Advanced Manufacturing of Metal-Free Near-Infrared Organic Luminescent Materials for Next-Gen Displays
Introduction to Breakthrough NIR Organic Luminescent Technology
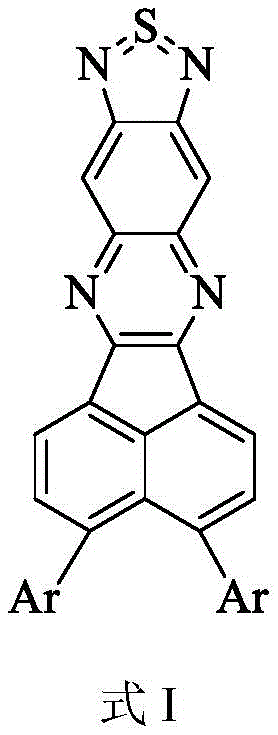
The rapid evolution of optoelectronic materials has placed significant emphasis on the development of efficient near-infrared (NIR) organic light-emitting diodes (OLEDs), driven by their critical applications in optical communication, infrared detection, and information security displays. Patent CN111574538A discloses a novel D-A type near-infrared organic luminescent material that addresses the longstanding challenges of low efficiency and limited wavelength availability in metal-free systems. This technology introduces a rigid large-plane naphthothiazoloquinoxaline structure as an electron acceptor, strategically combined with high HOMO energy level electron donors to optimize molecular orbital separation. For R&D directors and procurement specialists, this represents a pivotal shift away from expensive heavy metal phosphors toward sustainable, high-performance organic alternatives that promise both technical superiority and supply chain resilience.
The significance of this invention lies in its ability to achieve thermally activated delayed fluorescence (TADF), theoretically enabling 100% internal quantum efficiency without the need for scarce noble metals like platinum or iridium. By enhancing intermolecular torque through specific linking positions, the material effectively reduces the overlap between HOMO and LUMO molecular orbitals, a key mechanism for harvesting both singlet and triplet excitons. This patent not only provides a series of structurally diverse compounds but also outlines a robust preparation method characterized by readily available raw materials, simple reaction control, and high product purity, making it an attractive candidate for commercial scale-up in the competitive landscape of display and optoelectronic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of high-efficiency NIR OLEDs has been heavily reliant on phosphorescent heavy metal complexes, particularly those based on platinum(II) and iridium(III). While these materials have demonstrated impressive external quantum efficiencies exceeding 24%, their widespread adoption is severely hindered by the exorbitant cost and geological scarcity of the constituent noble metals. For procurement managers, the volatility in the supply of platinum group metals introduces significant financial risk and potential production bottlenecks. Furthermore, the environmental burden associated with mining and refining these metals, coupled with the complex waste treatment required for heavy metal residues during manufacturing, poses substantial regulatory and sustainability challenges for modern chemical enterprises aiming for green production standards.
Beyond economic and environmental concerns, conventional metal-based emitters often suffer from concentration quenching and stability issues in the solid state, limiting their operational lifetime in practical devices. The reliance on complex coordination chemistry also restricts the tunability of emission wavelengths, making it difficult to precisely target the specific near-infrared bands required for advanced telecommunications and sensing applications. Consequently, the industry has been actively seeking metal-free alternatives that can match the performance of phosphorescent complexes while offering the processing advantages and cost structures typical of purely organic semiconductors, a gap that previous generations of fluorescent materials failed to bridge due to their inherent limitation of utilizing only singlet excitons.
The Novel Approach
The methodology described in patent CN111574538A offers a transformative solution by employing a donor-acceptor (D-A) molecular architecture that facilitates efficient reverse intersystem crossing (RISC) through thermally activated delayed fluorescence. By constructing a rigid naphthothiazoloquinoxaline core as the electron acceptor and coupling it with strong electron-donating groups, the inventors have created a system where the energy gap between singlet and triplet states is minimized. This design allows for the up-conversion of non-radiative triplet excitons into radiative singlet states, effectively breaking the 25% efficiency limit of traditional fluorescence without incurring the cost penalty of heavy metals. The synthetic route is designed for scalability, utilizing standard cross-coupling and condensation reactions that are well-understood in industrial organic synthesis.
