Scalable Photocatalytic Synthesis of Chiral Azaarene Cyclobutanes for Advanced Drug Discovery
Scalable Photocatalytic Synthesis of Chiral Azaarene Cyclobutanes for Advanced Drug Discovery
The pharmaceutical industry continuously seeks robust methodologies for constructing complex chiral scaffolds, particularly those containing strained ring systems like cyclobutanes which are prevalent in bioactive molecules. Patent CN115894345A introduces a groundbreaking visible-light-driven protocol for the asymmetric synthesis of chiral azaarene-functionalized cyclobutanes. This technology leverages a dual catalytic system comprising an organic photosensitizer (DPZ) and a chiral phosphoric acid (CPA) to facilitate a highly stereoselective [2+2] cycloaddition. Unlike traditional thermal methods that often require harsh UV irradiation or stoichiometric chiral auxiliaries, this photo-redox approach operates under mild conditions with exceptional control over stereochemistry. The method enables the direct functionalization of nitrogen-containing heterocycles, generating valuable intermediates with all-carbon quaternary stereocenters or four adjacent stereocenters in high yields.
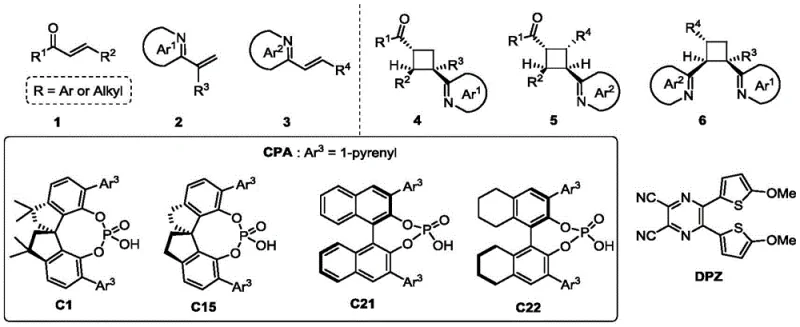
For R&D directors focusing on pipeline acceleration, this patent offers a versatile route to access diverse chemical space. The substrate scope is remarkably broad, accommodating various substituted enones and vinyl azaarenes, including pyridines, quinolines, and benzimidazoles. The ability to tune the electronic properties of the azaarene moiety without compromising reactivity is a significant advantage for medicinal chemists optimizing structure-activity relationships (SAR). Furthermore, the metal-free nature of the catalyst system addresses critical purity concerns, ensuring that the final active pharmaceutical ingredients (APIs) are free from toxic transition metal residues, thereby streamlining the regulatory approval process for new drug candidates targeting complex biological pathways.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of cyclobutane rings bearing nitrogen heterocycles has been fraught with synthetic challenges. Traditional thermal [2+2] cycloadditions often necessitate high-energy UV light, which can lead to substrate decomposition, polymerization, and poor functional group tolerance. Moreover, achieving high enantioselectivity in these reactions typically requires expensive chiral Lewis acids based on rare earth metals or transition metals like ruthenium and iridium. These metal catalysts not only increase the raw material cost but also introduce significant downstream processing burdens, such as the need for rigorous metal scavenging steps to meet strict ppm limits set by health authorities. Additionally, ground-state ionic pathways for activating electron-deficient azaarenes are limited due to their weak electron-withdrawing properties, often resulting in low conversion rates and sluggish reaction kinetics that hinder commercial viability.
The Novel Approach
The methodology disclosed in CN115894345A represents a paradigm shift by utilizing visible light energy to overcome activation barriers through an energy transfer mechanism. By employing DPZ, a dicyanopyrazine-derived organic dye, the system efficiently generates reactive diradical intermediates from enone substrates under benign blue LED irradiation. This avoids the damaging effects of UV light and allows for the use of simple, inexpensive glassware. Crucially, the integration of a chiral phosphoric acid catalyst creates a supramolecular assembly via hydrogen bonding, which precisely orients the vinyl azaarene for the cycloaddition event. This concerted catalysis strategy bypasses the need for heavy metals entirely, offering a greener, more sustainable alternative that aligns with modern principles of green chemistry while delivering superior stereocontrol compared to classical thermal methods.
