Advanced Manufacturing of 2-Hydroxyl Biphenyl Compounds via Copper-Catalyzed Decarboxylation for Global Supply Chains
The chemical industry is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthetic routes for critical intermediates. A pivotal advancement in this domain is documented in patent CN101955415B, which discloses a robust method for preparing 2-hydroxyl biphenyl compounds. This specific class of compounds serves as a vital building block in the synthesis of fungicides, food sanitizers, fiber and plastic modification agents, and various agrochemicals. The patented methodology represents a significant departure from traditional synthesis pathways by utilizing 9-fluorenone compounds as the primary starting material. This strategic choice of feedstock not only ensures a wide source and ready availability of raw materials but also establishes a foundation for a high-yield synthetic route that is exceptionally well-suited for large-scale industrial production. By leveraging a two-step process involving base-mediated ring opening followed by copper-catalyzed oxidative decarboxylation, this technology addresses long-standing challenges in purity and cost efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 2-hydroxyl biphenyl compounds has been plagued by significant technical and economic inefficiencies inherent in three primary conventional methods. The first method involves recovering phenol compounds from distillation residues generated during sulfonation processes; however, this approach suffers from a critical defect where 4-hydroxyl biphenyl compounds inevitably remain as residual impurities, drastically lowering the overall purity of the final product and restricting its application in high-value sectors. Furthermore, this method is severely constrained by the limited availability of suitable raw materials when attempting to produce on a massive scale. The second conventional technique relies on the gas-phase dehydrogenation of substituted cyclohexenyl hexanone in the presence of metal-loaded alumina catalysts such as copper or nickel. While chemically feasible, this route is economically prohibitive due to the exorbitant cost of the substituted cyclohexenyl hexanone raw material and the substantial energy consumption required to maintain gas-phase reaction conditions. The third method utilizes diazotization of substituted 2-phenylaniline with sodium nitrite in sulfuric acid, followed by hydrolysis. This pathway is equally problematic, characterized by low overall yields and the high market price of the 2-aminobiphenyl precursors, rendering it uncompetitive for modern cost-sensitive manufacturing environments.
The Novel Approach
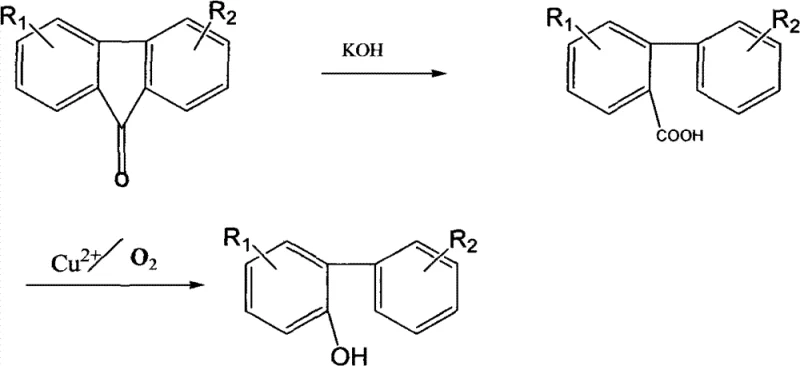
In stark contrast to these legacy technologies, the novel approach outlined in the patent data introduces a paradigm shift by employing 9-fluorenone compounds as the foundational starting material. This innovative route begins with the conversion of 9-fluorenone into biphenyl-2-carboxylic acid compounds through a highly efficient base-mediated ring-opening reaction. This intermediate is then subjected to a catalytic oxidation process to yield the target 2-hydroxyl biphenyl compound. The brilliance of this synthetic strategy lies in its ability to bypass the formation of difficult-to-separate isomers and avoid the use of expensive amines or energy-intensive gas-phase reactions. The process operates under relatively mild synthesis conditions compared to the extreme requirements of dehydrogenation, yet it achieves a high synthetic route yield that maximizes resource utilization. By integrating a copper-catalyzed decarboxylation step that utilizes air as the oxidant, the method significantly reduces reagent costs and simplifies the workup procedure, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations seeking to optimize their production lines.
