Advanced Synthesis of Alkylcyclohexylphenol Intermediates for High-Performance TFT-LCD Displays
Introduction to Next-Generation Liquid Crystal Intermediate Synthesis
The rapidly evolving landscape of display technology, particularly within the Thin Film Transistor-Liquid Crystal Display (TFT-LCD) sector, demands intermediates of exceptional purity and structural precision. Patent CN103553878A introduces a groundbreaking preparation method for alkylcyclohexylphenol liquid crystal intermediate compounds, addressing critical bottlenecks in existing manufacturing protocols. This innovation focuses on the synthesis of compounds characterized by the general formula (I), where specific alkyl chains and fluorine substitutions are meticulously arranged to optimize dielectric anisotropy and viscosity profiles. 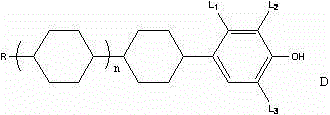 The disclosed methodology represents a significant leap forward for reliable electronic chemical suppliers, offering a pathway to high-quality materials that support the high voltage retention and fast response times required by advanced display panels. By streamlining the synthetic route and eliminating hazardous reagents, this technology not only enhances product quality but also aligns with modern environmental and safety standards essential for sustainable chemical manufacturing.
The disclosed methodology represents a significant leap forward for reliable electronic chemical suppliers, offering a pathway to high-quality materials that support the high voltage retention and fast response times required by advanced display panels. By streamlining the synthetic route and eliminating hazardous reagents, this technology not only enhances product quality but also aligns with modern environmental and safety standards essential for sustainable chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenyl dicyclic hexane liquid crystals, particularly those with difluoro-methoxy end groups, has been plagued by inefficient and hazardous chemical processes. Prior art, such as methods reported by Merck or academic institutions, often relies on the use of n-Butyl Lithium for lithiation steps, which necessitates cryogenic conditions below -70°C to maintain reagent stability and control reactivity. This requirement imposes severe energy burdens and safety risks, as n-Butyl Lithium is pyrophoric and reacts violently with moisture. Furthermore, traditional routes frequently involve multi-step sequences including separate etherification, reduction, and deprotection stages, leading to cumulative yield losses and complex purification challenges. The use of expensive catalysts like palladium on carbon in multiple steps, along with corrosive reagents such as aluminum chloride and hydrogen bromide, generates substantial acidic waste gas, creating significant environmental compliance hurdles for cost reduction in electronic chemical manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a robust Grignard reaction followed by a highly efficient one-step catalytic hydrogenation strategy. This method replaces the dangerous n-Butyl Lithium with magnesium chips, allowing the reaction to proceed at moderate temperatures ranging from 60°C to 85°C, thereby drastically simplifying the thermal management requirements. A pivotal innovation lies in the hydrogenation step, where a single catalytic operation simultaneously reduces the cyclohexenyl double bond and cleaves the benzyl protecting group. This convergence of reactions eliminates the need for intermediate isolation and separate deprotection steps, significantly shortening the overall production cycle. By employing neutral Raney nickel or palladium catalysts under controlled pressure, the process achieves high conversion rates while minimizing the formation of by-products, ensuring that the commercial scale-up of complex polymer additives and liquid crystal precursors remains economically viable and environmentally responsible.
Mechanistic Insights into Grignard Coupling and Catalytic Hydrogenation
The core of this synthetic breakthrough relies on the precise formation and utilization of a benzyloxyphenylmagnesium bromide Grignard reagent. In the initial stage, benzyloxybromobenzene reacts with magnesium chips in a mixed solvent system of tetrahydrofuran and toluene. The choice of solvent is critical; the mixture facilitates the initiation of the Grignard formation at 60°C and sustains the exothermic reaction safely up to 75°C. 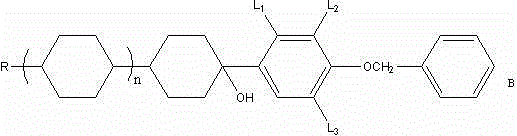 Once formed, this nucleophilic reagent attacks the carbonyl group of the alkyl dicyclo ketone (Raw Material A), forming a tertiary alcohol intermediate. Subsequent acidic hydrolysis yields compound B without the need for rigorous purification, as the crude product is directly fed into the dehydration step. This telescoping of steps minimizes material handling losses and solvent consumption, demonstrating a sophisticated understanding of process chemistry that prioritizes efficiency and throughput for high-purity OLED material and liquid crystal precursor production.