Mechanistic Insights into DPZ-Mediated Energy Transfer and H-Bond Catalysis
The success of this transformation relies on the intricate interplay between photo-excitation and chiral induction. Upon irradiation with visible light (450-455 nm), the DPZ photocatalyst absorbs photons to reach an excited triplet state. Through a Dexter energy transfer process, this excited state transfers energy to the enone substrate (compound 1), promoting it to a reactive triplet diradical species. This diradical is highly electrophilic and primed for cyclization. Simultaneously, the chiral phosphoric acid (CPA) engages the nitrogen atom of the vinyl azaarene (compound 2) or (E)-alkenyl azaarene (compound 3) through a specific hydrogen-bonding interaction. This activation lowers the LUMO energy of the azaarene and, more importantly, creates a rigid chiral pocket around the reaction site.
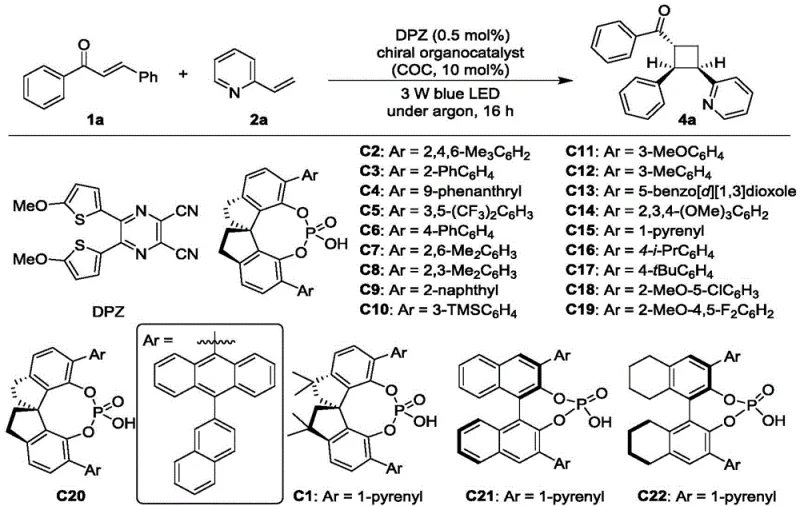
As the diradical approaches the activated azaarene, the steric bulk of the CPA's aryl substituents (such as the 1-pyrenyl group in C1 or the binaphthyl backbone in C15) dictates the facial selectivity of the bond formation. This ensures that the new carbon-carbon bonds are formed with high diastereoselectivity (dr) and enantiomeric excess (ee). The mechanism effectively decouples the radical generation step from the stereo-determining step, allowing for independent optimization of each component. For procurement managers, understanding this mechanism highlights the robustness of the supply chain; since the catalysts are small organic molecules rather than complex metal complexes, they are easier to synthesize and source reliably, reducing the risk of supply disruptions associated with geopolitical constraints on rare metal mining.
How to Synthesize Chiral Azaarene-Functionalized Cyclobutanes Efficiently
Implementing this photocatalytic protocol in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and optical purity. The process begins with the preparation of the reaction vessel under an inert atmosphere to prevent quenching of the excited states by oxygen. The precise molar ratios of the substrates and catalysts are critical; typically, a slight excess of the enone (1:1.5 ratio relative to the azaarene) drives the reaction to completion. The choice of solvent, specifically dry chloroform, plays a vital role in solubilizing the organic catalysts and maintaining the integrity of the hydrogen-bonding network. Detailed standardized operating procedures for scaling this reaction from milligram to kilogram quantities are outlined below to ensure reproducibility and safety.
- Dissolve the organic photocatalyst DPZ in toluene, transfer an aliquot to a Schlenk tube, and remove the solvent under vacuum to form a thin film.
