Advanced Enzymatic Resolution of Chiral Fatty Alcohols for Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access high-purity chiral building blocks, which are essential for the synthesis of bioactive molecules. Patent CN101045936B introduces a groundbreaking methodology for the preparation of chiral fatty alcohols utilizing acid anhydrides, specifically succinic anhydride, as acyl donors in lipase-catalyzed kinetic resolutions. This technology represents a significant departure from traditional acylating agents, addressing critical bottlenecks in downstream processing that have historically plagued the commercial production of optical isomers. By leveraging the unique reactivity of succinic anhydride in conjunction with robust biocatalysts such as Candida antarctica lipase B (CALB) or Pseudomonas fluorescens lipase (PFL), the process enables the facile separation of enantiomers through simple acid-base extraction rather than labor-intensive chromatography. For R&D directors and process chemists evaluating new routes for API intermediates, this patent offers a compelling blueprint for enhancing both purity profiles and operational efficiency without compromising on stereochemical integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the enzymatic resolution of racemic alcohols has frequently relied upon vinyl acetate as the standard acyl donor due to its irreversible reaction nature which drives equilibrium towards product formation. However, this conventional approach suffers from severe downstream processing limitations that become exponentially more problematic as production scales increase. The resulting vinyl esters and unreacted alcohols often possess similar physical properties, necessitating the use of silica gel column chromatography for effective separation and purification. This reliance on chromatography is not only prohibitively expensive in terms of stationary phase costs and solvent consumption but also introduces significant operational complexity and safety hazards associated with handling large volumes of flammable solvents in packed columns. Furthermore, the throughput of chromatographic processes is inherently low, creating a major bottleneck for supply chain managers who require consistent, high-volume delivery of chiral intermediates to meet aggressive drug development timelines.
The Novel Approach
The innovative strategy outlined in the patent data circumvents these traditional hurdles by employing succinic anhydride as the acyl donor, which fundamentally alters the physicochemical properties of the reaction products to enable superior separability. Upon enzymatic acylation, the resulting mono-ester contains a free carboxylic acid group that can be readily deprotonated using a mild base such as sodium carbonate or sodium bicarbonate. This chemical transformation converts the lipophilic ester into a water-soluble carboxylate salt, allowing for a clean partition between the organic phase containing the unreacted enantiomer and the aqueous phase containing the esterified enantiomer. This phase separation capability eliminates the need for column chromatography entirely, replacing it with standard liquid-liquid extraction unit operations that are well-understood, easily automated, and highly scalable in multi-purpose reactor systems. Consequently, this novel approach drastically simplifies the manufacturing workflow while simultaneously reducing the environmental footprint associated with solvent waste generation.
Mechanistic Insights into Lipase-Catalyzed Kinetic Resolution
The core of this technology lies in the exquisite stereoselectivity of lipase enzymes, which discriminate between the enantiomers of the racemic alcohol substrate within the active site. When racemic secondary alcohols, such as 2-octanol or 2-pentanol, are subjected to reaction with succinic anhydride in a non-polar solvent like isopropyl ether, the lipase selectively acylates one enantiomer (typically the R-enantiomer for many substrates using CALB) while leaving the other enantiomer (S-enantiomer) unreacted. This kinetic resolution proceeds efficiently at ambient temperatures, typically ranging from 20°C to 30°C, preserving the structural integrity of sensitive functional groups that might degrade under harsher chemical catalysis conditions. The reaction kinetics are carefully monitored, with the process ideally halted at a conversion rate between 40% and 55% to maximize the optical purity of both the remaining alcohol and the formed ester, ensuring that the enantiomeric excess (ee) values consistently exceed 95% and often reach as high as 98%.
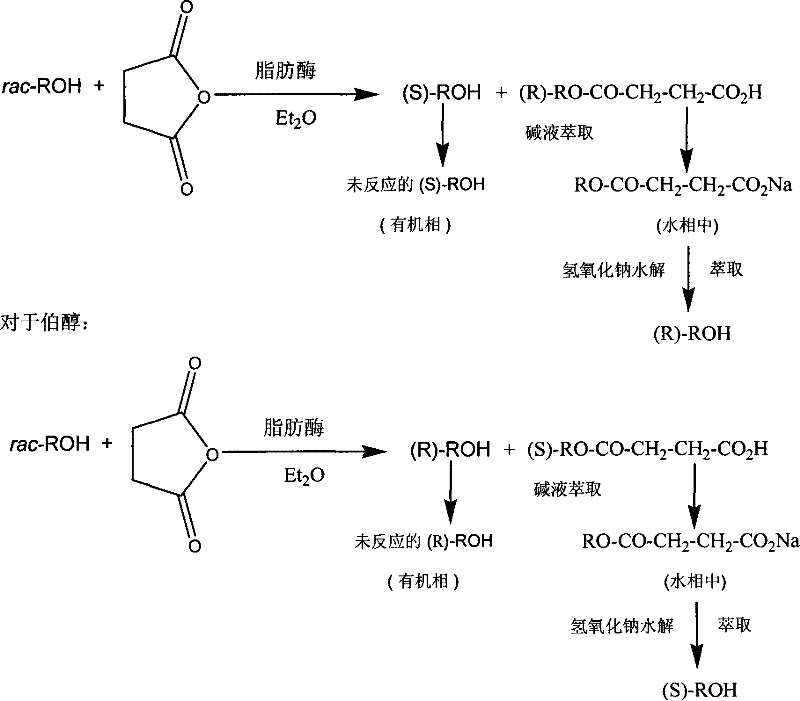
Beyond the initial enzymatic step, the mechanism includes a robust chemical workup that ensures the recovery of both optical isomers, thereby theoretically doubling the yield of useful chiral material from a single batch of racemate. The aqueous phase containing the succinate half-ester salt is subsequently treated with sodium hydroxide to effect hydrolysis, cleaving the ester bond and regenerating the chiral alcohol in its free form. This hydrolysis step is conducted under mild alkaline conditions at room temperature for approximately 8 to 12 hours, ensuring complete conversion without inducing racemization of the chiral center. The regenerated alcohol is then extracted back into an organic solvent, yielding the second enantiomer with high optical purity. This dual-recovery mechanism is critical for cost reduction in pharmaceutical intermediates manufacturing, as it transforms what would traditionally be a 50% yield process into one with near-quantitative utilization of the starting racemic material.
