Advanced Manufacturing of 1,3-Propanediol Gemini Surfactants via Solvent-Free Solid Acid Catalysis
The chemical industry is currently witnessing a paradigm shift towards greener synthesis methodologies, particularly in the realm of high-performance surfactants where environmental compliance and cost efficiency are paramount. Patent CN103435522A introduces a groundbreaking preparation method for 1,3-propanediol diisooctyl sodium sulfosuccinate, a novel gemini surfactant that exhibits superior surface activity compared to traditional single-chain analogues. This proprietary technology leverages a unique three-step reaction sequence that begins with the monoesterification of maleic anhydride and 1,3-propanediol, followed by a critical diesterification step utilizing a recyclable carbon-based solid acid catalyst. The final stage involves a conjugate addition sulfonation with sodium bisulfite to yield the target amphiphilic molecule. For R&D Directors and Procurement Managers seeking reliable specialty chemical suppliers, this patent represents a significant advancement in reducing the ecological footprint of surfactant manufacturing while maintaining rigorous quality standards. The elimination of hazardous solvents and the implementation of heterogeneous catalysis address key pain points in modern fine chemical production, offering a pathway to more sustainable and economically viable supply chains for personal care and industrial cleaning applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for gemini sulfosuccinate surfactants have historically been plagued by significant operational inefficiencies and environmental hazards that hinder large-scale commercial adoption. Conventional processes typically rely on homogeneous liquid acid catalysts, such as sulfuric acid or p-toluenesulfonic acid, which necessitate complex downstream neutralization and washing procedures to remove residual acidic species from the final product. Furthermore, these legacy methods often require the use of toxic organic solvents like toluene or acetone as water-carrying agents to drive the esterification equilibrium forward, introducing severe safety risks and requiring energy-intensive distillation units for solvent recovery. The presence of these solvents not only increases the overall production cost but also poses challenges in meeting stringent regulatory limits for residual volatiles in personal care ingredients. Additionally, the corrosive nature of liquid acids accelerates equipment degradation, leading to frequent maintenance downtime and potential metal contamination in the product stream. These cumulative factors result in a manufacturing process that is both economically burdensome and environmentally unsustainable, creating a pressing need for innovation in the sector of high-purity surfactant intermediates.
The Novel Approach
In stark contrast to these outdated methodologies, the novel approach detailed in the patent data utilizes a carbon-based solid acid catalyst derived from starch-immobilized p-toluenesulfonic acid, fundamentally transforming the reaction engineering landscape. This heterogeneous catalytic system allows the diesterification reaction to proceed efficiently under atmospheric pressure without the need for any auxiliary water-carrying solvents, thereby eliminating the risks associated with volatile organic compound emissions. The solid nature of the catalyst enables a simple filtration step for separation, allowing the catalytic material to be recovered and reused multiple times without significant loss of activity, which drastically reduces raw material consumption. By operating at moderate temperatures around 180°C and avoiding vacuum dehydration requirements, the process simplifies the reactor design and lowers the energy demand for heating and pressure control. This streamlined workflow not only enhances the overall yield by suppressing side reactions like ester hydrolysis but also ensures a lighter product color profile, which is critical for aesthetic applications in cosmetics and detergents. For supply chain heads, this translates to a more robust production capability with reduced dependency on complex utility infrastructure and hazardous waste management systems.
Mechanistic Insights into Carbon-Based Solid Acid Catalyzed Esterification
The core innovation of this synthesis lies in the mechanistic behavior of the carbon-based solid acid catalyst during the diesterification of 1,3-propanediol bismaleic acid monoester with iso-octanol. The catalyst functions by providing accessible proton donors on its solid surface, which activate the carbonyl group of the carboxylic acid moiety, facilitating nucleophilic attack by the hydroxyl group of the alcohol. Unlike liquid acids that disperse uniformly and require neutralization, the solid acid confines the catalytic activity to the interface, promoting selective esterification while minimizing unwanted polymerization or degradation of the unsaturated maleate double bonds. The immobilization of the active acid sites on a carbonized starch matrix enhances thermal stability and prevents leaching of acidic species into the reaction mixture, ensuring that the final product remains free from corrosive residues. This mechanism allows for precise control over the esterification rate, achieving conversion levels between 94% and 96% within a relatively short reaction timeframe of 3 hours. The ability to tune the acidity and pore structure of the carbon support offers further opportunities for optimizing reaction kinetics, making this catalytic system highly adaptable for various fatty alcohol chain lengths in surfactant design.