- Add the enone substrate (e.g., chalcone), vinyl azaarene, and chiral phosphoric acid catalyst (e.g., C1 or C15) to the tube, followed by dry chloroform solvent.
- Subject the mixture to freeze-pump-thaw cycles for argon protection, then irradiate with 3W blue LEDs at -35°C for 36 hours before purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this metal-free photocatalytic technology offers tangible strategic benefits beyond mere chemical novelty. The elimination of transition metals from the catalytic cycle fundamentally alters the cost structure of manufacturing these complex intermediates. By removing the need for expensive palladium, iridium, or rhodium catalysts, the direct material costs are significantly reduced. Furthermore, the absence of heavy metals simplifies the purification workflow, eliminating the need for specialized resin columns or extensive washing protocols required to reduce metal content to acceptable levels. This streamlining of the downstream process translates directly into shorter production cycles and lower operational expenditures, enhancing the overall margin profile for high-value pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The utilization of organic photocatalysts like DPZ, which are synthetically accessible and inexpensive compared to noble metal complexes, drastically lowers the catalyst loading cost per kilogram of product. Additionally, the mild reaction temperatures (-35°C to -20°C) reduce the energy consumption associated with cryogenic cooling compared to processes requiring extreme低温 or high heat. The high atom economy of the [2+2] cycloaddition minimizes waste generation, further contributing to cost efficiency by reducing waste disposal fees and maximizing the yield of valuable chiral building blocks from raw starting materials.
- Enhanced Supply Chain Reliability: Relying on organic small molecules for catalysis mitigates the supply risks associated with the volatile market prices of precious metals. The key reagents, including the chiral phosphoric acids and the DPZ dye, can be sourced from multiple chemical suppliers or manufactured in-house with standard organic synthesis equipment. This diversification of the supply base ensures business continuity and protects against shortages that frequently plague the fine chemical industry. The stability of the catalysts also allows for longer shelf life and easier logistics, facilitating just-in-time manufacturing strategies for global pharmaceutical clients.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable, as visible light penetration can be managed through the use of flow photoreactors or optimized batch vessels with high surface-area-to-volume ratios. The process generates minimal hazardous waste, aligning with increasingly stringent environmental regulations regarding solvent use and heavy metal discharge. The use of chloroform, while requiring careful handling, is a well-understood solvent in industrial settings with established recovery and recycling protocols. This environmental compatibility simplifies the permitting process for new manufacturing lines and supports corporate sustainability goals by reducing the overall carbon footprint of the synthesis.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this novel synthetic route for industrial applications.
Q: What are the advantages of using DPZ over transition metal photocatalysts?
A: DPZ is a metal-free organic chromophore that eliminates the risk of heavy metal contamination in pharmaceutical intermediates, simplifying downstream purification and regulatory compliance while maintaining high catalytic efficiency under visible light.
Q: How does the chiral phosphoric acid influence stereoselectivity?
A: The chiral phosphoric acid (CPA) acts as a hydrogen-bond donor, activating the imine group of the azaarene and creating a chiral environment that directs the approach of the diradical intermediate, resulting in high enantiomeric excess (ee) and diastereoselectivity (dr).
Q: Can this process be scaled for industrial production?
A: Yes, the reaction utilizes mild conditions (-35°C to -20°C) and standard blue LED irradiation, which are amenable to flow chemistry setups and large-scale batch reactors, ensuring consistent quality and supply continuity for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Cyclobutanes Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the photocatalytic synthesis method described in CN115894345A for producing high-value chiral intermediates. As a leading CDMO partner, we possess the technical expertise to translate this academic innovation into robust, GMP-compliant manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We operate stringent purity specifications and utilize rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee that every batch of chiral azaarene-functionalized cyclobutanes meets the highest quality standards required for drug development.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable pharmaceutical intermediate supplier committed to driving innovation and efficiency in your supply chain.
