Advanced Manufacturing of Crisaborole Key Intermediate via Selective Iodination and Miyaura Borylation
Introduction to Patent CN113214300A and Market Context
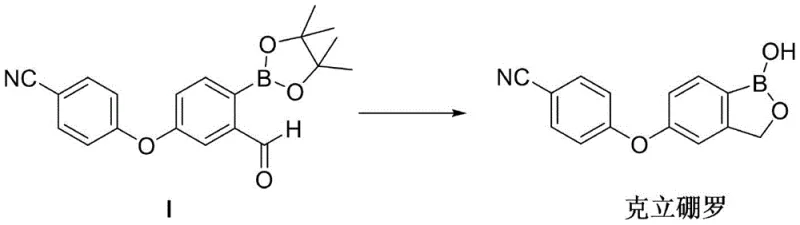
The pharmaceutical landscape for atopic dermatitis treatments has been significantly reshaped by the approval of Crisaborole (trade name Eucrisa), a non-steroidal phosphodiesterase-4 (PDE4) inhibitor. As demand for this active pharmaceutical ingredient (API) continues to grow globally, the efficiency of its supply chain relies heavily on the robustness of its key intermediates. Patent CN113214300A discloses a groundbreaking preparation method for the Crisaborole key intermediate, specifically the aldehyde-bearing boronic ester precursor (Compound I). This technology addresses critical bottlenecks in traditional synthesis, offering a pathway that balances high purity with industrial safety. By shifting away from hazardous halogenation reagents and cryogenic conditions, this innovation provides a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to secure long-term contracts with multinational generic and innovator companies.
The core of this technological advancement lies in a strategic three-step sequence that begins with the protection of m-hydroxybenzaldehyde, followed by selective iodination, etherification, and finally, a palladium-catalyzed Miyaura borylation. This approach not only circumvents the explosive risks associated with diazotization methods but also solves the regioselectivity issues plaguing direct bromination routes. For procurement strategists, understanding the nuances of this patent is essential, as it represents a shift towards greener, more cost-effective manufacturing protocols that align with modern ESG (Environmental, Social, and Governance) goals in the chemical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of boron-containing intermediates for Crisaborole has been fraught with significant technical and safety challenges. Traditional routes often relied on lithium-halogen exchange using n-butyllithium or tert-butyllithium, which necessitates ultra-low temperature reactions at -78°C under strictly anhydrous and oxygen-free conditions. These requirements impose a massive burden on capital expenditure for cryogenic reactors and inert gas systems, making the process economically unviable for large-scale production. Furthermore, alternative methods utilizing Grignard reagents, while slightly less sensitive, still operate at -20°C and struggle with moisture tolerance, leading to inconsistent batch quality.
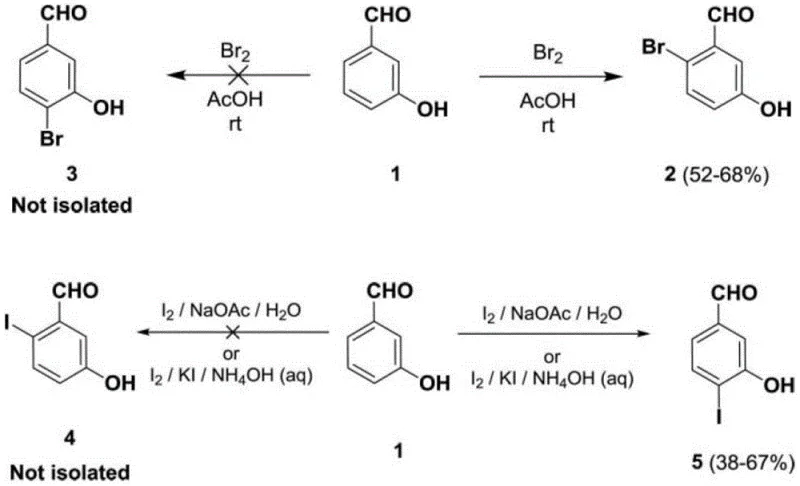
Another prevalent conventional approach involves direct bromination of m-hydroxybenzaldehyde. However, as illustrated in the side reaction analysis, direct halogenation often results in poor regioselectivity and dangerous side products. The use of elemental bromine introduces severe EHS hazards due to its corrosive nature and the generation of bromine-containing wastewater, which is costly to treat. Additionally, the unprotected aldehyde group in these traditional routes is prone to oxidation and self-condensation, drastically reducing the overall yield to often below 60%. These inefficiencies create substantial supply chain vulnerabilities, leading to potential shortages and price volatility for downstream API manufacturers.
