Advanced Biocatalytic Production of S-2-Hydroxy-4-Phenylbutyrate for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry's relentless pursuit of high-purity chiral intermediates has found a significant breakthrough in the biocatalytic synthesis methods detailed in patent CN101665811A. This intellectual property discloses a highly efficient method for preparing ethyl S-2-hydroxyl-4-phenylbutyrate, a critical chiral building block used extensively in the manufacture of Angiotensin-Converting Enzyme (ACE) inhibitors, such as the普利 class of medicines. The core innovation lies in the utilization of a novel dicarbonyl reductase coupled with a sophisticated coenzyme cyclic regeneration system mediated by formate dehydrogenase. This synergistic enzymatic approach allows for the catalytic synthesis of the target ester with an optical purity (ee value) reaching an impressive 99.5%, while simultaneously supporting substrate concentrations above 0.8 M. For R&D directors and process chemists, this represents a paradigm shift from traditional resolution or chemical reduction methods, offering a pathway to reduce impurity profiles and enhance overall process robustness. The ability to operate at such high substrate loadings without compromising stereoselectivity addresses a major bottleneck in industrial biocatalysis, making this technology a cornerstone for modern green chemistry initiatives in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of S-2-hydroxy-4-phenyl ethyl butyrate has relied heavily on resolution methods or harsh chemical synthesis, both of which present substantial drawbacks for large-scale commercial production. Traditional resolution methods, whether chemical or biological, are inherently limited by a maximum theoretical yield of 50%, which is economically inefficient and generates significant waste streams that require costly disposal. Furthermore, chemical synthesis routes often necessitate the use of expensive transition metal catalysts and require high hydrogen pressure conditions, imposing stringent safety requirements and capital expenditure on specialized high-pressure reactor equipment. Even when successful, these chemical routes frequently struggle to achieve high enantiomeric excess, with reported ee values typically hovering between 50% and 82%, necessitating additional purification steps that erode profit margins. Biological alternatives using hydroxynitrilase or lipase have also faced challenges; hydroxynitrilase pathways involve violent nitrile hydrolysis steps that can lead to product racemization and degradation, while lipase-mediated resolution remains capped at the 50% yield ceiling. These limitations collectively create a supply chain vulnerability, driving up costs and extending lead times for reliable pharmaceutical intermediate suppliers.
The Novel Approach
In stark contrast to these legacy technologies, the novel approach described in the patent utilizes a highly specific dicarbonyl reductase that directly reduces the keto-ester precursor with exceptional stereocontrol.  This enzymatic transformation is not only highly selective but is also driven by an integrated coenzyme regeneration system that recycles NAD(P)H in situ, eliminating the need for stoichiometric amounts of expensive cofactors. The process operates under mild reaction conditions, typically at ambient temperatures and atmospheric pressure, which drastically simplifies the engineering requirements for commercial scale-up. By employing a solvent system that balances organic solvents like toluene or ethanol with buffered aqueous phases, the method maintains enzyme stability while ensuring adequate substrate solubility. This dual-phase or mixed-solvent strategy allows for substrate concentrations to reach levels previously unattainable with older alcohol dehydrogenase systems, which were often limited to 0.1 M. The result is a streamlined, cost-effective manufacturing route that delivers high-purity products with minimal downstream processing, positioning it as a superior choice for cost reduction in pharmaceutical intermediate manufacturing.
