Scalable Synthesis of Isoflavone 7-O-Glucosides via Advanced Phase Transfer Catalysis for Global Markets
The pharmaceutical and functional food industries are constantly seeking robust synthetic routes for bioactive glycosides, particularly isoflavone derivatives known for their significant physiological benefits. Patent CN101450958B introduces a groundbreaking methodology for synthesizing isoflavone 7-O-glucosides, addressing critical challenges in regioselectivity and reaction stability that have long plagued conventional glycosylation techniques. This innovation leverages a sophisticated phase transfer catalysis system to achieve high synthesis rates under remarkably mild conditions, ensuring the structural integrity of the sensitive isoflavone core while maximizing yield. For R&D directors and procurement specialists, this represents a pivotal shift towards more reliable and cost-efficient manufacturing processes for high-value nutritional and pharmaceutical intermediates. The ability to produce these complex molecules with minimal byproduct formation directly translates to simplified downstream purification and enhanced overall process economics.
Furthermore, the strategic implementation of this technology supports the growing demand for high-purity isoflavone glycosides in the global market, where consistency and safety are paramount. By utilizing a fractional addition strategy for the glycosyl donor, the method effectively controls the reaction kinetics, preventing the accumulation of unstable intermediates that could compromise product quality. This level of control is essential for meeting the stringent regulatory standards required for ingredients destined for human consumption. As a reliable isoflavone glycoside supplier, understanding the nuances of such patented methodologies allows us to offer superior technical support and customized synthesis solutions to our partners. The transition from laboratory-scale discovery to commercial viability is seamless with this approach, offering a competitive edge in cost reduction in nutritional ingredient manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of flavonoid and isoflavone glycosides has historically relied on methods such as the Koenigs-Knorr reaction or direct esterification using trichloroacetimidates, which often suffer from significant drawbacks in terms of selectivity and operational safety. A primary concern in these conventional pathways is the lack of regioselectivity, as isoflavone aglycones typically possess multiple hydroxyl groups that can compete for glycosylation, leading to complex mixtures of isomers that are difficult and expensive to separate. Additionally, the glycosyl donors used in these classic methods, particularly halogenated sugars, are prone to rapid hydrolysis in the presence of the strong bases often required to drive the reaction, resulting in substantial material loss and reduced overall yields. The harsh reaction conditions, including elevated temperatures and strongly alkaline environments, can also induce degradation of the sensitive isoflavone skeleton, generating unknown impurities that complicate the safety profile of the final product. These technical limitations not only increase the cost of goods sold due to low efficiency but also pose significant risks for commercial scale-up of complex polymer additives and fine chemicals where purity is non-negotiable.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a mild phase transfer catalysis system that elegantly circumvents the pitfalls of traditional strong-base methodologies. By employing a specific combination of sodium bicarbonate and potassium chloride alongside the catalyst tris(3,6-dioxaheptyl)amine (TDA-1), the reaction environment remains sufficiently alkaline to activate the phenolic nucleophile without triggering the rapid decomposition of the glycosyl donor. This delicate balance allows for the selective glycosylation of the 7-hydroxyl position, which is the most biologically relevant site for isoflavone activity, while leaving other potential reaction sites untouched. The fractional addition of the 1-bromo-2,3,4,6-O-tetraacetyl-alpha-D-glucopyranose donor further optimizes the reaction stoichiometry, ensuring that the concentration of the reactive species is maintained at an ideal level to maximize conversion while minimizing side reactions. This methodological refinement results in a process that is not only safer and more environmentally friendly but also delivers consistently high yields that are critical for reducing lead time for high-purity bioactive compounds in a fast-paced supply chain.
