Advanced Synthesis of Degradable Photoresist Resin Monomers for High-Resolution Lithography
Advanced Synthesis of Degradable Photoresist Resin Monomers for High-Resolution Lithography
The semiconductor industry continuously demands higher resolution and precision in photolithography processes to manufacture increasingly complex integrated circuits. Patent CN111138287A introduces a groundbreaking approach to synthesizing photoresist resin monomers derived from hexahydro-1H-indene-1, 3(2H)-diketone. This innovation addresses critical limitations in current photoresist technologies by utilizing a unique molecular architecture that facilitates main-chain degradation upon exposure. The structural general formula of this novel resin monomer, designated as Formula I, features a robust bicyclic core substituted with polymerizable groups, enabling the formation of three-dimensional network structures rather than simple linear chains. 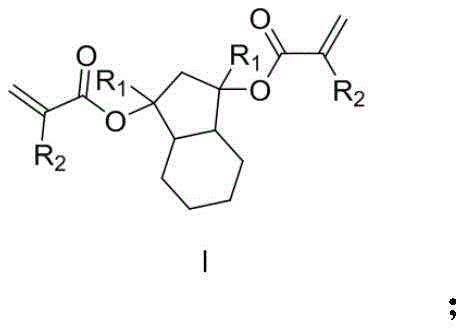 As a reliable photoresist resin monomer supplier, understanding the nuances of this chemistry is essential for delivering materials that meet the rigorous standards of modern electronic chemical manufacturing.
As a reliable photoresist resin monomer supplier, understanding the nuances of this chemistry is essential for delivering materials that meet the rigorous standards of modern electronic chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional photoresist resins are typically formed by copolymerizing monomers that possess only a single acid-sensitive group, resulting in linear polymer chains. While these materials have served the industry for years, they face significant challenges as feature sizes shrink to the nanometer scale. In conventional systems, the solubility difference between exposed and unexposed areas relies solely on the deprotection of side groups, leaving the polymer backbone intact. This limitation often leads to insufficient contrast in dissolution rates, causing issues such as line edge roughness and reduced pattern fidelity. Furthermore, the inability of the main chain to break down limits the extent of solubility change, thereby capping the achievable resolution of the photoetching patterns. These structural constraints necessitate the development of new monomer architectures that can fundamentally alter the dissolution mechanism during the development process.
The Novel Approach
The technology disclosed in CN111138287A offers a transformative solution by introducing a degradable resin monomer capable of forming crosslinked, three-dimensional networks. By incorporating two unsaturated carbon-carbon double bonds into the monomer structure, the resulting polymer resin exhibits superior etching resistance prior to exposure. Crucially, upon exposure to light and subsequent acid generation by the photoacid generator, the (methyl) acrylate groups on the main chain are cleaved under acidic conditions. This cleavage causes the main chain of the polymer resin to break, generating lower molecular weight fragments that dissolve much more readily in the developing solution. This mechanism dramatically increases the difference in dissolution speeds before and after exposure, directly translating to improved edge roughness and significantly enhanced resolution of the photoresist patterns. 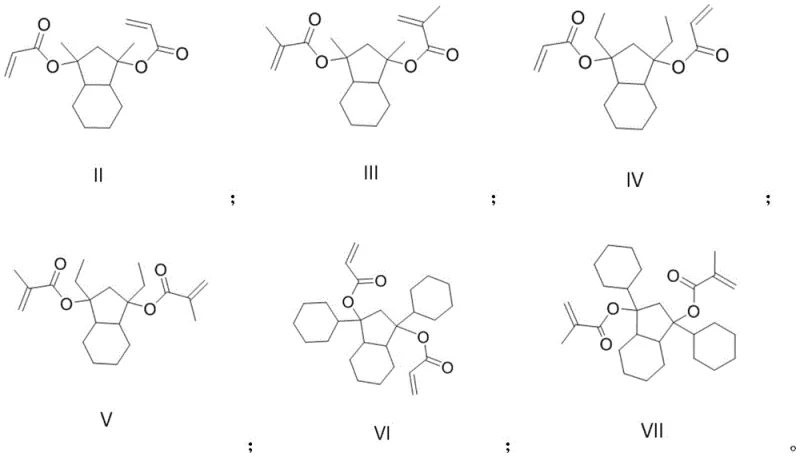 This approach represents a paradigm shift in cost reduction in electronic chemical manufacturing by improving yield and performance without necessitating entirely new processing equipment.
This approach represents a paradigm shift in cost reduction in electronic chemical manufacturing by improving yield and performance without necessitating entirely new processing equipment.
Mechanistic Insights into Grignard Addition and Esterification
The synthesis of these advanced monomers relies on a robust two-step chemical pathway that leverages well-understood organic transformations to ensure high purity and reproducibility. The process begins with the nucleophilic addition of a Grignard reagent to the carbonyl groups of hexahydro-1H-indene-1, 3(2H)-diketone. This reaction, conducted under inert gas protection in anhydrous ether at controlled temperatures between 0°C and 30°C, converts the diketone into a diol intermediate. The choice of Grignard reagent, whether alkyl or cycloalkyl, allows for precise tuning of the R1 substituent, providing flexibility in the final physical properties of the resin. Following the Grignard addition, the reaction mixture is quenched with water, and the intermediate is isolated through extraction and purification techniques such as column chromatography to remove magnesium salts and unreacted starting materials.