This enzymatic transformation is not only highly selective but is also driven by an integrated coenzyme regeneration system that recycles NAD(P)H in situ, eliminating the need for stoichiometric amounts of expensive cofactors. The process operates under mild reaction conditions, typically at ambient temperatures and atmospheric pressure, which drastically simplifies the engineering requirements for commercial scale-up. By employing a solvent system that balances organic solvents like toluene or ethanol with buffered aqueous phases, the method maintains enzyme stability while ensuring adequate substrate solubility. This dual-phase or mixed-solvent strategy allows for substrate concentrations to reach levels previously unattainable with older alcohol dehydrogenase systems, which were often limited to 0.1 M. The result is a streamlined, cost-effective manufacturing route that delivers high-purity products with minimal downstream processing, positioning it as a superior choice for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Dicarbonyl Reductase-Catalyzed Asymmetric Reduction
The heart of this technological advancement is the precise mechanistic interplay between the dicarbonyl reductase and the formate dehydrogenase-mediated cofactor regeneration loop. The dicarbonyl reductase specifically targets the pro-chiral ketone group of the substrate, transferring a hydride ion from the reduced cofactor NAD(P)H to generate the desired (S)-configured alcohol with rigorous stereochemical fidelity. 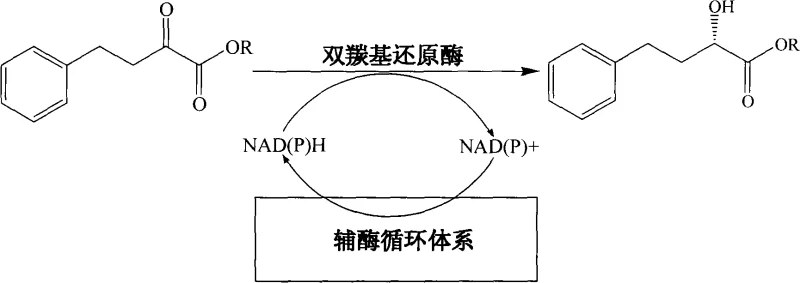 Crucially, the oxidation of NAD(P)H to NAD(P)+ during this reduction step is immediately counteracted by the formate dehydrogenase, which oxidizes sodium formate to carbon dioxide while regenerating the reduced cofactor. This cyclic regeneration ensures that the catalytic cycle can continue indefinitely without the accumulation of inhibitory oxidized cofactors or the depletion of the expensive reducing equivalents. The patent specifies that the dicarbonyl reductase used shares high homology with specific sequences (SEQ ID NO.1/2), indicating a tailored protein engineering approach that optimizes activity and stability in the presence of organic co-solvents. This robustness is essential for maintaining high reaction rates over the 1 to 18-hour reaction window described in the embodiments. Furthermore, the strict control of pH (between 4 and 7.5) and the specific ratio of organic to aqueous phases create a microenvironment that maximizes enzyme turnover number while minimizing non-enzymatic background reactions that could lead to racemization.
Crucially, the oxidation of NAD(P)H to NAD(P)+ during this reduction step is immediately counteracted by the formate dehydrogenase, which oxidizes sodium formate to carbon dioxide while regenerating the reduced cofactor. This cyclic regeneration ensures that the catalytic cycle can continue indefinitely without the accumulation of inhibitory oxidized cofactors or the depletion of the expensive reducing equivalents. The patent specifies that the dicarbonyl reductase used shares high homology with specific sequences (SEQ ID NO.1/2), indicating a tailored protein engineering approach that optimizes activity and stability in the presence of organic co-solvents. This robustness is essential for maintaining high reaction rates over the 1 to 18-hour reaction window described in the embodiments. Furthermore, the strict control of pH (between 4 and 7.5) and the specific ratio of organic to aqueous phases create a microenvironment that maximizes enzyme turnover number while minimizing non-enzymatic background reactions that could lead to racemization.
From an impurity control perspective, this mechanism offers distinct advantages over chemical hydrogenation. Because the enzyme active site acts as a chiral pocket, it physically excludes the formation of the (R)-enantiomer, leading to the observed 99.5% ee value without the need for chiral chromatography columns during the reaction phase. The mild conditions also prevent thermal degradation or side reactions such as ester hydrolysis, which are common pitfalls in acid or base-catalyzed chemical processes. The use of formate dehydrogenase also ensures that the only byproduct of the cofactor regeneration is carbon dioxide gas, which harmlessly vents from the reaction mixture, leaving no toxic residues in the final product. This clean reaction profile simplifies the work-up procedure, as the primary task becomes separating the product from the buffer salts and residual proteins rather than removing heavy metal catalysts or complex organic byproducts. For quality assurance teams, this translates to a much cleaner impurity spectrum, facilitating easier compliance with stringent ICH guidelines for residual solvents and elemental impurities in drug substances.
How to Synthesize Ethyl S-2-Hydroxyl-4-Phenylbutyrate Efficiently
The practical implementation of this synthesis route involves a straightforward sequence of mixing reagents, controlling reaction parameters, and executing a standard extraction protocol. The process begins with the preparation of a reaction mixture containing the keto-ester substrate, the specific dicarbonyl reductase enzyme, and the formate dehydrogenase system in a buffered solvent environment. Detailed operational parameters, such as the optimal oscillation speed of 200 rpm and the precise enzyme loading of 5 U/mL, are critical to achieving the reported conversion efficiencies of over 95%. The reaction proceeds at normal temperature, eliminating the need for energy-intensive heating or cooling cycles, and typically reaches completion within 9 to 18 hours depending on the substrate loading. Following the reaction, the product is isolated through liquid-liquid extraction, leveraging the solubility differences between the organic product and the aqueous enzyme solution. For a comprehensive, step-by-step guide including exact reagent quantities and purification details, please refer to the standardized protocol below.