Mechanistic Insights into TDA-1 Catalyzed Regioselective Glycosylation
The core of this synthetic breakthrough lies in the mechanistic action of the phase transfer catalyst TDA-1, which facilitates the transport of the phenoxide anion from the aqueous phase into the organic phase where the glycosylation occurs. In this biphasic system, the sodium bicarbonate serves as a mild base to deprotonate the 7-hydroxyl group of the isoflavone aglycone, generating a nucleophilic phenoxide ion that is then complexed by the lipophilic TDA-1 cation. This ion pair is soluble in the organic solvent, typically dichloromethane, bringing the nucleophile into close proximity with the glycosyl bromide donor dissolved in the same phase. The steric and electronic properties of TDA-1 are crucial, as they help to orient the reactants in a manner that favors the formation of the beta-anomer, which is the naturally occurring and biologically active configuration of isoflavone glycosides. This stereocontrol is vital for ensuring the efficacy of the final product, as the alpha-anomer often lacks the desired physiological activity and is considered an impurity in pharmaceutical applications.
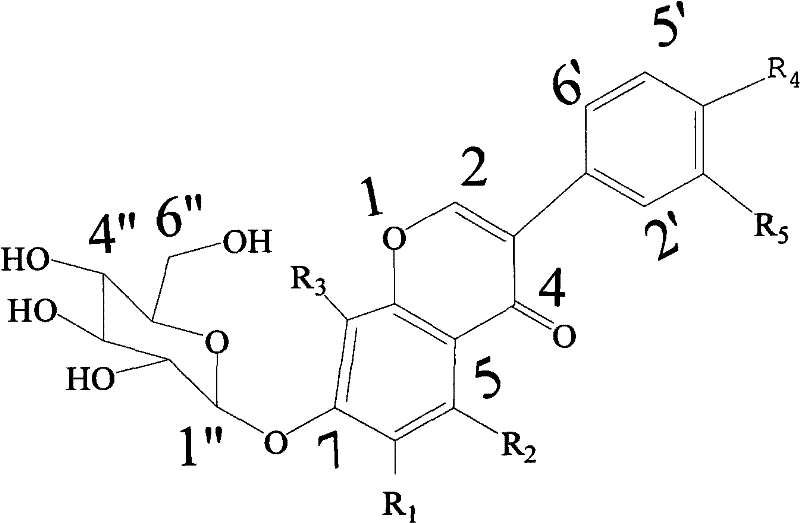
Beyond stereocontrol, the reaction conditions are meticulously designed to preserve the stability of the acetyl-protected sugar moiety throughout the transformation. The use of a mild alkaline medium prevents the saponification of the acetyl groups during the glycosylation step, which would otherwise lead to the formation of partially deprotected species that are difficult to purify. Following the coupling reaction, the deprotection step is carried out separately in methanol using ammonia, allowing for precise control over the removal of the acetyl groups without affecting the glycosidic bond. This two-stage strategy ensures that the final isoflavone 7-O-glucoside is obtained with exceptional purity, free from the complex impurity profiles often associated with one-pot synthesis attempts. For R&D teams, understanding this mechanism provides confidence in the reproducibility of the process and the ability to troubleshoot potential deviations during technology transfer to larger reactors.
How to Synthesize Isoflavone 7-O-Glucoside Efficiently
Implementing this synthesis route requires careful attention to the preparation of the reaction phases and the timing of reagent addition to maintain the optimal kinetic profile described in the patent. The process begins with the dissolution of the 7-hydroxyisoflavone aglycone in an aqueous solution containing equimolar potassium chloride and sodium bicarbonate, which establishes the necessary ionic strength and pH buffer for the phase transfer event. Once the aqueous phase is prepared, the organic phase containing the glycosyl donor and the TDA-1 catalyst is introduced, with the donor being added in two distinct fractions to manage the exotherm and reaction rate effectively. Maintaining the temperature strictly between 40-45°C is critical during this phase to ensure complete conversion while avoiding thermal degradation of the reactants. Detailed standardized synthesis steps see the guide below for exact parameters and workup procedures.
- Dissolve 7-hydroxyisoflavone in sodium bicarbonate solution with potassium chloride and TDA-1 catalyst, then add tetraacetyl-glucopyranose bromide fractionally at 40-45°C.
- Purify the resulting 7-O-beta-D-tetraacetylglucose isoflavone intermediate using silica gel chromatography.
