Advanced Manufacturing of Triazolinone Herbicide Intermediates via Palladium Catalysis
Advanced Manufacturing of Triazolinone Herbicide Intermediates via Palladium Catalysis
The global agrochemical sector continuously demands more efficient and safer pathways for producing high-value herbicide intermediates. Patent CN1159301C introduces a robust three-step synthetic route for preparing alkyl alpha-2-chloro-5-[4-(difluoromethyl)-4,5-dihydro-3-methyl-5-oxo-1H-1,2,4-triazol-1-yl]-2,4-substituted-benzene-propanoates, which are critical precursors for triazolinone herbicides. This technology represents a significant leap forward from older methodologies by replacing hazardous diazotization steps with a streamlined sequence of electrophilic halogenation, palladium-catalyzed coupling, and controlled chlorination. For R&D directors and procurement specialists, understanding this pathway is essential for securing a reliable agrochemical intermediate supplier capable of delivering high-purity materials with improved cost structures. The process specifically targets the synthesis of compounds where the benzene ring substituents can be independently selected from halo, alkyl, or alkoxy groups, providing versatility for various derivative productions.
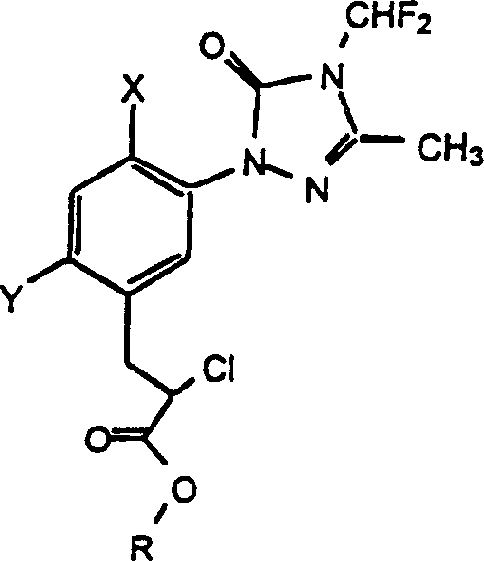
This innovative approach not only enhances the safety profile of the manufacturing process but also optimizes the impurity profile of the final active ingredient. By utilizing a palladium-catalyzed cross-coupling strategy, the method ensures precise construction of the carbon-carbon bond between the triazole core and the propanoate side chain. This level of control is paramount for meeting the stringent quality specifications required by major multinational crop protection companies. Furthermore, the final chlorination step employs sodium hypochlorite, a commodity chemical, rather than exotic chlorinating agents, which drastically simplifies the supply chain logistics and reduces raw material costs. As we delve deeper into the technical specifics, it becomes clear why this patent serves as a cornerstone for modern, scalable herbicide intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in US Patent 5,125,958, relied heavily on the diazotization of 5-amino intermediates using tert-butyl nitrite. This traditional approach presents significant safety and operational challenges for large-scale manufacturing. The use of tert-butyl nitrite involves handling unstable diazonium species, which poses thermal runaway risks and requires specialized equipment to manage exothermic events safely. Additionally, the subsequent reaction with ethyl acrylate necessitates the use of a large excess of the acrylate reagent to drive the reaction to completion, leading to substantial waste generation and increased downstream purification burdens. From a procurement perspective, the reliance on specialized diazotization reagents and the need for rigorous temperature control during the reaction create bottlenecks that limit production throughput and inflate the overall cost of goods sold. These factors collectively make the conventional route less attractive for high-volume commercial production where margin compression is a constant pressure.
The Novel Approach
In stark contrast, the methodology outlined in CN1159301C circumvents these hazards by employing a halogenation-first strategy. The process begins by introducing a halogen atom (bromine or iodine) directly onto the triazolone ring system using stable halogenating agents in the presence of strong acids like oleum. This transformation creates a highly reactive intermediate that is perfectly poised for the subsequent palladium-catalyzed coupling reaction. By shifting the synthetic logic from diazotization to cross-coupling, the new method eliminates the need for unstable diazonium salts and reduces the stoichiometric excess of reagents required. The coupling step utilizes readily available alkyl alkanoates and tertiary amines, which are far easier to source and handle than the reagents used in the legacy process. This strategic shift not only improves the inherent safety of the plant operations but also aligns with green chemistry principles by minimizing waste and improving atom economy, thereby offering a compelling value proposition for cost reduction in herbicide manufacturing.