- Prepare the reaction system by adding substrate (2-oxo-4-phenylbutyrate derivative), dicarbonyl reductase, and a coenzyme cyclic regeneration system mediated by formate dehydrogenase into a solvent mixture.
- Conduct an oscillatory reaction at ambient temperature for 1 to 18 hours, maintaining a rotation speed of 100 to 300 rpm to ensure proper mixing and enzyme activity.
- Separate and purify the resulting product by extracting the organic layer, washing with water, and performing column chromatography to obtain the final high-purity ethyl S-2-hydroxyl-4-phenylbutyrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biocatalytic technology offers transformative benefits that extend far beyond simple yield improvements. The shift from high-pressure chemical hydrogenation to ambient enzymatic reduction fundamentally alters the cost structure of manufacturing this key intermediate. By eliminating the requirement for expensive noble metal catalysts and specialized high-pressure reactors, the capital expenditure (CAPEX) and operational expenditure (OPEX) associated with production facilities are significantly reduced. Moreover, the ability to run reactions at high substrate concentrations (0.8 M and above) means that less solvent is required per kilogram of product, leading to substantial reductions in raw material costs and waste disposal fees. This efficiency gain is compounded by the high optical purity of the product, which minimizes the need for costly recrystallization or chiral separation steps downstream, thereby shortening the overall production cycle time. These factors combine to create a more resilient and cost-competitive supply chain, ensuring that buyers can secure high-quality materials at a more stable price point.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and high-pressure equipment removes two of the most significant cost drivers in traditional synthesis. Additionally, the high atom economy of the enzymatic reduction, coupled with the use of inexpensive sodium formate as the reducing agent, ensures that the variable cost of goods sold is drastically optimized compared to stoichiometric chemical reduction methods.
- Enhanced Supply Chain Reliability: Because the process operates under mild conditions with robust enzymes, the risk of batch failure due to equipment malfunction or safety incidents is minimized. The use of readily available starting materials and the scalability of fermentation-derived enzymes ensure a consistent and continuous supply of the intermediate, reducing the lead time for high-purity pharmaceutical intermediates and mitigating the risk of supply disruptions.
- Scalability and Environmental Compliance: The aqueous-organic solvent system and the generation of benign byproducts (CO2) make this process inherently greener and easier to scale from pilot to commercial production. This aligns perfectly with modern environmental regulations, reducing the burden of wastewater treatment and hazardous waste management, which in turn lowers compliance costs and enhances the sustainability profile of the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic process. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on performance metrics and operational feasibility. Understanding these details is crucial for technical teams evaluating the integration of this route into their existing manufacturing portfolios.
Q: What optical purity can be achieved with this biocatalytic method?
A: According to patent CN101665811A, this method achieves an exceptional enantiomeric excess (ee value) of up to 99.5%, significantly surpassing traditional chemical synthesis methods which typically range between 50% and 82%.
Q: How does this process improve upon conventional chemical synthesis?
A: Unlike conventional chemical synthesis that requires expensive catalysts and high hydrogen pressure equipment, this biocatalytic route operates under mild conditions with high substrate concentrations (up to 0.8M or higher), eliminating safety risks and reducing equipment costs.
Q: What is the maximum substrate concentration supported by this technology?
A: The technology supports substrate concentrations reaching above 0.8M, with some embodiments demonstrating efficient conversion even at higher molarities, which drastically reduces solvent usage and improves volumetric productivity compared to older enzymatic methods limited to 0.1M.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-2-Hydroxy-4-Phenylbutyrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity chiral intermediates in the development of life-saving cardiovascular medications. Our team of expert process chemists has extensively analyzed the biocatalytic pathways described in CN101665811A and possesses the technical capability to adapt and optimize this technology for your specific needs. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, consistently delivering products with the high ee values and low impurity profiles required by global regulatory agencies.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this enzymatic process for your specific volume requirements. We encourage you to contact us today to discuss your project needs,索取 specific COA data for our reference standards, and review our comprehensive route feasibility assessments. Let us help you secure a reliable, cost-effective, and sustainable supply of this vital pharmaceutical intermediate.
