Advanced Synthetic Route for N-Methyl-D-Threonine Enables Scalable Peptide Drug Manufacturing
Introduction to Novel N-Methyl-D-Threonine Synthesis Technology
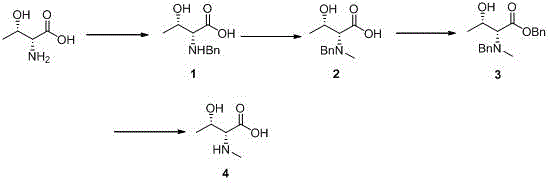
The pharmaceutical industry's relentless pursuit of potent antibacterial peptides and advanced polypeptide therapeutics has placed a premium on high-quality non-proteinogenic amino acid building blocks. Specifically, N-methyl-D-threonine (CAS: 2812-27-3) has emerged as a critical monomer for the synthesis of complex cyclic depsipeptides such as Petriellin A, which exhibits significant antifungal and anticancer properties. Addressing the historical challenges associated with the scalable production of this chiral intermediate, patent CN113773214A discloses a robust four-step synthetic methodology that fundamentally re-engineers the protection and deprotection strategy. This innovative approach leverages a benzyl-based protection group system coupled with optimized reductive amination conditions to overcome the severe purification bottlenecks inherent in previous art. By systematically modifying reaction parameters and reagent selection, the disclosed process achieves exceptional yields while maintaining stringent stereochemical integrity, thereby offering a viable pathway for industrial-scale manufacturing of this high-value API intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for N-methylated amino acids often suffer from significant downstream processing inefficiencies that render them economically unviable for large-scale production. A primary technical hurdle identified in prior art is the extreme water solubility and high polarity of key intermediates, particularly the N-methylated species prior to final deprotection. This physicochemical property makes liquid-liquid extraction notoriously difficult, often forcing manufacturers to rely on preparative column chromatography for purification. In a commercial context, column chromatography is a major bottleneck; it consumes vast quantities of silica gel and organic solvents, drastically increases cycle times, and limits batch sizes, effectively preventing the transition from laboratory grams to metric ton production. Furthermore, conventional methods frequently employ suboptimal bases or protecting group strategies that lead to racemization or incomplete reactions, resulting in complex impurity profiles that require additional, costly remediation steps to meet pharmaceutical grade specifications.
The Novel Approach
The methodology outlined in patent CN113773214A introduces a strategic paradigm shift by incorporating a temporary benzyl ester protection step that fundamentally alters the solubility profile of the intermediate stream. Instead of struggling with the polar intermediate directly, the novel process converts it into a lipophilic benzyl ester (Compound 3), which is readily soluble in organic solvents like ethyl acetate and easily separable from aqueous waste streams. This clever manipulation of polarity eliminates the absolute dependence on chromatographic purification, allowing for standard work-up procedures such as filtration, extraction, and crystallization. Additionally, the process utilizes a convergent deprotection strategy where a single catalytic hydrogenation step simultaneously cleaves both the nitrogen and oxygen benzyl groups. This consolidation of steps not only reduces the total number of unit operations but also minimizes material handling and potential yield losses, presenting a streamlined, cost-effective solution specifically designed for the rigorous demands of commercial API intermediate manufacturing.
Mechanistic Insights into Reductive Amination and Hydrogenolysis
The core of this synthetic success lies in the precise control of the initial reductive amination and the subsequent catalytic hydrogenolysis. In the first step, the reaction between D-threonine and benzaldehyde is facilitated by anhydrous potassium carbonate in methanol. Mechanistically, the carbonate base serves a dual purpose: it neutralizes the carboxylic acid to prevent unwanted side reactions and creates the necessary alkaline environment for imine formation without promoting epimerization of the alpha-carbon. The use of sodium triacetoxyborohydride as the reducing agent is critical here, as it is mild enough to selectively reduce the imine intermediate to the secondary amine (Compound 1) without affecting other functional groups, ensuring high chemoselectivity. The patent data explicitly highlights that substituting sodium bicarbonate with anhydrous potassium carbonate in this specific solvent system significantly enhances the reaction kinetics and crude product purity, suggesting a superior solubility match that drives the equilibrium forward more effectively than weaker bases.
Following the N-methylation via reductive amination with paraformaldehyde and sodium cyanoborohydride, the process employs a strategic esterification followed by a global deprotection. The esterification of Compound 2 with benzyl bromide in DMF creates Compound 3, masking the polar carboxylic acid and hydroxyl groups. The final transformation involves hydrogenation over a palladium-on-carbon catalyst. This step is mechanistically elegant as it exploits the lability of the benzyl group under hydrogenolytic conditions. The catalyst facilitates the cleavage of the C-N and C-O benzyl bonds simultaneously, regenerating the free amine, carboxylic acid, and hydroxyl functionalities of the target N-methyl-D-threonine in a single pot. This simultaneous removal is energetically favorable and kinetically efficient, preventing the accumulation of partially deprotected intermediates that could complicate purification. The result is a high-purity final product obtained simply by filtration of the catalyst and solvent removal, demonstrating a masterful application of orthogonal protection group chemistry to solve complex manufacturing problems.
How to Synthesize N-Methyl-D-Threonine Efficiently
Implementing this synthesis requires strict adherence to the optimized reaction conditions detailed in the patent to ensure maximum yield and purity. The process begins with the careful preparation of the reaction mixture in methanol, ensuring that the anhydrous potassium carbonate is fully dispersed before the addition of the reducing agent to prevent localized exotherms. Operators must monitor the temperature closely during the addition of sodium triacetoxyborohydride, maintaining the oil bath at 30°C to balance reaction rate with selectivity. Following the isolation of Compound 1, the N-methylation step utilizes paraformaldehyde as a stable source of formaldehyde, which depolymerizes in situ to react with the secondary amine. The subsequent esterification in DMF requires controlled addition of benzyl bromide to manage the exothermic nature of the alkylation, followed by a prolonged stirring period to ensure complete conversion. Finally, the hydrogenation step demands rigorous safety protocols regarding hydrogen gas handling, but offers the reward of a straightforward work-up that yields the target compound as a white solid with minimal downstream processing.