Mechanistic Insights into Copper-Catalyzed Oxidative Decarboxylation
The core of this technological breakthrough resides in the sophisticated mechanistic pathway that transforms the carboxylic acid intermediate into the final phenolic product. The first stage involves the nucleophilic attack of hydroxide ions on the carbonyl carbon of the 9-fluorenone ring system. Under basic conditions provided by solid potassium hydroxide in a toluene solvent at approximately 112°C, the strained five-membered ring of the fluorenone undergoes cleavage. This ring-opening reaction is thermodynamically favorable and proceeds with exceptional efficiency, often achieving yields exceeding 99%, to form the biphenyl-2-carboxylic acid salt, which is subsequently acidified to isolate the free acid. This step is crucial as it sets the stereochemical and structural stage for the subsequent transformation, ensuring that the carboxyl group is positioned ortho to the biphenyl linkage, a prerequisite for the final cyclization or decarboxylation event.
The second stage, which is the rate-determining and most chemically intricate part of the process, involves the copper-catalyzed oxidative decarboxylation of the biphenyl-2-carboxylic acid. In this step, a cupric salt, such as cupric chloride or neutralized verdigris, is introduced in mass ratios ranging from 1% to 5%. Upon heating the mixture to temperatures between 220°C and 300°C, specifically optimized at 250-280°C, the copper catalyst facilitates the removal of the carboxyl group as carbon dioxide. Simultaneously, air is steadily introduced into the reactor at controlled flow rates (0.2-3 liters/min) to serve as the terminal oxidant. This oxidative environment promotes the formation of a radical intermediate or a copper-carboxylate complex that collapses to form the carbon-oxygen bond of the phenol while releasing CO2. The use of copper is particularly advantageous as it is abundant and inexpensive compared to palladium or other precious metals, yet it provides sufficient catalytic activity to drive the reaction to completion within 1 to 3 hours.
Furthermore, the mechanism inherently supports high purity profiles by minimizing side reactions. The specific choice of cupric chloride or neutralized verdigris ensures better dispersibility within the reaction matrix, which enhances the contact between the catalyst and the substrate, thereby improving the yield to levels around 86%. The oxidative nature of the reaction also helps in consuming potential reduced byproducts, leading to a cleaner crude product that requires less intensive purification. Following the reaction, the mixture is cooled, dissolved in ethanol, and treated with activated carbon to remove trace colored impurities and inorganic salts. This straightforward workup procedure underscores the practical viability of the mechanism for industrial applications, ensuring that the final off-white crystalline product meets stringent purity specifications of up to 99.6% as determined by HPLC analysis.
How to Synthesize 2-Hydroxyl Biphenyl Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety. The process is designed to be scalable, moving seamlessly from laboratory benchtop experiments to multi-ton production batches. The initial preparation of the biphenyl-2-carboxylic acid intermediate is a critical quality gate; maintaining the temperature at 112°C during the addition of 9-fluorenone to the potassium hydroxide slurry ensures complete conversion without degradation. Subsequently, the oxidative decarboxylation step demands careful thermal management, as the reaction is endothermic and requires sustained heating to 250-280°C. The introduction of air must be regulated to prevent excessive oxidation of the phenolic product while ensuring sufficient oxygen is available for the catalytic cycle. For a comprehensive understanding of the operational parameters, safety protocols, and specific stoichiometric ratios required for GMP-compliant manufacturing, please refer to the detailed technical guide below.
- Prepare biphenyl-2-carboxylic acid by reacting 9-fluorenone with solid potassium hydroxide in toluene at 112°C, followed by acidification and filtration.
- Mix the biphenyl-2-carboxylic acid with 1-5% cupric salt catalyst in a reactor and heat to 220-300°C.