From a manufacturing perspective, this approach simplifies the supply chain by eliminating the need for specialized organometallic catalysts containing precious metals in the final product structure. The use of commercially available starting materials such as 4,5-dinitro-1,2-aminobenzene and 5,6-dibromoacenaphthylene ensures a stable and cost-effective feedstock base. Moreover, the reaction conditions are relatively mild, typically ranging from 60°C to 110°C, which reduces energy consumption and equipment stress compared to high-temperature pyrolysis methods. The resulting materials exhibit excellent thermal stability and emit in the desired near-infrared region, validating the strategy of using structural rigidity and electronic modulation to achieve high-performance optoelectronic properties in a metal-free framework.
Mechanistic Insights into Naphthothiazoloquinoxaline-Based TADF Synthesis
The core of this technological advancement is the precise construction of the naphthothiazoloquinoxaline moiety, which serves as a rigid, planar electron-accepting unit essential for stabilizing the excited states. The synthesis begins with the cyclization of 4,5-dinitro-1,2-aminobenzene using thionyl chloride, followed by a reduction step with iron powder to generate the diamino intermediate. This sequence is critical for establishing the heterocyclic framework that defines the electronic properties of the acceptor. Subsequently, the electron-deficient nature of this core is leveraged in a condensation reaction with electron-rich precursors, creating the push-pull electronic distribution necessary for TADF activity. The strategic placement of substituents allows for fine-tuning of the HOMO-LUMO gap, directly influencing the emission wavelength and ensuring it falls within the near-infrared spectrum.
Furthermore, the integration of strong electron donors via Suzuki-Miyaura cross-coupling reactions introduces the necessary steric bulk to prevent excessive aggregation, which can lead to quenching in the solid state. The patent details a multi-step pathway where intermediates are carefully purified to ensure high final product quality, a crucial factor for device performance. The reaction mechanism involves the formation of carbon-carbon bonds between the brominated acenaphthylene derivative and boronate-functionalized donors, catalyzed by palladium complexes. This step is pivotal for attaching the diverse array of donor groups shown in the patent, allowing for a library of materials with varying electronic characteristics. The final condensation step locks the D-A structure together, completing the conjugated system responsible for the material's luminescent properties.
How to Synthesize Naphthothiazoloquinoxaline Derivatives Efficiently
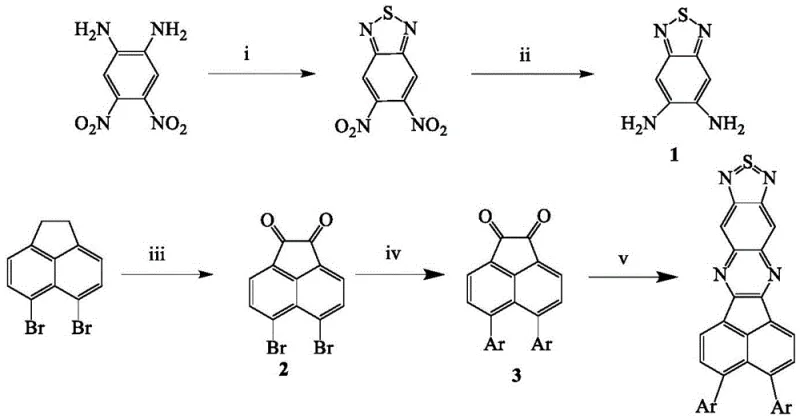
The preparation of these high-performance NIR materials follows a logical four-step sequence that balances yield with purity, making it suitable for both laboratory optimization and pilot-scale production. The process initiates with the formation of the heterocyclic acceptor core, followed by the functionalization of the acenaphthylene backbone, and concludes with the assembly of the final D-A architecture. Each step utilizes standard organic transformation techniques, ensuring that the protocol can be readily adopted by existing chemical manufacturing facilities without the need for exotic equipment. The detailed standardized synthesis steps are outlined in the guide below, providing a clear roadmap for replicating the high yields and purity levels reported in the patent data.
- Synthesize Intermediate 1 by reacting 4,5-dinitro-1,2-aminobenzene with thionyl chloride followed by reduction with iron powder.
