Advanced TS-1 Catalyzed Synthesis of 2,3-Dimethyl-2,3-Dinitrobutane for High-Security Applications
Introduction to Patent CN102267913B
The global demand for high-security materials, particularly explosive tracer agents mandated by the International Civil Aviation Organization (ICAO), requires manufacturing processes that prioritize both safety and exceptional purity. Patent CN102267913B introduces a groundbreaking synthetic method for 2,3-dimethyl-2,3-dinitrobutane (DMNB), a critical compound used not only as an explosive taggant but also as a precursor for nitric oxide scavengers in pharmaceutical applications. This technology represents a significant paradigm shift away from legacy chemistries that rely on hazardous nitroalkanes, offering a greener, more controllable pathway utilizing acetoxime and hydrogen peroxide. By leveraging a modified titanium silicalite (TS-1) catalyst, this process achieves superior product quality with a distinct melting point profile, addressing the long-standing industry challenge of impurity management in nitro-compound synthesis.
For R&D directors and procurement specialists, the implications of this patent extend beyond simple molecule construction; it offers a viable route to mitigate supply chain risks associated with regulated toxic precursors. The method operates under relatively mild alkaline conditions (pH 8-10) and moderate temperatures (60-90°C), avoiding the extreme hazards of sodium hydride or elemental bromine found in older literature. As a reliable explosive tracer intermediate supplier, understanding the nuances of this catalytic oxidation is essential for securing a stable supply of high-grade DMNB that meets rigorous international safety standards without compromising on production feasibility or environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3-dimethyl-2,3-dinitrobutane has been plagued by severe safety and quality issues inherent to the available chemical routes. Early methodologies, such as those described by Seigle and Hass, relied on the coupling of 2-nitropropane sodium salts with halogenated nitropropanes in alcohol solvents. As illustrated in the reaction scheme below, these pathways necessitate the use of highly toxic 2-nitropropane and hazardous halogens, resulting in poor yields ranging from merely 9% to 43% depending on the halogen used. Furthermore, subsequent improvements by researchers like Tsunoda, while claiming higher yields, still depended on toxic bromine and produced crude products with low melting points (129-130°C), indicating significant contamination that complicates downstream purification for sensitive applications.
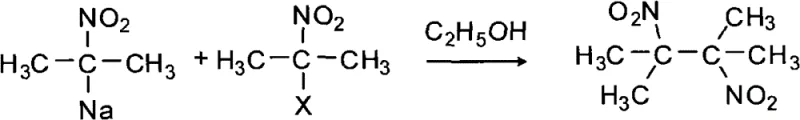
The reliance on these dangerous precursors creates substantial regulatory and logistical burdens for chemical manufacturers. The use of sodium hydride in conjunction with ceric ammonium nitrate, as reported in more recent literature, introduces expensive oxidants and pyrophoric reagents that escalate production costs and safety risks. These conventional methods often generate complex impurity profiles that are difficult to separate, leading to final products that may fail to meet the stringent physical specifications required for explosive detection systems. Consequently, the industry has urgently needed a alternative synthetic strategy that eliminates these toxic inputs while delivering a cleaner, more consistent product stream suitable for commercial scale-up of complex nitro compounds.
The Novel Approach
The innovative process detailed in CN102267913B fundamentally reimagines the construction of the dinitrobutane skeleton by utilizing acetoxime as the primary starting material. Instead of coupling toxic nitro fragments, this method employs a direct oxidative dimerization or functionalization strategy mediated by a modified TS-1 zeolite catalyst. The reaction proceeds in an aqueous environment using hydrogen peroxide as the terminal oxidant, generating water as the primary byproduct alongside recyclable acetone. This shift from halogenated/hydride chemistry to catalytic oxidation drastically simplifies the waste stream and removes the need for handling pyrophoric or highly toxic reagents, thereby enhancing overall plant safety and reducing the cost reduction in specialty chemical manufacturing associated with hazardous waste disposal.