How to Synthesize Chiral Fatty Alcohols Efficiently
The practical implementation of this resolution technology involves a straightforward sequence of reaction, filtration, and extraction steps that can be readily adapted to existing pilot and production facilities. The process begins by suspending the chosen lipase catalyst in a dry ether solvent, followed by the addition of the racemic alcohol and stoichiometric amounts of succinic anhydride. Reaction progress is meticulously tracked using gas chromatography (GC) to determine the precise endpoint where optimal enantiomeric enrichment is achieved. Once the target conversion is reached, the heterogeneous catalyst and any unreacted solid anhydride are removed via simple filtration, clarifying the reaction mixture for the subsequent separation stages.
- React racemic alcohol with succinic anhydride and lipase (e.g., CALB or PFL) in isopropyl ether at room temperature until 40-55% conversion.
- Filter the mixture to remove enzymes and unreacted anhydride, then extract with sodium carbonate solution to separate the organic phase (unreacted alcohol) from the aqueous phase (ester salt).
- Hydrolyze the aqueous phase ester with sodium hydroxide to recover the opposite enantiomer of the alcohol, followed by final extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this succinic anhydride-based resolution process offers transformative advantages in terms of cost structure and operational reliability. The elimination of column chromatography represents a massive reduction in variable manufacturing costs, removing the need for expensive silica gel, specialized equipment, and the extensive labor required for fraction collection and analysis. Furthermore, the solvents utilized in this process, such as isopropyl ether or methyl tert-butyl ether, are commodity chemicals that are readily available in bulk quantities, ensuring stable pricing and minimizing the risk of supply disruptions compared to specialized HPLC-grade solvents. The ability to operate at room temperature also translates to significant energy savings, as there is no requirement for cryogenic cooling or high-temperature heating, further contributing to a lower overall cost of goods sold (COGS) for the final chiral intermediates.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the substitution of complex purification methods with simple extraction, which drastically lowers capital expenditure requirements for processing equipment. By avoiding chromatography, manufacturers can utilize standard stainless steel reactors and centrifuges, assets that are already prevalent in most fine chemical plants, thereby accelerating the timeline for technology transfer and commercial scale-up. Additionally, the recovery and potential recycling of unreacted succinic anhydride from the filtration step provide further opportunities for raw material cost optimization, creating a more circular and efficient production loop.
- Enhanced Supply Chain Reliability: The robustness of the enzymatic process under mild conditions ensures high batch-to-batch consistency, a critical factor for maintaining qualified supplier status with major pharmaceutical clients. Since the reaction does not rely on sensitive transition metal catalysts that can be poisoned by trace impurities, the process tolerates a wider range of raw material specifications, reducing the risk of batch failures due to feedstock variability. This resilience enhances supply security, allowing for longer production campaigns and larger batch sizes that can effectively buffer against market fluctuations and unexpected demand surges for key chiral alcohol intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns perfectly with green chemistry principles by reducing solvent intensity and eliminating solid waste streams associated with spent chromatography media. The aqueous waste streams generated during the extraction phases are primarily composed of benign salts and can be treated using standard wastewater protocols, simplifying regulatory compliance and reducing disposal fees. The inherent scalability of liquid-liquid extraction means that moving from kilogram-scale development to multi-ton commercial production involves linear scaling factors rather than the exponential complexities often encountered when scaling chromatographic separations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic resolution technology, drawing directly from the experimental data and process descriptions provided in the patent literature. Understanding these nuances is essential for project managers evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is succinic anhydride preferred over vinyl acetate for enzymatic resolution?
A: Succinic anhydride forms a half-ester salt that is water-soluble under basic conditions, allowing for simple liquid-liquid extraction separation. In contrast, vinyl acetate products often require expensive and difficult column chromatography for purification.
Q: What level of enantiomeric excess (ee) can be achieved with this process?
A: According to patent data, this method consistently achieves high optical purity, with enantiomeric excess (ee) values reaching up to 97-98% for various substrates like 2-octanol and 3-methyl-2-butanol.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates at room temperature using common solvents like isopropyl ether and avoids complex purification steps like chromatography, making it highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Fatty Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced biocatalytic processes in securing a competitive edge in the global supply of pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN101045936B can be seamlessly translated into robust industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art chiral HPLC and GC capabilities to guarantee that every batch of chiral fatty alcohol meets the exacting standards required for GMP synthesis. Our commitment to quality assurance extends beyond mere compliance, as we proactively monitor impurity profiles to ensure full alignment with the regulatory expectations of our international partners.
We invite forward-thinking R&D and procurement leaders to collaborate with us on leveraging this efficient resolution technology for your next project. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this anhydride-based route for your specific target molecules. We encourage you to contact us today to discuss your specific requirements,索取 specific COA data for our available chiral alcohol inventory, and review detailed route feasibility assessments tailored to your unique synthetic challenges.