Mechanistic Insights into Palladium-Catalyzed Coupling and Halogenation
The core of this synthetic innovation lies in the efficient functionalization of the triazolone scaffold. The first critical transformation involves the electrophilic aromatic substitution where a halogen atom is installed at the 5-position of the phenyl ring. As illustrated in the reaction scheme below, the triazolone precursor reacts with a halogenating agent such as iodine or bromine in a medium of fuming sulfuric acid. This acidic environment activates the halogen species, facilitating the attack on the electron-rich aromatic ring while the triazolone moiety remains stable under these conditions. The choice of oleum as a solvent is particularly advantageous as it ensures complete solubility of the starting material and drives the reaction to high conversion rates, often exceeding 90% as evidenced by gas chromatography analysis in the patent examples. This high conversion is crucial for minimizing the carryover of unreacted starting material into subsequent steps, which could otherwise complicate purification.
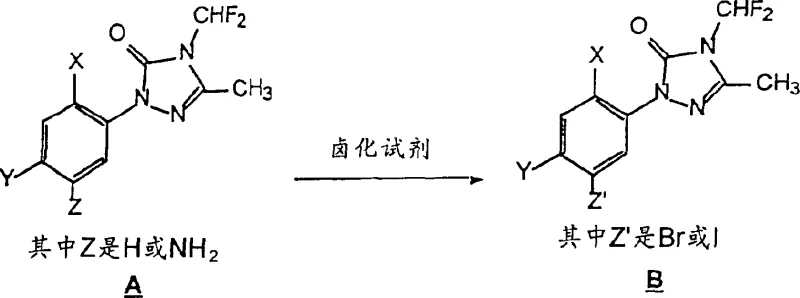
Following halogenation, the resulting 5-halo-triazolone intermediate undergoes a palladium-catalyzed coupling with an alkyl alkanoate, such as ethyl 3-hydroxy-2-methylenebutyrate or ethyl acrylate. This step likely proceeds via a Heck-type or similar palladium-catalyzed insertion mechanism, where the palladium catalyst oxidative adds into the carbon-halogen bond. The presence of a tertiary amine base, such as triethylamine or tributylamine, is essential to neutralize the acid generated during the catalytic cycle and to facilitate the regeneration of the active palladium species. The reaction is typically conducted at elevated temperatures ranging from 120°C to 135°C, which provides the necessary activation energy for the coupling to proceed efficiently. Impurity control is managed by carefully selecting the catalyst loading, typically between 0.01 to 0.03 molar equivalents, which balances reaction rate with the minimization of metal residues in the final product. This precise control over the catalytic cycle ensures that the final intermediate possesses the high purity levels required for downstream herbicide synthesis.
How to Synthesize Triazolinone Herbicide Intermediate Efficiently
Executing this synthesis requires careful attention to reaction parameters, particularly during the halogenation and coupling stages. The process begins with the dissolution of the triazolone starting material in fuming sulfuric acid, followed by the controlled addition of the halogen source at low temperatures to manage exotherms. Once the halogenated intermediate is isolated, it is subjected to the coupling reaction in a polar aprotic solvent like acetonitrile, using a palladium catalyst and a stoichiometric amount of the alkanoate coupling partner. The final step involves a one-pot chlorination and cyclization sequence using sodium hypochlorite and a base, which converts the acetyl side chain into the desired alpha-chloro propionate motif. For a detailed breakdown of the specific reagent quantities, temperature profiles, and workup procedures, please refer to the standardized synthesis guide below.
- Step 1: Halogenation of the triazolone precursor using iodine or bromine in fuming sulfuric acid to introduce a leaving group at the 5-position.