Once formed, this nucleophilic reagent attacks the carbonyl group of the alkyl dicyclo ketone (Raw Material A), forming a tertiary alcohol intermediate. Subsequent acidic hydrolysis yields compound B without the need for rigorous purification, as the crude product is directly fed into the dehydration step. This telescoping of steps minimizes material handling losses and solvent consumption, demonstrating a sophisticated understanding of process chemistry that prioritizes efficiency and throughput for high-purity OLED material and liquid crystal precursor production.
Following dehydration to form the olefinic compound C, the process enters its most technically distinct phase: the catalytic hydrogenation. Unlike conventional methods that might require distinct conditions for alkene reduction and benzyl ether cleavage, this protocol achieves both transformations in a single reactor vessel. Using a neutral Raney nickel catalyst at temperatures around 80°C and pressures of 0.2 MPa, the system effectively saturates the cyclohexene ring while concurrently hydrogenolyzing the benzyl-oxygen bond. 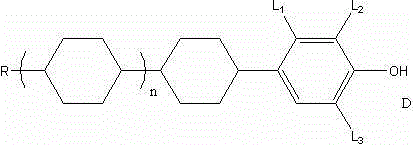 This dual-functionality is mechanistically advantageous as it prevents the accumulation of partially reduced intermediates that could complicate downstream isomerization. The resulting cis-trans mixture (Compound D) is then subjected to a base-catalyzed isomerization in a polar aprotic solvent like DMF. The use of potassium hydroxide facilitates the thermodynamic equilibration towards the more stable trans-isomer, which is crucial for the mesogenic properties of the final liquid crystal material. This meticulous control over stereochemistry ensures the final product meets the rigorous structural specifications demanded by display manufacturers.
This dual-functionality is mechanistically advantageous as it prevents the accumulation of partially reduced intermediates that could complicate downstream isomerization. The resulting cis-trans mixture (Compound D) is then subjected to a base-catalyzed isomerization in a polar aprotic solvent like DMF. The use of potassium hydroxide facilitates the thermodynamic equilibration towards the more stable trans-isomer, which is crucial for the mesogenic properties of the final liquid crystal material. This meticulous control over stereochemistry ensures the final product meets the rigorous structural specifications demanded by display manufacturers.
How to Synthesize Alkylcyclohexylphenol Efficiently
The synthesis of these high-value intermediates requires strict adherence to the optimized reaction parameters outlined in the patent to ensure maximum yield and purity. The process begins with the careful preparation of the Grignard reagent under nitrogen protection, followed by the controlled addition of the ketone substrate to manage exotherms. Detailed standard operating procedures regarding temperature gradients, solvent ratios, and catalyst loading are essential for reproducibility. For a comprehensive guide on the specific molar ratios, reaction times, and workup procedures including recrystallization solvents, please refer to the standardized synthesis steps provided below.
- React benzyloxybromobenzene with magnesium chips to form a Grignard reagent, then couple with alkyl dicyclo ketone followed by acidic hydrolysis to obtain compound B.
- Perform reflux dehydration on compound B in an organic solvent and organic acid system to synthesize the olefinic compound C with high purity.
- Conduct a one-step catalytic hydrogenation on compound C to simultaneously reduce the cyclohexenyl double bond and remove the benzyl protecting group, yielding cis-trans compound D.
