Advanced Nickel-Catalyzed Isomerization for Scalable Production of Complex Pharmaceutical Intermediates
The chemical landscape for synthesizing polysubstituted α,β-unsaturated ketones has undergone a significant transformation with the disclosure of patent CN110790649A, published on February 14, 2020. This pivotal intellectual property introduces a robust methodology that leverages nickel catalysis to drive the thermodynamic isomerization of β,γ-unsaturated ketones into their more stable conjugated α,β-isomers. For R&D directors and process chemists, this represents a paradigm shift away from the restrictive and often hazardous traditional routes involving cyclopropane ring-opening or non-selective Aldol condensations. The core innovation lies in the utilization of a Ni(cod)2 catalyst system paired with bulky N-heterocyclic carbene (NHC) ligands, which effectively activates carbon-carbon bonds to facilitate double bond migration. This technical breakthrough not only expands the accessible chemical space for complex synthons but also aligns perfectly with modern green chemistry principles by utilizing base metals and stable substrates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trisubstituted α,β-unsaturated ketones has been plagued by significant synthetic bottlenecks that hinder efficient commercial production. Traditional approaches predominantly rely on Aldol condensation followed by dehydration, a pathway that suffers from severe selectivity issues unless the reacting ketones are identical or one lacks α-protons. When dissimilar ketones are employed, the reaction invariably yields complex mixtures of regioisomers and self-condensation byproducts, drastically reducing isolated yields and complicating downstream purification. Furthermore, alternative literature methods involving the ring-opening of cyclopropanes, while chemically elegant, introduce substantial supply chain vulnerabilities due to the inherent instability and high energy of the three-membered ring precursors. These substrates are often difficult to store, transport, and handle on a multi-kilogram scale, posing safety risks and inflating raw material costs for procurement teams managing large-scale API intermediate manufacturing.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes readily available and thermally stable β,γ-unsaturated ketones as the primary feedstock, effectively bypassing the hazards associated with cyclopropane chemistry. As illustrated in the reaction scheme below, the process employs a catalytic amount of Ni(cod)2 and an NHC ligand to drive the isomerization, a transformation powered by the thermodynamic stability gained upon forming the conjugated enone system. This approach decouples the synthesis from the strict structural requirements of Aldol chemistry, allowing for the modular assembly of diverse substitution patterns on the alkene backbone. The ability to start from simple, commercially viable precursors like benzaldehyde derivatives and allyl bromides, as shown in the precursor synthesis pathway, ensures a reliable supply chain foundation. This strategic shift enables manufacturers to produce high-purity intermediates with fewer unit operations and reduced waste generation.
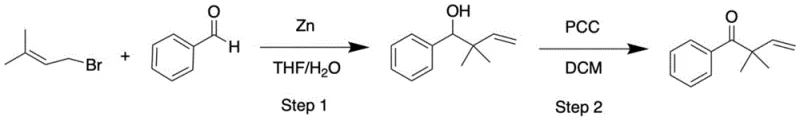
Mechanistic Insights into Ni(cod)2-Catalyzed Double Bond Isomerization
The mechanistic underpinning of this transformation involves a sophisticated interplay between the zero-valent nickel center and the olefinic substrate, mediated by the steric and electronic properties of the N-heterocyclic carbene ligand. The catalytic cycle is initiated by the coordination of the Ni(0) species to the electron-deficient double bond of the β,γ-unsaturated ketone, potentially forming a π-allyl nickel intermediate. The presence of the bulky NHC ligand, such as SIPr·HCl or IPr, is critical as it modulates the electron density at the metal center, facilitating the oxidative addition or hydrometalation steps required for C-H or C-C bond activation. The driving force for the reaction is the irreversible migration of the double bond towards the carbonyl group, establishing a conjugated system that is thermodynamically favored by approximately 10-15 kcal/mol. This isomerization is further promoted by the basic additive Cs2CO3, which may assist in proton shuttling or stabilizing charged intermediates within the catalytic manifold, ensuring high turnover numbers even at elevated temperatures.
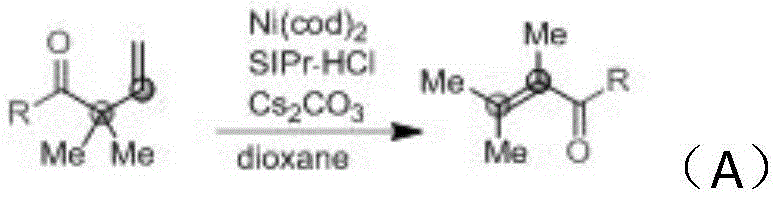
From an impurity control perspective, this mechanism offers distinct advantages over acid or base-catalyzed thermal isomerizations which often lead to polymerization or degradation. The transition metal-mediated pathway is highly specific, minimizing side reactions such as oligomerization of the terminal alkene or over-reduction of the carbonyl group. The use of non-polar aprotic solvents like dioxane or toluene further suppresses polar side reactions, resulting in a cleaner reaction profile that simplifies the final purification step. For quality assurance teams, this means the impurity profile is predictable and manageable, with the primary contaminants being residual catalyst and unreacted starting material, both of which are easily removed via standard silica gel chromatography or crystallization techniques. This level of control is essential for meeting the stringent purity specifications required for pharmaceutical intermediates destined for clinical trials.