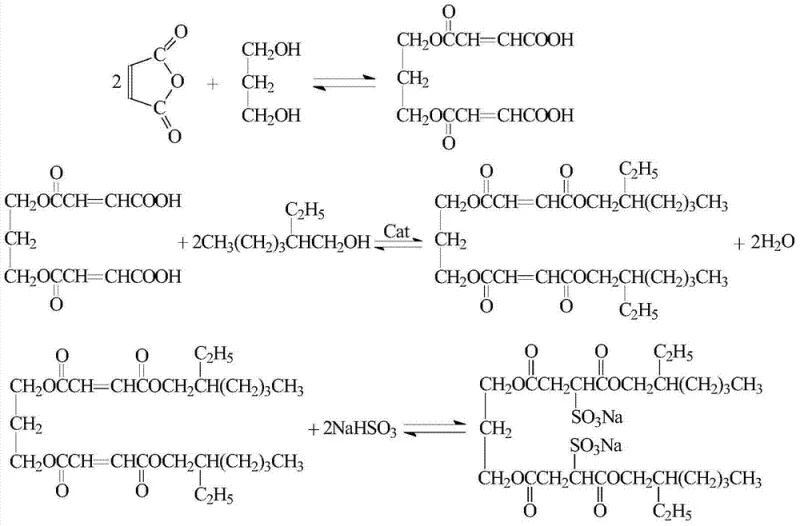
Furthermore, the subsequent sulfonation step employs a clever strategy where the monoester formed during the diesterification phase is neutralized to act as an intrinsic phase transfer catalyst, eliminating the need for external additives. This internal phase transfer mechanism facilitates the interaction between the organic diester phase and the aqueous sodium bisulfite solution, promoting efficient conjugate addition across the carbon-carbon double bond. The reaction proceeds via a Michael-type addition where the sulfite ion attacks the beta-carbon of the alpha,beta-unsaturated ester, forming the stable sulfonate head group characteristic of this gemini surfactant class. By controlling the molar ratio of sodium bisulfite to maleic anhydride at 1.00:1.05, the process ensures complete conversion while minimizing excess salt formation that could complicate purification. The absence of external phase transfer agents reduces the impurity profile significantly, resulting in a product with consistent critical micelle concentration (CMC) and surface tension properties. This level of mechanistic control is essential for R&D teams aiming to replicate performance benchmarks in formulation testing without batch-to-batch variability caused by residual catalysts or unreacted intermediates.
How to Synthesize 1,3-Propanediol Diisooctyl Sodium Sulfosuccinate Efficiently
The synthesis of this high-value gemini surfactant follows a meticulously optimized three-stage protocol designed to maximize yield while adhering to green chemistry principles. The process initiates with the monoesterification of maleic anhydride and 1,3-propanediol at 90°C, followed by the critical diesterification step where the carbon-based solid acid catalyst is introduced at 180°C under nitrogen protection. The final sulfonation is conducted at 104°C with aqueous sodium bisulfite to complete the molecular architecture. Detailed standardized synthetic steps, including precise temperature ramping rates, stirring speeds, and workup procedures, are outlined in the technical guide below to ensure reproducibility for process chemists.
- Conduct monoesterification of maleic anhydride and 1,3-propanediol at 90°C with a molar ratio of 2.1: 1.0 to form the bismaleic acid monoester intermediate.
- Perform diesterification with iso-octanol using 1% carbon-based solid acid catalyst at 180°C under atmospheric pressure, avoiding toxic solvents like toluene.
- Execute sulfonation by reacting the diester with sodium bisulfite at 104°C to yield the final gemini surfactant product with low toxicity and high surface activity.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this solvent-free, solid-acid catalyzed process offers substantial advantages in terms of total cost of ownership and supply chain resilience. The elimination of toxic solvents like toluene and acetone removes the need for expensive solvent recovery systems and reduces the regulatory burden associated with volatile organic compound (VOC) emissions, leading to significant operational expenditure savings. Moreover, the reusability of the carbon-based solid acid catalyst drastically cuts down on raw material costs compared to single-use liquid acids, while the simplified filtration process reduces labor and processing time. For supply chain managers, the ability to operate under atmospheric pressure lowers the capital investment required for specialized pressure-rated reactors, making it easier to scale production capacity to meet fluctuating market demands. The robustness of the process also ensures consistent product quality, reducing the risk of batch rejections and enhancing reliability for long-term contracts with major personal care brands. These factors collectively contribute to a more agile and cost-effective manufacturing model that can withstand raw material price volatility and regulatory changes.
