Advanced Aminosulfone Synthesis: Technical Breakthroughs for Commercial Scale-Up
Advanced Aminosulfone Synthesis: Technical Breakthroughs for Commercial Scale-Up
The pharmaceutical industry's relentless pursuit of potent anti-inflammatory agents has placed significant focus on Phosphodiesterase 4 (PDE4) inhibitors, a class of compounds critical for treating asthma, COPD, and autoimmune disorders. Central to the synthesis of these advanced therapeutics is the efficient production of specific aminosulfone intermediates. Patent CN102209709A introduces a transformative methodology for preparing these vital building blocks, specifically targeting compounds like 2-(3-ethoxy-4-methoxyphenyl)-1-(methylsulfonyl)ethan-2-amine. This patent outlines a robust synthetic pathway that bypasses the inefficiencies of traditional racemic synthesis and chiral resolution, offering a direct route to high-purity intermediates essential for downstream isoindoline-based PDE4 modulator production.
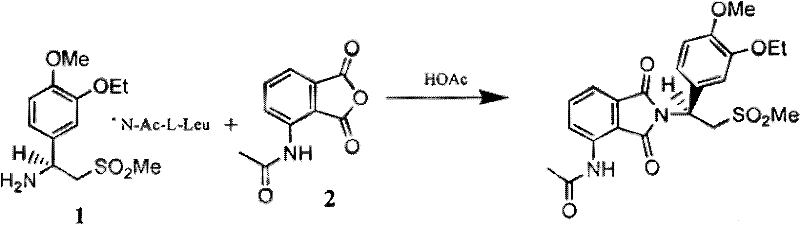
For R&D directors and process chemists, the significance of this disclosure lies in its ability to streamline the supply chain for complex API intermediates. The traditional reliance on resolving racemic mixtures often introduces bottlenecks related to yield loss and the procurement of costly chiral resolving agents. By shifting the paradigm to a constructive synthesis via nitrile and enamine intermediates, this technology not only enhances chemical efficiency but also aligns with modern green chemistry principles by reducing waste streams associated with resolution byproducts. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic shifts is crucial for evaluating long-term process viability and cost structures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of chiral aminosulfones required for PDE4 inhibitors involved a cumbersome multi-step sequence starting from substituted benzaldehydes. The conventional route typically proceeds through the formation of a racemic amino sulfone, which subsequently necessitates a resolution step using chiral acids such as N-Ac-L-Leucine. This approach is inherently flawed from a manufacturing perspective because the maximum theoretical yield for the desired enantiomer is capped at 50%, assuming perfect resolution efficiency, which is rarely the case in practice. Furthermore, the recycling of the unwanted enantiomer adds additional processing steps, solvent consumption, and energy usage, drastically inflating the cost of goods sold (COGS). The extended process cycle time associated with crystallization and mother liquor recovery also creates significant lead time challenges for supply chain managers trying to meet tight clinical or commercial deadlines.
The Novel Approach
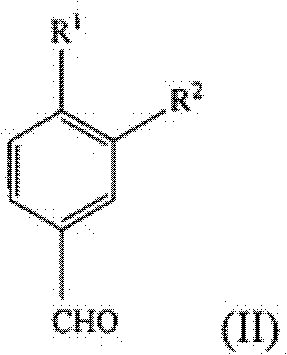
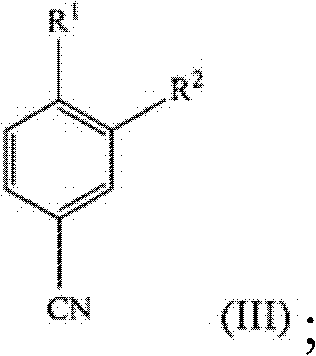
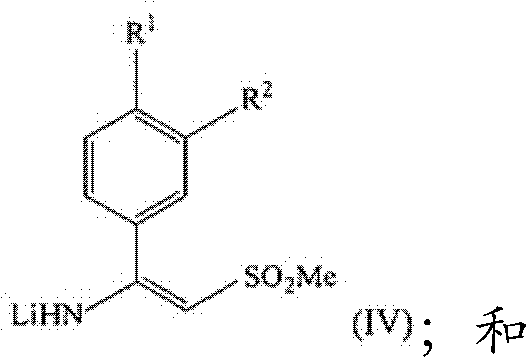
In stark contrast, the methodology disclosed in the patent data presents a streamlined, non-racemic synthetic strategy that constructs the chiral center or preserves stereochemistry more effectively through selective reduction. The new route initiates with the conversion of a substituted benzaldehyde (Formula II) directly into a nitrile (Formula III) using hydroxylamine derivatives. This nitrile is then subjected to nucleophilic addition with lithiated methyl sulfone to generate an enamine intermediate (Formula IV), which is subsequently reduced to the target amine. This sequence eliminates the need for early-stage resolution, thereby theoretically doubling the material throughput compared to racemic routes. The process utilizes widely available reagents and operates under mild conditions, making it an attractive candidate for cost reduction in API manufacturing where margin compression is a constant pressure.
Mechanistic Insights into Nitrile Formation and Reductive Amination
The core of this technological advancement rests on the efficient transformation of the aldehyde functionality into a nitrile group, followed by a controlled carbon-carbon bond formation. The first critical step involves the reaction of the aldehyde of general formula (II) with hydroxylamine or its salts. This condensation reaction is facilitated in polar aprotic or protic solvents such as acetonitrile, formic acid, or acetic acid. The reaction kinetics are favorable at elevated temperatures, typically between 65°C to 85°C, allowing for substantial completion within approximately 5 hours. This step is pivotal as it activates the aromatic ring for the subsequent nucleophilic attack while establishing the nitrogen framework required for the final amine structure.
