Industrial Scale-Up of N-Nitroisourea Intermediates via Advanced SO3-Assisted Nitration Technology
Introduction to Advanced Isourea Nitration Technology
The chemical landscape for producing critical heterocyclic intermediates is constantly evolving, driven by the dual needs of environmental compliance and economic efficiency. Patent CN101400647A introduces a transformative methodology for the industrial production of N-nitroisoureas, a class of compounds that serves as indispensable building blocks in the synthesis of next-generation pharmaceuticals and high-performance agrochemicals. This specific intellectual property addresses the longstanding inefficiencies associated with traditional nitration protocols, which often suffer from excessive acid consumption and difficult downstream processing. By leveraging the unique chemical properties of sulfur trioxide within a sulfuric acid medium, this technology enables a more atom-economical pathway that significantly curtails the generation of hazardous waste streams. For global procurement leaders and R&D directors, understanding the nuances of this patent is essential for securing a reliable agrochemical intermediate supplier capable of delivering consistent quality while adhering to stringent environmental standards. The following analysis dissects the technical merits and commercial implications of this novel synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-nitroisourea derivatives has relied heavily on classical nitration conditions involving concentrated sulfuric acid and fuming nitric acid. While chemically feasible, these legacy processes are plagued by substantial operational drawbacks that impact both cost and sustainability profiles. The primary issue lies in the stoichiometric generation of water during the nitration event, which dilutes the sulfuric acid solvent and necessitates the use of vast excesses of acid to maintain reaction viability. This results in the production of massive quantities of spent acid waste, creating a severe burden on waste treatment facilities and inflating the overall cost of goods sold. Furthermore, the harsh acidic environment often leads to side reactions and decomposition of sensitive functional groups, complicating the purification process and reducing the overall yield of the desired active pharmaceutical ingredient precursor. These factors collectively render conventional methods increasingly obsolete in a modern regulatory environment focused on green chemistry principles.
The Novel Approach
In stark contrast to traditional methodologies, the process disclosed in CN101400647A utilizes a strategic addition of sulfur trioxide to the reaction matrix, fundamentally altering the thermodynamics and kinetics of the nitration. 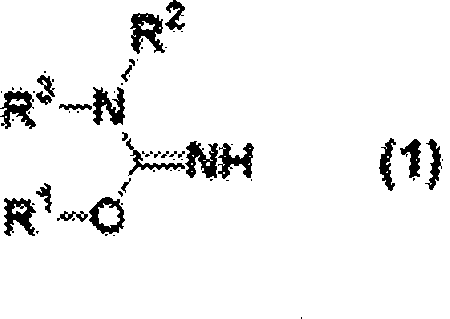 As illustrated by the general structure of the starting material, the isourea backbone is susceptible to hydrolysis and degradation under prolonged exposure to aqueous acidic conditions. The introduction of sulfur trioxide acts as a potent dehydrating agent, effectively scavenging the water molecules produced during the conversion of the isourea to the nitro-isourea derivative. This mechanism allows the reaction to proceed efficiently with a drastically reduced molar ratio of sulfuric acid, often lowering the requirement from traditional excesses to near-stoichiometric levels relative to the substrate. This innovation not only minimizes waste acid volume but also enhances the stability of the reaction mixture, leading to cleaner product profiles and simplified isolation procedures.
As illustrated by the general structure of the starting material, the isourea backbone is susceptible to hydrolysis and degradation under prolonged exposure to aqueous acidic conditions. The introduction of sulfur trioxide acts as a potent dehydrating agent, effectively scavenging the water molecules produced during the conversion of the isourea to the nitro-isourea derivative. This mechanism allows the reaction to proceed efficiently with a drastically reduced molar ratio of sulfuric acid, often lowering the requirement from traditional excesses to near-stoichiometric levels relative to the substrate. This innovation not only minimizes waste acid volume but also enhances the stability of the reaction mixture, leading to cleaner product profiles and simplified isolation procedures.
Mechanistic Insights into SO3-Assisted Electrophilic Nitration
The core of this technological advancement lies in the precise manipulation of the nitrating species' activity through water management. In a standard nitration, the active electrophile is the nitronium ion (NO2+), generated by the protonation of nitric acid by sulfuric acid. However, as the reaction progresses, water is released, shifting the equilibrium back towards unreactive nitric acid and requiring immense volumes of sulfuric acid to push the equilibrium forward. 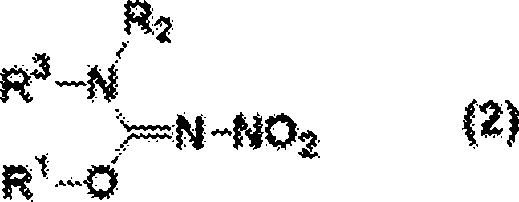 The patented method circumvents this by introducing sulfur trioxide, which reacts rapidly with the generated water to form additional sulfuric acid in situ. This dynamic regeneration of the acidic medium ensures that the concentration of the nitronium ion remains high throughout the reaction course without the need for initial bulk solvent loading. Consequently, the reaction can be conducted at milder temperatures, typically between 10°C and 25°C, which preserves the integrity of thermally labile substituents on the isourea ring. This level of control is paramount for R&D teams aiming to synthesize complex analogs where functional group tolerance is critical for biological activity.
The patented method circumvents this by introducing sulfur trioxide, which reacts rapidly with the generated water to form additional sulfuric acid in situ. This dynamic regeneration of the acidic medium ensures that the concentration of the nitronium ion remains high throughout the reaction course without the need for initial bulk solvent loading. Consequently, the reaction can be conducted at milder temperatures, typically between 10°C and 25°C, which preserves the integrity of thermally labile substituents on the isourea ring. This level of control is paramount for R&D teams aiming to synthesize complex analogs where functional group tolerance is critical for biological activity.
