Advanced Synthesis of Asymmetric Fluorinated Diamine Monomers for High-Performance Polyimides
Advanced Synthesis of Asymmetric Fluorinated Diamine Monomers for High-Performance Polyimides
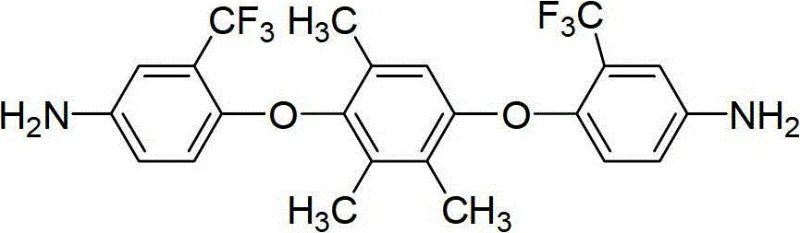
The development of advanced polyimide materials for next-generation electronics hinges critically on the molecular architecture of the constituent monomers. Patent CN102796015A introduces a breakthrough in this domain by disclosing a robust synthetic route for 1,4-bis(4-amino-2-trifluoromethylphenoxy)-2,3,5-trimethylbenzene, an asymmetric aromatic diamine monomer designed to overcome the inherent limitations of traditional polyimides. Conventional polyimides often suffer from poor solubility and deep coloration due to rigid chain stacking and strong charge-transfer complexes, restricting their application in optoelectronics. This patent addresses these challenges by integrating bulky trifluoromethyl groups and an asymmetric trimethylbenzene core, which effectively disrupts molecular packing. For R&D directors and procurement specialists seeking a reliable electronic chemical supplier, understanding the nuances of this synthesis is vital for securing high-purity polyimide monomer supplies that enable superior film-forming properties and optical transparency in final products.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional aromatic diamine monomers used in polyimide synthesis typically possess symmetrical structures lacking substantial steric bulk, which leads to tightly packed polymer chains with strong intermolecular forces. This structural rigidity results in polyimides that are insoluble in most organic solvents, often requiring aggressive solvents like concentrated sulfuric acid for processing, which is environmentally hazardous and operationally difficult. Furthermore, the extensive conjugation in these symmetric systems facilitates intramolecular charge transfer, causing the resulting films to exhibit deep yellow or brown colors and poor optical transparency, rendering them unsuitable for display applications. The oxidative instability of many conventional diamines at room temperature also poses significant storage and handling challenges, complicating the supply chain for high-purity OLED material precursors. These cumulative drawbacks necessitate a fundamental redesign of the monomer structure to achieve the desired balance of thermal stability and processability.
The Novel Approach
The innovative strategy outlined in the patent leverages a dual-modification approach, incorporating both electron-withdrawing trifluoromethyl groups and an asymmetric methyl-substituted central ring to fundamentally alter the polymer's physical properties. By synthesizing 1,4-bis(4-amino-2-trifluoromethylphenoxy)-2,3,5-trimethylbenzene, the method introduces significant steric hindrance that prevents close packing of the polymer chains, thereby drastically improving solubility in common solvents like NMP and chloroform. The presence of fluorine atoms also lowers the dielectric constant and reduces color formation by inhibiting charge-transfer complexation, directly addressing the optical clarity issues plaguing earlier generations of materials. This novel approach not only enhances the functional performance of the resulting polyimides but also simplifies the downstream processing, offering a compelling value proposition for cost reduction in electronic chemical manufacturing through improved yield and easier purification workflows.
Mechanistic Insights into Nucleophilic Substitution and Catalytic Reduction
The synthesis mechanism begins with a nucleophilic aromatic substitution where 2,3,5-trimethylhydroquinone reacts with 2-chloro-5-nitrotrifluorotoluene under alkaline conditions, typically utilizing potassium carbonate in polar aprotic solvents like DMF. The electron-withdrawing nature of the nitro and trifluoromethyl groups on the chlorobenzene ring activates the position ortho to the chlorine, facilitating the attack by the phenoxide anions generated in situ. This step is critical for establishing the ether linkages that provide flexibility to the polymer backbone, while the specific reaction temperatures of 100-150°C ensure complete conversion without degrading the sensitive nitro functionalities. Following this, the intermediate dinitro compound undergoes a catalytic reduction using hydrazine hydrate and palladium on carbon, a method chosen for its selectivity and mild conditions compared to harsh metal-acid reductions. This two-stage mechanism ensures high fidelity in constructing the complex asymmetric architecture required for advanced material performance.
Controlling impurities during this synthesis is paramount, particularly given the sensitivity of the amino groups to oxidation and the potential for incomplete reduction of the nitro intermediates. The patent specifies rigorous purification protocols, including hot water washing to remove inorganic salts and recrystallization from specific solvent mixtures like DMF and methanol, which selectively precipitate the desired product while leaving impurities in solution. The use of hydrazine hydrate as a reducing agent, rather than hydrogen gas, minimizes the risk of over-reduction or side reactions that could compromise the integrity of the trifluoromethyl groups. Furthermore, the final recrystallization from ethanol ensures that the resulting white powdery solid meets stringent purity specifications, with melting points sharply defined between 233-234°C, indicating a highly crystalline and pure material suitable for commercial scale-up of complex fluorinated monomers.
