Advanced One-Pot Synthesis of 2-Aryl-2,2-Dimethyl Methyl Acetate for Commercial Scale-Up
The landscape of fine chemical manufacturing is constantly evolving, driven by the urgent need for more efficient and sustainable synthetic pathways. A significant breakthrough in this domain is documented in patent CN102675092A, which details a novel method for preparing 2-aryl-2,2-dimethyl methyl acetate. This compound serves as a critical building block in the synthesis of various active pharmaceutical ingredients and agrochemical agents. The traditional approaches to synthesizing this sterically hindered ester often involve cumbersome multi-step procedures that suffer from low atom economy and harsh reaction conditions. By contrast, the methodology outlined in this intellectual property introduces a streamlined one-pot strategy that leverages sodium hydride and dimethyl sulfate to achieve simultaneous esterification and alpha-methylation. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this route is essential for optimizing supply chain resilience and reducing overall production costs in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical precedents for synthesizing 2-aryl-2,2-dimethyl acetates, such as those reported by the Arthur Gomtsyan group, typically rely on a disjointed two-step sequence that introduces significant operational inefficiencies. In the conventional pathway, the carboxylic acid substrate must first undergo esterification using reagents like acetyl chloride in methanol, requiring a reflux step followed by extraction and concentration to isolate the intermediate methyl ester. This isolated intermediate is then subjected to a separate alkylation step using strong bases like sodium hydride and alkylating agents such as methyl iodide in solvents like tetrahydrofuran. This fragmentation of the process not only doubles the handling time and labor requirements but also exposes the intermediate to potential degradation or contamination during isolation. Furthermore, the reliance on acetyl chloride generates stoichiometric amounts of hydrochloric acid gas, necessitating specialized scrubbing equipment and posing safety risks that complicate commercial scale-up of complex organic intermediates.

The Novel Approach
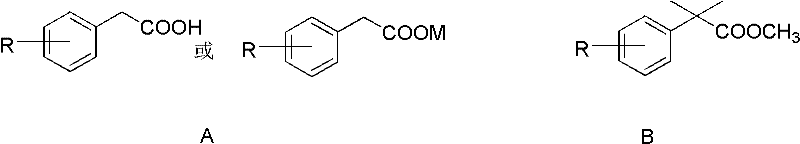
The innovative protocol described in the patent data fundamentally reengineers this workflow by consolidating the esterification and methylation events into a single reaction vessel. By utilizing 2-arylphenylacetic acid or its metal salts directly with sodium hydride in a non-polar solvent, the system generates the enolate species in situ. The subsequent addition of dimethyl sulfate acts dually as both the methylating agent for the alpha-position and the source of the methyl ester group, effectively bypassing the need for a separate esterification reagent. This convergence of steps eliminates the intermediate purification operation entirely, which is a major bottleneck in traditional processing. The result is a drastic simplification of the technique, where the process of methyl esterification and methylation is completed in the same reaction kettle, leading to products with high purity and significantly improved yields without the burden of isolating unstable intermediates.
Mechanistic Insights into NaH-Catalyzed One-Pot Esterification and Methylation
At the core of this chemical transformation lies a sophisticated interplay between strong base deprotonation and nucleophilic substitution mechanisms. The reaction initiates with the addition of sodium hydride to the solution of 2-arylphenylacetic acid, where the hydride ion abstracts the acidic proton from the carboxylic acid group to form a carboxylate salt and hydrogen gas. As the reaction progresses and dimethyl sulfate is introduced, the system facilitates a nucleophilic attack on the sulfur center, transferring a methyl group to the carboxylate oxygen to form the methyl ester functionality. Simultaneously, the strong basic environment promotes the deprotonation of the alpha-carbon adjacent to the newly formed ester, generating a reactive enolate intermediate. This enolate then undergoes a second methylation event, either from excess dimethyl sulfate or subsequently added methyl iodide, to install the gem-dimethyl group. This cascade ensures that the sterically demanding quaternary carbon center is constructed efficiently under controlled thermal conditions.
Controlling the impurity profile in such a reactive system is paramount for meeting the stringent specifications required for high-purity API intermediate production. The use of non-polar solvents like tetrahydrofuran or 1,4-dioxane helps to moderate the reactivity of the sodium hydride, preventing excessive side reactions such as over-alkylation or solvent decomposition. By maintaining the temperature within a specific range, typically initiating at low temperatures like 10°C and gradually warming to reflux, the kinetic profile is managed to favor the desired double methylation over competing hydrolysis or elimination pathways. The absence of water during the critical reaction phase is ensured by the use of anhydrous solvents and nitrogen protection, which prevents the quenching of the reactive enolate species. This meticulous control over the reaction environment allows for the direct attainment of high-purity products, often exceeding 90% purity by GC analysis before final distillation, thereby reducing the load on downstream purification units.