- Perform reductive amination of D-threonine with benzaldehyde using anhydrous potassium carbonate and sodium triacetoxyborohydride in methanol to generate Compound 1.
- Conduct N-methylation of Compound 1 using paraformaldehyde and sodium cyanoborohydride in methanol to yield Compound 2.
- Execute esterification of Compound 2 with benzyl bromide in DMF containing sodium bicarbonate to form Compound 3, reducing polarity for easier purification.
- Complete the synthesis by hydrogenating Compound 3 with palladium on carbon in methanol to simultaneously remove benzyl groups and obtain the target Compound 4.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the technical refinements in this patent translate directly into tangible operational efficiencies and risk mitigation. The most significant commercial advantage is the complete elimination of column chromatography, which is traditionally one of the most expensive and time-consuming unit operations in fine chemical synthesis. By replacing chromatography with crystallization and extraction, the process drastically reduces the consumption of silica gel and high-grade organic solvents, leading to substantial cost savings in raw materials and waste disposal. Furthermore, the simplified workflow shortens the overall production cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand fluctuations. The high yields reported in each step, particularly the near-quantitative conversion in the methylation and hydrogenation stages, minimize the loss of valuable chiral starting materials, ensuring that the cost of goods sold remains competitive even for complex peptide building blocks.
- Cost Reduction in Manufacturing: The strategic design of this synthetic route inherently lowers production costs by removing the need for expensive chromatographic purification media and the associated labor. The use of commercially available and relatively inexpensive reagents such as benzaldehyde, paraformaldehyde, and benzyl bromide ensures that raw material costs remain stable and predictable. Additionally, the ability to perform a simultaneous deprotection of two functional groups in the final step reduces the number of reactor charges and solvent swaps required, further driving down utility and operational expenses. This lean manufacturing approach allows suppliers to offer high-purity N-methyl-D-threonine at a more attractive price point compared to legacy methods that suffer from low throughput and high waste generation.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the robustness of this four-step sequence, which relies on stable, shelf-stable reagents that are readily sourced from multiple global vendors. The avoidance of sensitive or hazardous reagents that require special handling or cold chain logistics simplifies inventory management and reduces the risk of production stoppages due to material shortages. The high purity of the intermediates achieved through the optimized base selection means that quality control failures are less likely, reducing the incidence of batch rejections that can disrupt delivery schedules. This reliability makes the supplier a more dependable partner for long-term contracts, ensuring that pharmaceutical clients can maintain their own production schedules for critical polypeptide drugs without fear of raw material interruptions.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process is exceptionally well-suited for expansion from pilot plant to multi-ton commercial production. The reduction in solvent usage and the elimination of silica waste align perfectly with modern green chemistry principles and increasingly stringent environmental regulations. The process generates less hazardous waste, simplifying effluent treatment and lowering the environmental compliance burden on the manufacturing facility. The physical state of the intermediates, transitioning from solids to oils and back to solids, is manageable in standard stainless steel reactors without requiring specialized glass-lined equipment for highly corrosive conditions. This ease of scale-up ensures that supply can be rapidly ramped up to meet surging demand for antibacterial peptides and cancer therapeutics without requiring massive capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of N-methyl-D-threonine using this patented methodology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing transparency into the process capabilities. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their own supply chains and assess the quality standards achievable through this route. We encourage technical teams to review these points to gain a deeper appreciation of the process advantages.
Q: Why is anhydrous potassium carbonate preferred over sodium bicarbonate in the first step?
A: The patent data indicates that using anhydrous potassium carbonate in the initial reductive amination step significantly improves the purity and yield of the crude product compared to sodium bicarbonate, addressing solubility and extraction challenges early in the process.
Q: How does this synthesis method avoid column chromatography?
A: By converting the highly water-soluble intermediate Compound 2 into the esterified Compound 3, the polarity is effectively reduced. This modification allows for standard extraction and crystallization techniques, eliminating the need for expensive and time-consuming column chromatography during scale-up.
Q: What is the advantage of the final hydrogenation step in this route?
A: The final step utilizes palladium-carbon catalyzed hydrogenation to simultaneously remove both the N-benzyl and O-benzyl protecting groups in a single operation. This convergent deprotection strategy streamlines the workflow, reduces solvent usage, and maximizes overall process efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Methyl-D-Threonine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful development of next-generation polypeptide drugs relies heavily on the availability of high-quality, scalable amino acid derivatives. Our technical team has thoroughly analyzed the synthetic route disclosed in CN113773214A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this process to life. We are committed to delivering N-methyl-D-threonine that meets stringent purity specifications, utilizing our rigorous QC labs to ensure every batch is free from critical impurities and ready for immediate use in solid-phase peptide synthesis. Our facility is equipped to handle the specific solvent systems and hydrogenation requirements of this route safely and efficiently, guaranteeing a consistent supply of this vital building block for your R&D and commercial manufacturing needs.
We invite you to collaborate with us to leverage this advanced technology for your upcoming projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data from our pilot runs and comprehensive route feasibility assessments to demonstrate how our implementation of this patent can optimize your supply chain. Let us be your partner in accelerating the development of life-saving antibacterial and anticancer therapies through superior chemical manufacturing.