- Introduce air at a steady rate for 1-3 hours to effect oxidative decarboxylation, then purify via ethanol recrystallization and activated carbon treatment.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method offers transformative benefits that directly impact the bottom line and operational resilience. The shift away from expensive and scarce raw materials like substituted cyclohexenyl hexanone or 2-aminobiphenyl to widely available 9-fluorenone derivatives fundamentally alters the cost structure of production. This transition mitigates the risk of supply disruptions caused by raw material shortages and reduces exposure to volatile pricing in niche chemical markets. Moreover, the elimination of complex purification steps required to remove 4-hydroxyl biphenyl isomers significantly streamlines the manufacturing workflow, reducing both processing time and utility consumption. These factors collectively contribute to a more robust and predictable supply chain capable of meeting the rigorous demands of global pharmaceutical and agrochemical clients.
- Cost Reduction in Manufacturing: The economic advantages of this process are profound, primarily driven by the substitution of high-cost precursors with commodity-grade 9-fluorenone. By eliminating the need for precious metal catalysts and replacing them with inexpensive copper salts, the direct material costs are substantially lowered. Additionally, the high yield of the ring-opening step (over 99%) minimizes waste generation, ensuring that nearly all input material is converted into valuable intermediate. The simplified downstream processing, which avoids complex chromatographic separations or multiple recrystallizations, further reduces operational expenditures related to solvents and energy. Consequently, manufacturers can achieve significant cost savings in fine chemical intermediates manufacturing without compromising on the quality or purity of the final output.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of raw materials that are produced on a massive industrial scale for other applications, ensuring a stable and diversified supply base. Unlike specialized amines or ketones that may have limited suppliers, 9-fluorenone is a standard chemical with a mature global market. This abundance reduces lead times for high-purity pharmaceutical intermediates and allows for larger inventory buffers against market fluctuations. The robustness of the reaction conditions also means that production is less susceptible to minor variations in feedstock quality, enhancing the overall reliability of the manufacturing schedule. This stability is crucial for long-term contracts where consistent delivery is paramount.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns well with modern green chemistry principles. The use of air as an oxidant generates water as the primary byproduct of reduction, avoiding the heavy metal waste streams associated with stoichiometric oxidants. The high atom economy of the decarboxylation step ensures minimal waste generation per unit of product. Furthermore, the process is inherently scalable; the reaction conditions (temperature and pressure) are compatible with standard stainless steel reactors used in the fine chemical industry, facilitating the commercial scale-up of complex organic molecules. This ease of scale-up allows producers to rapidly respond to increased market demand, ensuring that capacity constraints do not become a bottleneck for growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-hydroxyl biphenyl compounds using this advanced methodology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear picture of the technology's capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What are the primary advantages of using 9-fluorenone as a starting material over traditional sulfonation methods?
A: Using 9-fluorenone avoids the formation of difficult-to-remove 4-hydroxyl biphenyl isomers common in sulfonation residues, resulting in significantly higher purity (up to 99.6%) and eliminating complex separation steps.
Q: Why is copper catalysis preferred for the decarboxylation step in this process?
A: Copper salts act as efficient, low-cost catalysts that facilitate oxidative decarboxylation under air atmosphere, avoiding the need for expensive precious metals or hazardous diazotization reagents required in older methods.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process utilizes readily available raw materials like 9-fluorenone and operates under conditions (220-300°C) that are manageable with standard industrial reactors, offering high yields suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxyl Biphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of our clients' drug development and manufacturing programs. Our technical team has thoroughly analyzed the potential of the copper-catalyzed decarboxylation route and is prepared to leverage this technology to deliver superior value. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-hydroxyl biphenyl we produce meets the highest international standards for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. By partnering with NINGBO INNO PHARMCHEM, you gain access to our deep technical expertise and our commitment to continuous improvement. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term business goals.