- Execute an isomerization reaction on compound D in a strong alkaline system to convert the cis-isomer to the thermodynamically stable trans-product, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers transformative benefits that extend beyond simple chemical yield improvements. The elimination of n-Butyl Lithium removes a major supply chain vulnerability, as this reagent is not only expensive but also subject to strict transportation and storage regulations due to its hazardous classification. By substituting it with magnesium chips, the process leverages a commodity chemical that is widely available, stable, and cost-effective, leading to substantial cost savings in raw material procurement. Furthermore, the simplification of the synthetic route from five or more steps down to a streamlined four-step sequence reduces the total processing time and equipment occupancy, enhancing overall plant throughput and reducing lead time for high-purity electronic chemicals.
- Cost Reduction in Manufacturing: The consolidation of the reduction and deprotection steps into a single hydrogenation event significantly lowers utility costs and solvent consumption. Traditional methods often require separate reactors and extensive washing procedures for each discrete step, generating large volumes of wastewater and spent solvent. By telescoping these operations, the new method minimizes the physical footprint required for production and reduces the load on waste treatment facilities. Additionally, the avoidance of expensive transition metal catalysts in favor of recyclable Raney nickel further drives down the cost of goods sold, making the final liquid crystal intermediate more competitive in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous reagents like magnesium and toluene ensures a more resilient supply chain compared to routes dependent on cryogenic reagents. The operational simplicity allows for easier scaling from pilot batches to full commercial production, mitigating the risks associated with technology transfer. Since the process operates at atmospheric or low pressures and moderate temperatures, it can be implemented in standard stainless steel reactors without the need for specialized Hastelloy equipment required for highly corrosive acids or extreme cold, thereby increasing the pool of qualified contract manufacturing organizations capable of producing these intermediates reliably.
- Scalability and Environmental Compliance: Environmental sustainability is a critical metric for modern chemical supply chains, and this process excels by minimizing the generation of acidic waste gases and heavy metal contaminants. The absence of aluminum chloride and hydrogen bromide eliminates the need for complex scrubbing systems to handle corrosive off-gases, simplifying regulatory compliance. Moreover, the high purity achieved through recrystallization reduces the need for energy-intensive chromatographic purification, lowering the carbon footprint of the manufacturing process. This alignment with green chemistry principles not only future-proofs the supply against tightening environmental regulations but also appeals to end-users who prioritize sustainability in their sourcing decisions for display and optoelectronic materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these alkylcyclohexylphenol intermediates. The answers are derived directly from the experimental data and process descriptions found in the underlying patent literature, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is vital for evaluating the feasibility of integrating this technology into existing production lines or sourcing strategies for next-generation display materials.
Q: How does this new method improve safety compared to traditional n-Butyl Lithium routes?
A: Traditional methods often rely on n-Butyl Lithium, which requires extremely low temperatures and poses significant safety hazards due to its pyrophoric nature. This novel protocol utilizes a Grignard reagent formed from magnesium chips, which operates at much milder temperatures (60°C to 85°C) and eliminates the need for cryogenic conditions, drastically reducing operational risks and energy consumption associated with cooling.
Q: What is the key advantage of the hydrogenation step in this synthesis?
A: The process features a unique one-step catalytic hydrogenation that achieves two critical transformations simultaneously: it reduces the double bond in the cyclohexenyl ring and removes the benzyl protecting group. This convergence of steps significantly shortens the synthetic route, reduces solvent usage, and minimizes waste generation compared to sequential reduction and deprotection strategies found in older patents.
Q: What purity levels can be achieved for TFT-LCD applications?
A: Through optimized recrystallization processes using specific solvent systems like toluene and isopropanol, the final trans-alkylcyclohexylphenol intermediates achieve purity levels exceeding 99.85%. This high degree of chemical purity is essential for meeting the stringent voltage holding ratio and response time requirements of modern Thin Film Transistor Liquid Crystal Displays (TFT-LCD).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkylcyclohexylphenol Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing the technical expertise to translate complex patent methodologies like CN103553878A into robust commercial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacture is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of alkylcyclohexylphenol intermediate meets the exacting standards required for TFT-LCD applications. Our commitment to quality assurance guarantees that the dielectric and optical properties of the final liquid crystal mixtures remain consistent and reliable.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance your supply chain resilience and product performance in the competitive electronic materials market.