How to Synthesize 1,4-bis(4-amino-2-trifluoromethylphenoxy)-2,3,5-trimethylbenzene Efficiently
Executing this synthesis requires precise control over reaction stoichiometry and temperature profiles to maximize yield and minimize byproduct formation. The process initiates with the careful mixing of 2,3,5-trimethylhydroquinone and 2-chloro-5-nitrotrifluorotoluene in a molar ratio of approximately 1:2.0 to 1:2.3 under a nitrogen atmosphere to prevent oxidative degradation. Detailed standardized synthesis steps are provided below to guide process engineers in replicating this high-efficiency route.
- Conduct nucleophilic substitution of 2,3,5-trimethylhydroquinone with 2-chloro-5-nitrotrifluorotoluene using potassium carbonate in DMF at 100-150°C.
- Purify the resulting dinitro intermediate via hot water washing and recrystallization in a DMF/methanol mixture.
- Reduce the dinitro compound using hydrazine hydrate and a palladium carbon catalyst in ethanol at 60-100°C to yield the final diamine.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial advantages by utilizing readily available starting materials and avoiding the need for exotic or prohibitively expensive catalysts. The reliance on standard base-mediated coupling and catalytic hydrogenation means that the process can be easily integrated into existing fine chemical manufacturing facilities without requiring massive capital expenditure on specialized high-pressure reactors. This accessibility translates directly into a more resilient supply chain, as the raw materials such as chloronitrotrifluorotoluene and trimethylhydroquinone are commodity chemicals with stable market availability. For supply chain heads focused on reducing lead time for high-purity polyimide monomers, the simplicity of the workup procedures—primarily filtration and recrystallization—allows for faster batch turnover and quicker response to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of complex multi-step protection and deprotection sequences, which are common in the synthesis of functionalized aromatics, significantly streamlines the production workflow. By achieving high yields in both the substitution and reduction steps through optimized conditions, the overall material throughput is maximized, leading to substantial cost savings per kilogram of finished monomer. Additionally, the use of hydrazine hydrate as a reducing agent avoids the safety and infrastructure costs associated with high-pressure hydrogenation units, further lowering the barrier to entry for production and enhancing the economic viability of the final polyimide products.
- Enhanced Supply Chain Reliability: The stability of the final diamine product at room temperature is a critical logistical advantage, as it eliminates the need for cold chain storage or inert atmosphere shipping that unstable intermediates often require. This robustness ensures that inventory can be held safely for extended periods, buffering against supply disruptions and allowing for strategic stockpiling. The straightforward purification via recrystallization also means that quality consistency is easier to maintain across different batches, reducing the risk of supply rejection due to specification deviations and fostering stronger trust between suppliers and downstream polymer manufacturers.
- Scalability and Environmental Compliance: The process operates at moderate temperatures and pressures, making it inherently safer and easier to scale from pilot plant to full commercial production volumes. The solvent systems employed, primarily alcohols and amides, are well-understood in terms of waste management and recycling, facilitating compliance with increasingly stringent environmental regulations. The ability to recover and reuse solvents like ethanol and DMF further contributes to a greener manufacturing footprint, aligning with the sustainability goals of modern electronics companies and reducing the overall environmental impact of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized diamine monomer. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for industry stakeholders.
Q: How does the asymmetric structure improve polyimide solubility?
A: The introduction of bulky trifluoromethyl groups and the asymmetric 2,3,5-trimethylbenzene core disrupts the regular packing of polymer chains. This increases free volume and reduces intermolecular charge transfer interactions, significantly enhancing solubility in common organic solvents compared to traditional symmetric diamines.
Q: What are the purity specifications for this monomer?
A: The patented process utilizes rigorous recrystallization steps, specifically using DMF/methanol for the intermediate and ethanol for the final product. This ensures high purity suitable for electronic-grade applications, minimizing impurities that could affect the optical transparency and dielectric properties of the final polyimide film.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process relies on standard unit operations such as stirred tank reactions, filtration, and recrystallization. The use of commercially available reagents like hydrazine hydrate and palladium carbon, along with moderate temperature ranges (60-150°C), facilitates straightforward commercial scale-up without requiring exotic high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-bis(4-amino-2-trifluoromethylphenoxy)-2,3,5-trimethylbenzene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance monomers play in the advancement of electronic materials and polyimide technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global supply chains. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest industry standards. Our capability to handle complex fluorinated chemistries positions us as a strategic partner for companies looking to innovate in the fields of flexible displays and high-temperature coatings.
We invite you to collaborate with us to optimize your material sourcing strategy and accelerate your product development timelines. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term growth and stability in the competitive electronics market.