How to Synthesize 2-Aryl-2,2-Dimethyl Methyl Acetate Efficiently
Implementing this synthesis route requires precise adherence to the order of addition and thermal management to ensure safety and reproducibility. The process begins by charging an anhydrous non-polar solvent into a reactor under inert atmosphere, followed by the careful addition of sodium hydride to establish the basic medium. The 2-arylphenylacetic acid substrate is then introduced, allowing the formation of the carboxylate salt before the methylating agent is added. Dimethyl sulfate is dosed slowly to manage the exotherm, followed by a period of reflux to drive the esterification to completion. Once the initial methylation is exhausted, methyl iodide may be added to ensure full conversion to the gem-dimethyl species. The detailed standardized synthesis steps, including specific molar ratios and quenching procedures, are provided in the guide below for technical teams to follow.
- Prepare the reaction vessel with anhydrous non-polar solvent and add sodium hydride under nitrogen protection at controlled low temperatures.
- Introduce the 2-arylphenylacetic acid substrate followed by dimethyl sulfate to initiate simultaneous esterification and methylation.
- Heat the mixture to reflux, then quench, extract with toluene, and purify via distillation to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this one-pot methodology offers profound benefits that extend beyond mere chemical yield improvements. For procurement managers focused on cost reduction in fine chemical manufacturing, the elimination of the intermediate isolation step translates directly into reduced solvent consumption, lower energy usage for evaporation, and decreased labor hours per batch. The ability to perform both esterification and methylation in a single vessel reduces the equipment footprint required for production, allowing existing reactor trains to handle higher throughput without capital expansion. Furthermore, the avoidance of hazardous reagents like acetyl chloride simplifies regulatory compliance and waste disposal logistics, contributing to substantial cost savings in environmental management. These factors combine to create a more robust and economically viable supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The consolidation of two distinct chemical transformations into a single operational unit significantly lowers the variable costs associated with production. By removing the need to isolate and dry the intermediate methyl ester, the process saves considerable amounts of drying agents, filtration media, and solvent volumes that would otherwise be lost during transfer. Additionally, the replacement of expensive alkylating agents in some conventional routes with the more economical dimethyl sulfate for the esterification step optimizes the raw material cost structure. This qualitative improvement in process efficiency ensures that the final cost of goods sold is minimized, providing a competitive edge in the global market for specialty chemicals.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route inherently reduces the number of potential failure points within the manufacturing process. Fewer unit operations mean fewer opportunities for equipment malfunction, human error, or quality deviations that could lead to batch rejection. The use of readily available starting materials like 2-arylphenylacetic acid and common solvents ensures that raw material sourcing remains stable even during market fluctuations. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream drug manufacturers receive their key inputs on schedule without unexpected delays caused by complex synthesis bottlenecks.
- Scalability and Environmental Compliance: The reaction conditions described, which avoid extreme temperatures and high-pressure requirements, are ideally suited for scaling from pilot plant to commercial tonnage. The use of standard solvents like THF and toluene facilitates easy solvent recovery and recycling, aligning with green chemistry principles and reducing the environmental footprint of the operation. Moreover, the reduction in waste generation, particularly the avoidance of acidic byproducts from acetyl chloride, simplifies effluent treatment processes. This scalability and environmental compatibility make the technology a sustainable choice for long-term production partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating the feasibility of this route. Understanding these nuances is vital for making informed decisions about process adoption and supplier qualification.
Q: What are the primary advantages of this one-pot method over conventional routes?
A: This method eliminates the need for intermediate purification and avoids hazardous reagents like acetyl chloride, significantly simplifying the workflow and reducing waste generation.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the use of common solvents like THF and manageable temperature profiles makes this protocol highly suitable for commercial scale-up in standard reactor systems.
Q: How does the impurity profile compare to traditional multi-step syntheses?
A: By avoiding isolation of unstable intermediates, the one-pot approach minimizes side reactions, resulting in a cleaner crude product with higher GC purity prior to final distillation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aryl-2,2-Dimethyl Methyl Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthesis pathways play in the success of modern drug development programs. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory methods like the one described in CN102675092A can be successfully translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify identity and assay. Our infrastructure is designed to handle complex organic intermediates with the utmost care, guaranteeing consistency and quality in every batch we deliver to our global partners.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this optimized route can impact your bottom line. Please contact us to request specific COA data and route feasibility assessments for 2-aryl-2,2-dimethyl methyl acetate. By partnering with us, you gain access to a supply chain that values innovation, quality, and reliability, ensuring your projects proceed without interruption.