How to Synthesize Polysubstituted Alpha,Beta-Unsaturated Ketones Efficiently
The operational simplicity of this nickel-catalyzed protocol makes it an attractive candidate for technology transfer from laboratory benchtop to pilot plant reactors. The procedure eliminates the need for cryogenic conditions or sensitive organometallic reagents like Grignards, relying instead on robust heating protocols in sealed vessels. The reaction tolerates a wide range of functional groups, allowing for late-stage functionalization strategies in complex molecule synthesis. While the specific stoichiometry and conditions can be tuned based on the substrate's electronic nature, the general workflow remains consistent, providing a standardized platform for process development teams. For a detailed breakdown of the standardized operating procedures, including precise reagent grades and safety protocols, please refer to the technical guide below.
- Prepare the catalytic mixture by combining Ni(cod)2, an N-heterocyclic carbene ligand (e.g., SIPr·HCl), and Cs2CO3 in a non-polar aprotic solvent like dioxane.
- Add the beta,gamma-unsaturated ketone substrate to the dried reaction vessel and heat the mixture to between 120°C and 180°C for 10 to 40 hours under inert atmosphere.
- Upon completion, cool the reaction to room temperature, filter off the catalyst residues, concentrate the filtrate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel-catalyzed isomerization technology translates into tangible strategic advantages regarding cost structure and vendor reliability. The substitution of precious metal catalysts like Palladium or Rhodium with Nickel represents a fundamental shift in raw material economics, as nickel salts are orders of magnitude cheaper and less subject to geopolitical supply volatility. Furthermore, the stability of the β,γ-unsaturated ketone starting materials allows for bulk purchasing and long-term storage without the degradation risks associated with reactive cyclopropanes or enolizable aldehydes. This stability significantly reduces lead times for high-purity pharmaceutical intermediates, as suppliers can maintain safety stock of key precursors without fear of spontaneous decomposition, ensuring continuity of supply even during market fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive noble metals and the simplification of the synthetic route. By avoiding multi-step sequences involving unstable intermediates, the overall cost of goods sold (COGS) is drastically simplified. The use of common industrial solvents like toluene and dioxane, combined with a straightforward filtration workup, removes the need for specialized equipment or complex aqueous waste treatment streams associated with strong acids or bases. This streamlined process flow directly contributes to substantial cost savings in API intermediate manufacturing, allowing for more competitive pricing models in contract manufacturing agreements.
- Enhanced Supply Chain Reliability: The reliance on stable, commodity-grade chemicals enhances the resilience of the supply chain against disruptions. Unlike methods requiring custom-synthesized, high-energy building blocks, this approach utilizes substrates that are either commercially available or easily prepared from bulk chemicals like benzaldehyde. This accessibility reduces the dependency on single-source vendors for exotic reagents, mitigating the risk of production stoppages. Additionally, the robustness of the reaction conditions (120-180°C) ensures consistent batch-to-batch reproducibility, a critical factor for maintaining qualified supplier status with major pharmaceutical clients who demand rigorous consistency.
- Scalability and Environmental Compliance: Scaling this reaction to commercial volumes is facilitated by the absence of exothermic hazards typically associated with organometallic additions. The reaction operates under thermal control in standard stainless steel reactors, and the catalyst loading is low enough to minimize heavy metal contamination in the final product. From an environmental standpoint, the atom economy is favorable as the reaction is an isomerization with no loss of molecular fragments, and the solvent systems are amenable to recycling. This alignment with green chemistry principles supports corporate sustainability goals and simplifies regulatory compliance regarding waste disposal and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed technology in industrial settings. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a factual basis for feasibility assessments. Understanding these nuances is crucial for project managers evaluating the integration of this chemistry into existing production pipelines.
Q: What are the primary advantages of this Nickel-catalyzed method over traditional Aldol condensation?
A: Unlike traditional Aldol condensations which often require identical ketones or lack selectivity leading to mixed products, this Ni-catalyzed isomerization offers high regioselectivity. It utilizes stable beta,gamma-unsaturated ketones as substrates rather than unstable cyclopropanes, significantly simplifying the supply chain and reducing safety risks associated with high-energy intermediates.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly scalable. It employs inexpensive base metal catalysts (Nickel) instead of precious metals, uses common industrial solvents like toluene or dioxane, and operates at robust temperatures (120-180°C). The workup involves simple filtration and chromatography, avoiding complex aqueous quenches typical of organometallic processes.
Q: What is the substrate scope for this isomerization reaction?
A: The method demonstrates broad substrate tolerance, accommodating various functional groups including halides, ethers, and nitriles on the aromatic ring. It is effective for synthesizing both acyclic and cyclic polysubstituted alpha,beta-unsaturated ketones, making it versatile for diverse pharmaceutical and material science applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Alpha,Beta-Unsaturated Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic methodologies play in accelerating drug discovery and development timelines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale optimization to tonnage manufacturing is seamless and risk-mitigated. We are committed to delivering high-purity organic intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our facility is designed to handle complex organometallic chemistry safely, with dedicated containment systems for nickel processing and solvent recovery units that maximize efficiency and minimize environmental impact.
We invite you to collaborate with us to leverage this cutting-edge isomerization technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce your overall development costs.