The Novel Approach
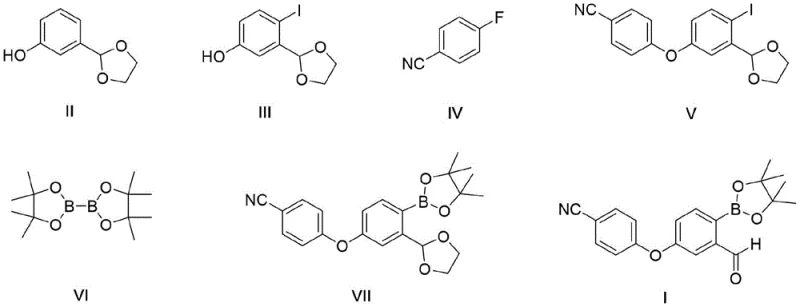
The methodology disclosed in CN113214300A offers a transformative solution by introducing an acetal protection strategy coupled with selective iodination. By first converting m-hydroxybenzaldehyde into its ethylene glycol acetal (Compound II), the reactive aldehyde moiety is masked, effectively preventing the self-condensation and oxidation side reactions that plague unprotected routes. This protection step is followed by a highly selective iodination using silver catalysts and solid iodine sources, which avoids the handling risks of liquid bromine. The resulting iodo-intermediate possesses a C-I bond with lower bond dissociation energy than a C-Br bond, making it significantly more reactive in the subsequent cross-coupling steps.
The culmination of this novel route is the Miyaura borylation reaction, which proceeds under mild thermal conditions (80-100°C) rather than cryogenic temperatures. This shift allows for the use of standard stainless steel reactors and simplifies the purification process, as the intermediate (Compound VII) can be deprotected in situ without isolation. This telescoped process design minimizes solvent usage and waste generation, directly contributing to cost reduction in API manufacturing. The overall synthetic pathway is depicted below, highlighting the streamlined progression from simple starting materials to the high-value boronic ester intermediate.
Mechanistic Insights into Selective Iodination and Miyaura Borylation
The success of this synthetic route hinges on the precise mechanistic control exerted during the iodination and borylation steps. In the initial functionalization, the use of silver trifluoroacetate or similar silver salts acts as a Lewis acid catalyst, activating the iodine source (such as KI or N-iodosuccinimide) towards electrophilic aromatic substitution. The acetal group at the meta-position exerts a specific electronic and steric influence that directs the iodine atom exclusively to the para-position relative to the hydroxyl group. This regioselectivity is crucial; unlike bromination, which can yield mixtures of isomers requiring difficult chromatographic separation, this iodination produces a single, high-purity isomer (Compound III) suitable for direct use in the next step.
Following the etherification with 4-fluorobenzonitrile, the Miyaura borylation step utilizes a palladium catalyst, typically [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium(II) (Pd(dppf)Cl2), to facilitate the transmetallation of the boron species. The mechanism involves the oxidative addition of the aryl iodide to the Pd(0) species, followed by ligand exchange with the bis(pinacolato)diboron reagent. The lower bond energy of the C-I bond compared to C-Br accelerates this oxidative addition step, allowing the reaction to proceed efficiently at 80-100°C. Finally, the acidic deprotection step hydrolyzes the acetal back to the aldehyde, yielding the target Compound I. This sequence ensures that impurities generated in earlier steps do not carry through, as the final crystallization or extraction effectively purifies the product.