- Prepare Intermediate 2 via oxidation of 5,6-dibromoacenaphthylene using chromium trioxide in acetic anhydride.
- Perform Suzuki coupling between Intermediate 2 and boronate-functionalized electron donors to form Intermediate 3.
- Condense Intermediate 1 and Intermediate 3 in glacial acetic acid and chloroform to obtain the final D-A type TADF material.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this metal-free NIR material offers compelling economic and logistical benefits that extend beyond simple raw material costs. The elimination of platinum and iridium from the molecular structure removes the exposure to volatile precious metal markets, stabilizing the cost of goods sold over long-term production contracts. Additionally, the synthetic pathway relies on commodity chemicals such as toluene, acetic acid, and dichloromethane, which are widely available globally, reducing the risk of supply disruptions associated with specialized reagents. The simplicity of the purification process, primarily involving silica gel column chromatography with common solvent systems, further streamlines operations and minimizes the need for complex downstream processing infrastructure.
- Cost Reduction in Manufacturing: The absence of expensive noble metal catalysts in the final product structure significantly lowers the material cost per gram, a critical metric for high-volume display manufacturing. By utilizing iron powder for reduction and standard palladium catalysts for coupling which can potentially be recovered, the process avoids the high capital lock-up associated with precious metal inventory. The moderate reaction temperatures (60-110°C) also contribute to lower energy utility costs compared to high-energy processes, resulting in substantial overall cost savings in electronic chemical manufacturing without compromising on the quality or performance of the emitted light.
- Enhanced Supply Chain Reliability: The reliance on readily available organic building blocks such as dinitroaminobenzene and dibromoacenaphthylene ensures a robust and diversified supply base. Unlike rare earth elements or platinum group metals which are geographically concentrated and subject to geopolitical trade restrictions, these organic precursors can be sourced from multiple global suppliers, enhancing supply continuity. The straightforward synthesis steps reduce the complexity of the manufacturing workflow, minimizing the potential for batch failures and ensuring consistent delivery schedules for downstream OLED device manufacturers who require reliable volumes of high-purity emissive materials.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, as demonstrated by the use of standard reaction vessels and workup procedures like filtration and extraction. The avoidance of heavy metals in the final product simplifies waste management and disposal, aligning with increasingly stringent environmental regulations regarding hazardous substances in electronics. This eco-friendly profile not only reduces compliance costs but also enhances the brand value of the end-products, appealing to consumers and regulators who prioritize sustainability in the supply chain of advanced display technologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this D-A type near-infrared organic luminescent technology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of adopting this new material class for next-generation optoelectronic applications. Understanding these details is essential for stakeholders evaluating the integration of metal-free TADF materials into their product portfolios.
Q: What are the advantages of this metal-free NIR material over traditional phosphorescent complexes?
A: Unlike traditional platinum or iridium-based phosphorescent complexes which suffer from high costs and resource scarcity, this metal-free D-A type material utilizes abundant organic raw materials, significantly reducing production costs and supply chain risks while maintaining high thermal stability and emission efficiency.
Q: How does the molecular structure enhance device performance?
A: The material features a rigid large-plane naphthothiazoloquinoxaline electron acceptor combined with strong electron donors. This D-A structure enhances intermolecular torque and effectively separates HOMO/LUMO energy levels, facilitating thermally activated delayed fluorescence (TADF) for higher quantum efficiency.
Q: Is the synthesis process suitable for large-scale commercial production?
A: Yes, the preparation process involves standard organic reactions such as Suzuki coupling and condensation under mild conditions (60-110°C). The use of common solvents like toluene and acetic acid, along with easy product separation via column chromatography, makes it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable NIR OLED Material Supplier
As a leading CDMO expert in the fine chemical sector, NINGBO INNO PHARMCHEM is uniquely positioned to support the commercialization of these advanced naphthothiazoloquinoxaline derivatives. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for high-performance electronic materials, guaranteeing that every batch delivered meets the exacting standards of the global display industry.
We invite you to collaborate with our technical team to explore how this cost-effective and high-efficiency material can enhance your product offerings. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and innovation in your supply chain for high-purity organic luminescent materials.