Moreover, the product quality achieved through this novel route is demonstrably superior, with the synthesized DMNB exhibiting a sharp melting point range of 210-212°C. This significant increase in melting point compared to the 129-130°C of prior art serves as a robust indicator of high purity and crystalline integrity. The ability to produce such high-quality material directly from the reactor reduces the burden on downstream purification units, making the process economically attractive despite the specific stoichiometric requirements. By adopting this methodology, manufacturers can secure a supply of high-purity 2,3-dimethyl-2,3-dinitrobutane that is fully compliant with modern safety regulations and ready for immediate application in high-value security and medical sectors.
Mechanistic Insights into TS-1 Catalyzed Oxidative Functionalization
The core of this technological advancement lies in the unique activity of the modified titanium silicalite (TS-1) catalyst, which facilitates the activation of hydrogen peroxide under mild alkaline conditions. In this mechanistic pathway, acetoxime serves as both the carbon source and the nitrogen donor, undergoing a complex transformation in the presence of the catalyst and oxidant. The TS-1 framework provides specific active sites that likely stabilize reactive oxygen species, enabling the selective formation of the gem-dinitro motif without over-oxidation or degradation of the carbon backbone. The reaction is conducted at a controlled temperature of 60-90°C and a pH of 8-10, conditions that are critical for maintaining catalyst stability while ensuring sufficient reaction kinetics for the conversion of acetoxime to the target dinitro compound.
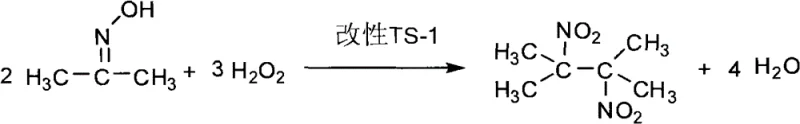
However, the reaction system is not without competing pathways that must be carefully managed to ensure high yield and selectivity. A significant side reaction involves the hydrolysis of the acetoxime starting material, which leads to the formation of acetone and hydroxylamine derivatives, effectively consuming the feedstock without generating the desired product. As shown in the equilibrium diagram below, this hydrolysis is reversible but can be driven forward by reaction conditions, representing a key loss channel that the process parameters aim to minimize. Understanding this balance is crucial for R&D teams optimizing the process, as the molar ratio of hydrogen peroxide to acetoxime (optimized between 1:1 and 3:1) plays a pivotal role in suppressing hydrolysis while driving the oxidative coupling forward.

Control of these impurity profiles is achieved through precise regulation of the pH and temperature during the addition of reagents. Maintaining the pH between 9 and 10 prevents the dissolution and deactivation of the TS-1 catalyst while minimizing the rate of de-oximation. Furthermore, the post-reaction processing involves a strategic distillation step at temperatures not exceeding 80°C to remove the acetone byproduct, which shifts the equilibrium and aids in the isolation of the solid DMNB product. This meticulous control over the reaction environment ensures that the final material possesses the high thermal stability and purity required for its intended use as an explosive taggant, where consistency is paramount for reliable detection performance.
How to Synthesize 2,3-Dimethyl-2,3-Dinitrobutane Efficiently
The practical implementation of this synthesis requires strict adherence to the optimized parameters defined in the patent to maximize efficiency and product quality. The process begins with the preparation of the reaction vessel containing acetoxime, water, and the modified TS-1 catalyst, followed by the controlled addition of alkali and hydrogen peroxide at elevated temperatures. Operators must monitor the pH closely to stay within the 8-10 window and ensure the temperature remains between 60-90°C throughout the addition and subsequent holding period. Detailed standardized operating procedures regarding catalyst loading, addition rates, and workup protocols are essential for reproducing the high melting point and purity characteristics described in the intellectual property.
- Prepare the reaction system by mixing acetoxime, water, and modified TS-1 catalyst at room temperature, ensuring the water weight is 1 to 10 times that of the acetoxime.