How to Synthesize Crisaborole Key Intermediate Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly the choice of solvent and catalyst loading during the borylation step. The patent details specific embodiments where solvents like dioxane or toluene provide optimal solubility for the organic intermediates while maintaining the stability of the palladium catalyst. The process is designed to be robust, tolerating minor variations in stoichiometry without compromising the final purity profile. For process chemists looking to replicate or scale this technology, the following standardized workflow outlines the critical operational phases derived from the patent examples.
- Protect m-hydroxybenzaldehyde with ethylene glycol to form the acetal intermediate (Compound II), then perform selective iodination using silver catalysts to obtain Compound III.
- Condense the iodo-intermediate (III) with 4-fluorobenzonitrile (IV) under basic conditions to form the ether linkage (Compound V).
- Execute Miyaura borylation on Compound V using pinacol diboron and a palladium catalyst, followed by acidic deprotection to yield the final aldehyde intermediate (Compound I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits beyond mere technical feasibility. The shift from cryogenic lithiation to thermal borylation fundamentally alters the cost structure of production by eliminating the need for specialized low-temperature infrastructure and the associated high energy consumption. Furthermore, the replacement of hazardous liquid bromine with solid iodine salts simplifies logistics, storage, and regulatory compliance, thereby reducing the total cost of ownership for the manufacturing facility. These factors combine to create a more resilient supply chain capable of withstanding market fluctuations.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like n-butyllithium and elemental bromine leads to significant raw material cost savings. Additionally, the higher selectivity of the iodination step reduces the burden on downstream purification processes, minimizing solvent consumption and waste disposal fees. The ability to telescope the borylation and deprotection steps without isolating the unstable boronic ester intermediate further streamlines operations, reducing labor hours and equipment occupancy time, which translates to substantial operational expenditure (OPEX) reductions.
- Enhanced Supply Chain Reliability: By utilizing commercially available and stable starting materials such as m-hydroxybenzaldehyde and ethylene glycol, the process mitigates the risk of raw material shortages often associated with custom-synthesized complex bromides. The robustness of the reaction conditions (80-100°C) ensures consistent batch-to-batch reproducibility, a critical factor for maintaining uninterrupted API supply. This reliability allows pharmaceutical partners to plan their production schedules with greater confidence, reducing the need for excessive safety stock and freeing up working capital.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind. The avoidance of genotoxic nitrosamine impurities, which can form in DMF-based high-temperature condensation reactions used in older methods, simplifies the regulatory filing process. Moreover, the reduced generation of halogenated wastewater aligns with increasingly stringent environmental regulations, ensuring long-term operational continuity without the risk of shutdowns due to environmental non-compliance. This makes the technology future-proof for sustainable manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this method represents a superior choice for modern pharmaceutical manufacturing.
Q: Why is the iodination route preferred over bromination for this intermediate?
A: The iodination route utilizes C-I bonds which have lower bond energy than C-Br bonds, facilitating easier cleavage during the subsequent Miyaura borylation. Additionally, solid iodine sources are safer to transport and handle than liquid bromine, significantly reducing EHS risks and waste treatment costs associated with bromine-containing effluents.
Q: How does the acetal protection group improve the synthesis yield?
A: Protecting the aldehyde group as an acetal prevents unwanted side reactions such as self-condensation between the hydroxyl and aldehyde groups, or activation of the benzene ring by the aldehyde which leads to isomeric impurities. This protection strategy ensures higher regioselectivity and purity in the final product.
Q: What are the scalability advantages of this method compared to lithiation methods?
A: Unlike lithiation methods that require cryogenic conditions (-78°C) and strictly anhydrous environments, this Miyaura borylation route operates at moderate temperatures (80-100°C) in common solvents like dioxane or toluene. This eliminates the need for expensive cryogenic infrastructure and allows for safer, more robust commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Crisaborole Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team has extensively analyzed the pathways described in CN113214300A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity Crisaborole intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to ensure every batch complies with global pharmacopeial standards.
We invite you to collaborate with us to optimize your supply chain for dermatology APIs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive value for your organization.