Following nitrile formation, the process advances to the generation of the enamine intermediate. This involves the deprotonation of dimethyl sulfone using a strong base like n-butyllithium to form the lithiated species LiCH2SO2Me. This nucleophile attacks the nitrile carbon of Formula (III) to yield the enamine of Formula (IV). The control of temperature during this exothermic step is critical, with optimal ranges identified between 0°C to 25°C to prevent side reactions and ensure high regioselectivity. The final reduction of the enamine to the amine is achieved using hydride sources such as sodium borohydride (NaBH4) or sodium triacetoxyborohydride in the presence of an acid source like trifluoroacetic acid or acetic acid. This reductive step is highly chemoselective, reducing the C=N bond without affecting other sensitive functional groups on the aromatic ring, thus ensuring the high purity specifications required for pharmaceutical applications.
How to Synthesize 2-(3-ethoxy-4-methoxyphenyl)-1-(methylsulfonyl)ethan-2-amine Efficiently
The practical implementation of this synthesis requires precise control over stoichiometry and thermal profiles to maximize yield and safety. The patent provides detailed embodiments demonstrating the scalability of the route, with specific examples showing yields exceeding 95% for the nitrile formation and over 76% for the final amination step. These high yields are indicative of a robust process capable of handling large-scale batches with minimal variance. For process engineers looking to implement this technology, the detailed standardized synthesis steps provided in the patent serve as a foundational protocol that can be further optimized for specific reactor configurations and throughput requirements.
- Convert the substituted benzaldehyde (Formula II) to the corresponding nitrile (Formula III) using hydroxylamine or its salt in a solvent like acetonitrile or formic acid at 65-85°C.
- React the nitrile (Formula III) with lithiated methyl sulfone (LiCH2SO2Me) in THF/hexane at 0-25°C to form the enamine intermediate (Formula IV).
- Reduce the enamine (Formula IV) using sodium borohydride or sodium triacetoxyborohydride in the presence of an acid source, followed by hydrolysis to yield the final aminosulfone (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthetic route offers profound strategic advantages that extend beyond simple chemical yield improvements. The elimination of chiral resolution agents represents a direct removal of a high-cost, low-availability input from the bill of materials. Resolving agents like N-Ac-L-Leucine are not only expensive but can also be subject to supply volatility due to their fermentation-based production. By circumventing this dependency, manufacturers can achieve significant cost savings and stabilize their raw material sourcing strategies. Furthermore, the simplified workflow reduces the number of unit operations, which translates to lower utility consumption, reduced labor hours, and decreased facility occupancy time, all of which contribute to a leaner and more cost-effective manufacturing model.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the synthetic sequence. By removing the resolution step, the process avoids the inherent 50% yield loss associated with separating enantiomers from a racemic mixture. This effectively doubles the output per kilogram of starting material compared to traditional methods. Additionally, the reagents used, such as hydroxylamine hydrochloride and dimethyl sulfone, are commodity chemicals with stable pricing and abundant global supply. The reduction in solvent usage and waste treatment costs, stemming from fewer isolation and purification steps, further enhances the overall economic viability of producing these high-value pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, non-specialized reagents and solvents. The process relies on standard organic solvents like THF, hexane, and acetonitrile, which are readily available from multiple global suppliers, mitigating the risk of single-source bottlenecks. The reaction conditions are moderate, avoiding the need for extreme cryogenic temperatures or high-pressure equipment that often complicate logistics and increase capital expenditure. This operational simplicity ensures that production schedules can be maintained consistently, reducing lead times for high-purity pharmaceutical intermediates and enabling faster response to market demand fluctuations.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly improved due to the reduction in waste generation. Traditional resolution processes generate substantial amounts of salt waste and mother liquors containing the unwanted enantiomer, posing disposal challenges. The new route's atom economy is superior, generating fewer byproducts and simplifying effluent treatment. The ability to operate at near-ambient temperatures for key steps reduces the energy footprint of the manufacturing process. These factors collectively facilitate easier regulatory approval and compliance with increasingly stringent environmental regulations, making the process sustainable for long-term commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aminosulfone synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the process capabilities and limitations. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this new aminosulfone synthesis route over conventional resolution methods?
A: The novel route described in patent CN102209709A eliminates the need for chiral resolution using expensive agents like N-Ac-L-Leu. By proceeding through a nitrile and enamine intermediate, the process achieves significantly higher overall yields (up to 95.5% for the nitrile step and 76.1% for the final amine) compared to the low-efficiency racemic synthesis and splitting cycles of traditional methods.
Q: What represent the critical reaction conditions for the nitrile formation step?
A: The conversion of the aldehyde to the nitrile is optimally performed in solvents such as acetonitrile, formic acid, or acetic acid. The reaction temperature is maintained between 65°C to 85°C, typically requiring about 5 hours to reach substantial completion, ensuring high purity without the need for complex purification techniques.
Q: Is this process scalable for industrial manufacturing of PDE4 inhibitor intermediates?
A: Yes, the process utilizes common industrial solvents like THF, hexane, and acetonitrile, and operates at moderate temperatures (0°C to 60°C). The elimination of difficult separation steps and the use of robust reagents like sodium borohydride make it highly suitable for commercial scale-up from kilogram to multi-ton production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(3-ethoxy-4-methoxyphenyl)-1-(methylsulfonyl)ethan-2-amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of next-generation anti-inflammatory therapies. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications and utilizing rigorous QC labs to guarantee that every batch of aminosulfone compounds meets the exacting standards required by global regulatory bodies. Our infrastructure is designed to support the complex chemistry involved in PDE4 inhibitor synthesis, providing a secure and reliable foundation for your drug development pipeline.
We invite you to collaborate with us to leverage these advanced synthetic methodologies for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both the technical and commercial outcomes of your pharmaceutical manufacturing initiatives.