- Cost Reduction in Manufacturing: The implementation of a reusable heterogeneous catalyst system fundamentally alters the cost structure by eliminating the recurring expense of purchasing fresh liquid acid catalysts for every batch. By avoiding the use of auxiliary solvents, the process removes the energy-intensive distillation steps required for solvent recovery, resulting in lower utility consumption and reduced carbon footprint. The simplified downstream processing, which relies on filtration rather than complex extraction and washing, further decreases labor costs and wastewater treatment fees. These cumulative efficiencies translate into a more competitive pricing model for the final surfactant product without compromising on purity or performance specifications.
- Enhanced Supply Chain Reliability: Operating under atmospheric pressure and moderate temperatures reduces the mechanical stress on production equipment, leading to longer asset life and fewer unplanned maintenance shutdowns. The absence of hazardous solvents simplifies logistics and storage requirements, mitigating risks associated with the transportation and handling of flammable materials. This operational simplicity ensures a more stable production schedule, allowing suppliers to maintain consistent inventory levels and meet tight delivery deadlines even during periods of high market demand. The reduced dependency on complex utility infrastructure also makes the manufacturing process more resilient to external disruptions, securing the supply continuity for downstream formulators.
- Scalability and Environmental Compliance: The green nature of this synthesis route aligns perfectly with increasingly stringent global environmental regulations, future-proofing the production facility against potential compliance issues. The lack of solvent emissions and the minimal generation of acidic wastewater simplify the permitting process for capacity expansion, enabling faster scale-up from pilot to commercial production volumes. The solid waste generated from the spent catalyst is non-hazardous and easier to dispose of or regenerate, further enhancing the sustainability profile of the manufacturing site. This environmental stewardship not only reduces liability but also strengthens the brand value for customers seeking eco-friendly ingredients for their sustainable product lines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel gemini surfactant, based on the specific advantages outlined in the patent literature. These insights are designed to clarify the operational benefits and performance characteristics for stakeholders evaluating this technology for integration into their supply chains. Understanding these details is crucial for making informed decisions about sourcing high-performance surfactants that meet both economic and environmental criteria.
Q: What are the environmental advantages of using carbon-based solid acid catalysts in surfactant production?
A: The use of carbon-based solid acid catalysts eliminates the need for corrosive liquid acids, thereby preventing equipment corrosion and simplifying the separation process through simple filtration. This technology significantly reduces wastewater treatment burdens and allows for catalyst reuse, aligning with green chemistry principles for sustainable surfactant manufacturing.
Q: How does the solvent-free process impact the purity profile of the final gemini surfactant?
A: By operating without auxiliary solvents like toluene or acetone, the process avoids residual solvent contamination in the final product. This solvent-free approach, combined with controlled esterification rates, minimizes side reactions and hydrolysis, resulting in a high-purity surfactant with consistent surface tension properties and reduced color formation.
Q: Is this synthesis route scalable for industrial production of specialty chemicals?
A: Yes, the process operates under atmospheric pressure and moderate temperatures, removing the need for complex vacuum dehydration systems. The robustness of the solid acid catalyst and the elimination of solvent recovery steps make this route highly amenable to commercial scale-up, ensuring reliable supply continuity for large-volume personal care applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Propanediol Diisooctyl Sodium Sulfosuccinate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into commercially viable products that meet the rigorous demands of the global personal care industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity 1,3-propanediol diisooctyl sodium sulfosuccinate that adheres to stringent purity specifications, supported by our rigorous QC labs that perform comprehensive testing on every batch. Our expertise in solid acid catalysis and solvent-free processing allows us to offer this advanced surfactant at a competitive price point while maintaining the highest standards of quality and consistency. By partnering with us, you gain access to a supply chain that is not only cost-effective but also aligned with the latest trends in green chemistry and sustainable manufacturing practices.
We invite you to collaborate with our technical procurement team to explore how this innovative synthesis route can optimize your formulation costs and enhance your product performance. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate the tangible benefits of switching to this superior manufacturing technology. Let us help you secure a reliable supply of high-performance gemini surfactants that will give your products a competitive edge in the marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