- Step 2: Palladium-catalyzed coupling of the halogenated intermediate with an alkyl alkanoate in the presence of a tertiary amine base.
- Step 3: Chlorination of the coupled intermediate using sodium hypochlorite followed by base treatment to yield the final alpha-chloro propionate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers tangible benefits that extend beyond mere technical feasibility. The elimination of hazardous diazotization reagents translates directly into reduced safety compliance costs and lower insurance premiums for manufacturing facilities. Furthermore, the use of commodity chemicals like sodium hypochlorite and common organic acids ensures a stable and resilient supply chain, mitigating the risk of production stoppages due to raw material shortages. The robustness of the palladium-catalyzed step allows for flexible sourcing of catalysts, as the process tolerates various palladium sources including heterogeneous catalysts like palladium on carbon, which can be easily filtered and potentially recycled. This flexibility empowers procurement teams to negotiate better pricing and secure long-term supply agreements without being locked into single-source proprietary reagents.
- Cost Reduction in Manufacturing: The streamlined three-step sequence significantly lowers the overall cost of production by reducing the number of unit operations and eliminating the need for expensive, specialized reagents associated with diazotization. The high conversion rates observed in the halogenation and coupling steps minimize raw material waste, leading to substantial cost savings in material consumption. Additionally, the ability to perform the final chlorination in the same reaction vessel as the preceding steps (telescoping) reduces solvent usage and energy consumption associated with isolation and drying processes. These efficiencies collectively contribute to a lower cost of goods sold, allowing for more competitive pricing in the global agrochemical market.
- Enhanced Supply Chain Reliability: By relying on widely available industrial chemicals such as bromine, iodine, acetic acid, and sodium hypochlorite, the process decouples production from volatile specialty chemical markets. The tolerance for various palladium catalysts further diversifies the supply base, ensuring that catalyst shortages do not impact production schedules. The moderate reaction conditions, particularly the ambient temperature options for the halogenation step, reduce the strain on utility infrastructure, making the process suitable for a wider range of manufacturing sites globally. This geographical flexibility enhances supply chain resilience and shortens lead times for delivering high-purity agrochemical intermediates to customers.
- Scalability and Environmental Compliance: The process is inherently scalable, with reaction conditions that translate well from laboratory benchtop to multi-ton commercial reactors. The use of aqueous workups and standard extraction techniques simplifies waste management and facilitates the recovery of solvents for reuse. The avoidance of heavy metal contaminants in the final product, achieved through optimized catalyst loading and purification, ensures compliance with stringent environmental regulations regarding metal residues in agricultural products. This alignment with regulatory standards reduces the burden of environmental reporting and permits, accelerating the time to market for new herbicide formulations derived from these intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is critical for R&D teams evaluating process transfer and for commercial teams assessing the viability of long-term supply partnerships. The answers reflect the practical realities of scaling this chemistry while maintaining the high quality standards expected in the fine chemical industry.
Q: How does this synthesis method improve safety compared to prior art?
A: Unlike previous methods relying on hazardous diazotization with tert-butyl nitrite and large excesses of ethyl acrylate, this patent utilizes a controlled halogenation and palladium-catalyzed coupling sequence, significantly reducing thermal risks and reagent hazards.
Q: What catalysts are suitable for the coupling step?
A: The process supports various palladium sources including palladium(II) acetate, palladium on carbon, and dibenzonitrile dichloropalladium(II), offering flexibility in catalyst selection based on cost and availability.
Q: Can this process be scaled for commercial production?
A: Yes, the reaction conditions operate at moderate temperatures (ambient to 135°C) and utilize standard industrial reagents like sodium hypochlorite and common solvents, making it highly amenable to multi-ton scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazolinone Herbicide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of robust intermediate supply chains for the global agrochemical industry. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our facility is equipped to handle the specific requirements of palladium-catalyzed reactions and halogenation processes, guaranteeing consistent quality and reliability for your herbicide production needs.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to validate our capabilities against your internal quality metrics. Let us collaborate to drive efficiency and innovation in your herbicide manufacturing operations.