Furthermore, the impurity profile of the resulting N-nitroisourea is significantly improved due to the suppression of hydrolytic side pathways. In conventional acidic media, the O-alkyl bond of the isourea can be prone to cleavage, leading to urea byproducts that are difficult to separate. The anhydrous conditions promoted by the sulfur trioxide additive mitigate this risk, ensuring that the nitrogen atom is selectively nitrated while the oxygen-carbon bond remains intact. This selectivity translates directly into higher crude purity, reducing the load on downstream purification units such as crystallization or chromatography columns. For manufacturing scientists, this means a more robust process window where minor deviations in temperature or addition rates do not catastrophically impact the final quality attributes of the batch. The ability to consistently produce high-purity intermediates is a key differentiator for any contract development and manufacturing organization (CDMO) serving the regulated pharmaceutical market.
How to Synthesize N-Nitroisourea Efficiently
Implementing this synthesis route requires careful attention to reagent quality and addition protocols to maximize the benefits of the sulfur trioxide effect. The process begins with the dissolution of the isourea sulfate salt in a pre-mixed solvent system containing concentrated sulfuric acid and oleum, ensuring that the free sulfur trioxide concentration is sufficient to handle the theoretical water output of the reaction. Detailed standardized synthesis steps see the guide below. This controlled environment allows for the safe handling of energetic nitration reactions while maintaining the high throughput necessary for commercial viability. Operators must monitor the exotherm closely during the addition of the nitric acid, as the rapid consumption of water by sulfur trioxide can initially mask the heat of reaction before the system stabilizes.
- Dissolve the isourea sulfate salt (Formula 1) in a mixture of concentrated sulfuric acid and oleum (fuming sulfuric acid) to ensure the presence of free sulfur trioxide.
- Maintain the reaction temperature between 10°C and 25°C while slowly adding a nitrating agent, preferably 90-100% nitric acid, over a period of 1 hour.
- Stir the reaction mixture for 6 to 8 hours to ensure complete conversion, then quench into ice water and neutralize to isolate the crude N-nitroisourea product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, adopting this SO3-assisted nitration technology offers profound advantages that extend beyond simple yield improvements. The reduction in sulfuric acid usage directly correlates to a decrease in raw material procurement costs and, more significantly, a drastic reduction in waste disposal fees associated with spent acid neutralization. Cost Reduction in Manufacturing: By minimizing the volume of hazardous waste generated per kilogram of product, manufacturers can achieve substantial cost savings in environmental compliance and waste treatment operations. The elimination of excess acid handling also reduces the corrosion load on reactor vessels and piping, extending equipment lifespan and lowering maintenance capital expenditures. Furthermore, the higher selectivity of the reaction reduces the loss of expensive starting materials to byproduct formation, optimizing the overall material balance and improving the gross margin potential for high-value intermediates.
Enhanced Supply Chain Reliability: The robustness of this chemical process contributes to greater supply chain stability by reducing the risk of batch failures due to impurity buildup. Traditional nitration methods often require extensive rework or recycling of off-spec material, which can disrupt delivery schedules and strain inventory buffers. The cleaner reaction profile enabled by sulfur trioxide ensures that batches meet specification on the first pass, facilitating just-in-time delivery models for downstream drug substance manufacturers. Additionally, the use of commodity chemicals like sulfuric acid and nitric acid, rather than exotic catalysts, ensures that the supply of reagents remains secure and unaffected by niche market fluctuations. This reliability is crucial for long-term supply agreements where continuity of supply is a primary contractual obligation.
Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production often reveals hidden bottlenecks related to heat transfer and mixing, particularly in highly exothermic nitrations. The moderated reaction conditions of this patent, operating effectively at near-ambient temperatures, simplify the engineering requirements for large-scale reactors. This ease of scale-up allows for faster technology transfer and quicker time-to-market for new products entering the pipeline. Moreover, the alignment with green chemistry principles by reducing waste acid generation positions the manufacturer favorably against increasingly strict environmental regulations globally. Companies that proactively adopt such cleaner technologies future-proof their operations against regulatory tightening, ensuring uninterrupted production capabilities in jurisdictions with rigorous environmental oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitration technology in an industrial setting. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios or outsourcing strategies.
Q: What is the primary advantage of using sulfur trioxide in isourea nitration?
A: The addition of sulfur trioxide (SO3) effectively scavenges water produced during the nitration reaction, maintaining the strength of the nitrating mixture and allowing for a significant reduction in the total volume of sulfuric acid solvent required, thereby minimizing waste acid disposal costs.
Q: Can this process be scaled for commercial production of agrochemical intermediates?
A: Yes, the process operates at mild temperatures (10-25°C) and utilizes standard industrial reagents like oleum and nitric acid, making it highly suitable for large-scale commercial manufacturing of complex agrochemical and pharmaceutical intermediates.
Q: What purity levels can be achieved with this nitration method?
A: Experimental data from the patent indicates reaction yields exceeding 94%, and subsequent purification steps such as crystallization or chromatography can achieve high-purity specifications required for GMP-grade pharmaceutical synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Nitroisourea Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent theory to commercial reality requires deep technical expertise and state-of-the-art infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the nuances of SO3-assisted nitration are managed with precision and safety. We operate stringent purity specifications and maintain rigorous QC labs equipped to analyze complex impurity profiles, guaranteeing that every batch of N-nitroisourea intermediate meets the exacting standards required for pharmaceutical and agrochemical applications. Our commitment to process optimization allows us to deliver cost-effective solutions without compromising on quality or regulatory compliance.
We invite potential partners to engage with our technical procurement team to discuss how this advanced nitration technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this greener, more efficient synthetic route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your supply chain.