The second critical stage involves the esterification of the diol intermediate with acryloyl chloride or methacryloyl chloride. This step introduces the polymerizable vinyl groups necessary for crosslinking and subsequent degradation. Conducted in solvents like anhydrous tetrahydrofuran, toluene, or chloroform at temperatures ranging from 0°C to 70°C, the reaction utilizes bases such as triethylamine or pyridine to scavenge the generated hydrochloric acid. 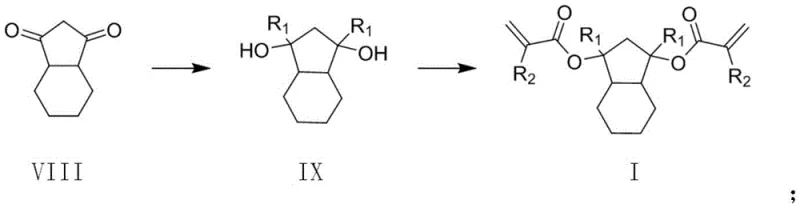 The mechanistic precision of this esterification ensures that both hydroxyl groups are converted efficiently, maximizing the functionality of the monomer. This high degree of conversion is vital for achieving the desired three-dimensional network structure in the final photoresist, ensuring consistent performance across different batches and facilitating the commercial scale-up of complex electronic chemicals.
The mechanistic precision of this esterification ensures that both hydroxyl groups are converted efficiently, maximizing the functionality of the monomer. This high degree of conversion is vital for achieving the desired three-dimensional network structure in the final photoresist, ensuring consistent performance across different batches and facilitating the commercial scale-up of complex electronic chemicals.
How to Synthesize Photoresist Resin Monomer Efficiently
The practical implementation of this synthesis route requires careful attention to reaction conditions, particularly moisture control during the Grignard step and temperature management during esterification. The patent provides detailed experimental examples demonstrating the feasibility of producing various derivatives, such as those with methyl, ethyl, or cyclohexyl substituents. For instance, the preparation of the methyl-substituted monomer involves generating methyl magnesium bromide in situ, followed by addition to the diketone to yield the intermediate in high yield. Subsequent reaction with acryloyl chloride affords the final monomer with excellent purity suitable for semiconductor applications.
- Perform a Grignard reaction between hexahydro-1H-indene-1, 3(2H)-diketone and an alkyl or cycloalkyl Grignard reagent in anhydrous ether at 0-30°C to form a diol intermediate.
- Quench the reaction with water, separate the organic layer, and purify the intermediate via column chromatography.
- React the purified intermediate with acryloyl chloride or methacryloyl chloride in a solvent like THF or toluene with a base (triethylamine) at 0-70°C to obtain the final resin monomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis route offers distinct strategic advantages rooted in process efficiency and raw material accessibility. The reliance on standard organic reactions eliminates the need for expensive transition metal catalysts or specialized high-pressure equipment, which are often bottlenecks in fine chemical production. By utilizing widely available reagents like magnesium, alkyl halides, and acid chlorides, the supply chain becomes more resilient to fluctuations in the availability of exotic precursors. This simplification of the bill of materials directly contributes to substantial cost savings in the overall manufacturing process, allowing for more competitive pricing structures without compromising on the high-purity specifications required for electronic grade chemicals.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes the necessity for costly metal scavenging steps and reduces the environmental burden associated with heavy metal waste disposal. Furthermore, the high yields reported in the patent examples indicate an efficient atom economy, minimizing raw material waste and lowering the cost per kilogram of the final product. The use of common solvents like ether and THF, which can be recovered and recycled in large-scale operations, further enhances the economic viability of the process. These factors collectively drive down the operational expenditure, making high-performance photoresist monomers more accessible for mass production.
- Enhanced Supply Chain Reliability: The starting material, hexahydro-1H-indene-1, 3(2H)-diketone, and the requisite Grignard reagents are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate standard laboratory and plant equipment, ensures that production can be scaled rapidly to meet surging demand in the semiconductor sector. This flexibility allows manufacturers to maintain continuous supply lines even during periods of market volatility, ensuring that downstream photoresist formulators receive their critical intermediates on schedule.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-ton commercial production without fundamental changes to the chemistry. The absence of toxic heavy metals simplifies wastewater treatment and aligns with increasingly stringent environmental regulations governing chemical manufacturing. Additionally, the ability to produce degradable monomers supports the broader industry trend towards more sustainable lithography processes, as the resulting photoresists offer better performance with potentially lower usage rates due to their enhanced sensitivity and resolution.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these novel photoresist monomers. Understanding these details is crucial for R&D teams evaluating the integration of this technology into their existing formulations.
Q: What is the primary advantage of this new photoresist monomer structure?
A: Unlike traditional linear polymers, this monomer contains two unsaturated carbon-carbon double bonds that form a three-dimensional network. Upon exposure, the main chain breaks under acidic conditions, significantly increasing solubility differences and improving pattern resolution.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the process utilizes standard organic synthesis techniques such as Grignard reactions and esterification, which are well-established and easily scalable from laboratory to commercial tonnage without requiring exotic catalysts.
Q: What are the key raw materials required for this synthesis?
A: The core starting material is hexahydro-1H-indene-1, 3(2H)-diketone, which reacts with alkyl or cycloalkyl Grignard reagents and subsequently with acryloyl or methacryloyl chloride.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Photoresist Resin Monomer Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and manufacturing for the electronic chemicals sector, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the rigorous purity specifications and stringent QC labs required for semiconductor-grade materials, ensuring that every batch of photoresist monomer meets the highest international standards. We understand the critical nature of supply continuity in the chip manufacturing supply chain and are committed to delivering consistent quality and reliability to our global partners.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next-generation photoresist formulations. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and production goals, ensuring a seamless transition from laboratory innovation to industrial reality.