- Heat the mixture to 60-90°C and simultaneously add aqueous alkali and hydrogen peroxide while maintaining a pH between 8 and 10 and a molar ratio of H2O2 to acetoxime of 1: 1 to 3:1.
- After addition, maintain reaction temperature for 0-2 hours, then cool, filter to recover the catalyst, remove acetone byproduct via distillation, and crystallize the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this TS-1 catalyzed process offers tangible strategic benefits that extend well beyond the laboratory bench. The elimination of highly regulated and dangerous raw materials like 2-nitropropane and sodium hydride significantly lowers the barrier to entry for production facilities, reducing the need for specialized containment infrastructure and expensive safety protocols. This simplification of the raw material basket allows for more flexible sourcing strategies, as acetoxime and hydrogen peroxide are commodity chemicals available from a broad global supplier base, thereby enhancing supply chain reliability and reducing the risk of production stoppages due to precursor shortages.
- Cost Reduction in Manufacturing: The economic model of this new process is strengthened by the removal of expensive and hazardous reagents such as ceric ammonium nitrate and elemental bromine, which historically drove up the cost of goods sold. By utilizing a heterogeneous catalyst that can be separated via filtration and potentially regenerated, the process minimizes consumable costs and reduces the volume of hazardous waste requiring costly disposal. Furthermore, the high purity of the crude product reduces the energy and solvent consumption associated with extensive recrystallization or chromatographic purification steps, leading to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: Relying on commodity feedstocks like acetoxime and hydrogen peroxide insulates the production schedule from the volatility often seen in the market for specialized nitro-chemicals. The robust nature of the TS-1 catalyst and the aqueous reaction medium allow for scalable operations that are less prone to the batch-to-batch variability common in sensitive organometallic reactions. This stability ensures consistent delivery timelines for downstream customers in the security and pharmaceutical sectors, fostering stronger long-term partnerships and reducing the lead time for high-purity 2,3-dimethyl-2,3-dinitrobutane.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scalability, utilizing water as the primary solvent and generating benign byproducts that simplify effluent treatment. The absence of halogenated waste streams and heavy metal residues aligns perfectly with increasingly stringent environmental regulations, reducing the compliance burden and potential liability for manufacturing sites. This green chemistry approach not only future-proofs the production asset against regulatory tightening but also enhances the corporate sustainability profile, a key factor for multinational corporations seeking responsible suppliers for their critical material needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2,3-dimethyl-2,3-dinitrobutane via this novel catalytic route. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on process capabilities and product attributes. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this material into their supply chains or product formulations.
Q: Why is the TS-1 catalyzed route safer than traditional 2-nitropropane methods?
A: Traditional methods rely on highly toxic and hazardous raw materials like 2-nitropropane, sodium hydride, and bromine. The patented TS-1 route utilizes acetoxime and hydrogen peroxide, significantly reducing operational risks and eliminating the need for dangerous halogenated intermediates.
Q: How does this method improve product purity for explosive tracer applications?
A: The new synthesis method produces 2,3-dimethyl-2,3-dinitrobutane with a melting point of 210-212°C, significantly higher than the 129-130°C observed in prior art. This indicates a much lower impurity profile, which is critical for meeting International Civil Aviation Organization (ICAO) standards for explosive detection.
Q: Can the catalyst be recovered and reused in this process?
A: Yes, the modified TS-1 catalyst is heterogeneous and can be separated via filtration or centrifugation. The patent details a washing process using acetone to recover residual product from the catalyst, allowing the solid catalyst to be potentially regenerated or disposed of more safely than homogeneous heavy metal wastes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dimethyl-2,3-Dinitrobutane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing high-quality intermediates for security and pharmaceutical applications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering 2,3-dimethyl-2,3-dinitrobutane that meets stringent purity specifications, verified through our rigorous QC labs to guarantee the high melting point and low impurity profiles demanded by international standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain resilience and product performance.