- Deacetylate the intermediate in methanol with ammonia solution to yield the final high-purity isoflavone 7-O-glucoside.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis method offers profound advantages that directly address the pain points of procurement managers and supply chain heads in the fine chemical sector. The elimination of harsh reagents and the use of mild reaction conditions significantly reduce the safety hazards associated with manufacturing, lowering insurance costs and simplifying regulatory compliance for facility operations. Moreover, the high selectivity of the reaction minimizes the generation of waste streams and reduces the burden on wastewater treatment systems, aligning with modern environmental sustainability goals that are increasingly important for corporate social responsibility reporting. The simplicity of the post-treatment process, which involves standard extraction and crystallization techniques, means that the technology can be implemented in existing multipurpose plants without requiring specialized or expensive equipment upgrades. These factors combine to create a robust supply chain reliability that ensures consistent delivery of high-quality materials to downstream customers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the high yield and the use of cost-effective, commercially available starting materials that do not require custom synthesis. By avoiding the use of expensive heavy metal catalysts or exotic reagents, the raw material costs are kept low, and the absence of toxic metals eliminates the need for costly scavenging steps to meet residual metal specifications. The high conversion rate means that less raw material is wasted, and the simplified purification process reduces the consumption of solvents and silica gel, further driving down the variable costs per kilogram. These efficiencies allow for substantial cost savings that can be passed on to customers or reinvested into further process optimization and capacity expansion.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents such as sodium bicarbonate and potassium chloride ensures that the supply chain is not vulnerable to the volatility often seen with specialized or hazardous chemicals. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by minor variations in raw material quality or environmental conditions, providing a predictable output rate. This stability is crucial for maintaining long-term contracts with major pharmaceutical and nutraceutical companies who require guaranteed availability of key intermediates to support their own production lines. Consequently, partnering with a manufacturer utilizing this technology mitigates the risk of supply shortages and ensures business continuity for all stakeholders involved in the value chain.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is straightforward due to the absence of extreme pressures or temperatures that typically complicate reactor design and operation. The mild nature of the chemistry allows for the use of standard glass-lined or stainless steel reactors, facilitating rapid scale-up to meet increasing market demand without significant capital expenditure. Furthermore, the aqueous workup and the ability to recover and recycle solvents contribute to a greener manufacturing footprint, helping companies meet stringent environmental regulations and sustainability targets. This alignment with green chemistry principles not only reduces disposal costs but also enhances the brand reputation of the end-product in markets that prioritize eco-friendly sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of isoflavone 7-O-glucosides using this advanced phase transfer catalysis method. These insights are derived directly from the patent data and practical experience in scaling similar glycosylation reactions for industrial clients. Understanding these details helps stakeholders make informed decisions about integrating this technology into their supply chains and product development pipelines. We encourage technical teams to review these points to fully appreciate the value proposition of this synthetic route.
Q: How does this method improve regioselectivity compared to traditional Koenigs-Knorr reactions?
A: This method utilizes a specific phase transfer catalyst system (TDA-1/KCl/NaHCO3) that selectively targets the 7-hydroxyl group on the isoflavone aglycone, minimizing side reactions at other phenolic positions which are common in harsher traditional methods.
Q: What are the stability advantages of using the tetraacetyl protected sugar donor?
A: Using 1-bromo-2,3,4,6-O-tetraacetyl-alpha-D-glucopyranose prevents premature hydrolysis of the glycosyl donor in the alkaline medium, ensuring higher conversion rates and reducing the formation of hydrolytic impurities.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process operates at mild temperatures (40-45°C) and uses readily available reagents with simple aqueous workups, making it safe, cost-effective, and highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoflavone 7-O-Glucoside Supplier
At NINGBO INNO PHARMCHEM, we leverage deep technical expertise to transform patented innovations like CN101450958B into commercial realities that drive value for our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity observed in the lab are faithfully reproduced at an industrial scale. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and quality of every batch before it leaves our facility. This commitment to excellence ensures that the isoflavone derivatives we supply meet the exacting standards required for pharmaceutical and high-end nutraceutical applications, providing you with a foundation of trust and reliability.
We invite you to collaborate with us to explore how this efficient synthesis route can optimize your specific product requirements and cost structures. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals for high-purity isoflavone derivatives. Let us be your partner in navigating the complexities of fine chemical manufacturing and securing a competitive advantage in the marketplace.
