Revolutionizing Exatecan Intermediate Production via Efficient Negishi Coupling Technology
Introduction to Advanced Intermediate Synthesis
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing complex oncology agents, particularly topoisomerase I inhibitors like Exatecan (DX-8951). A pivotal development in this domain is disclosed in patent CN112300019A, which outlines a novel preparation method for o-acetamidophenylbutyric acid compounds. These compounds serve as critical building blocks in the total synthesis of Exatecan, a potent antitumor agent used for treating advanced soft tissue sarcoma and various carcinomas. The traditional manufacturing landscape for these intermediates has been plagued by lengthy synthetic sequences and hazardous reagents, creating bottlenecks for reliable pharmaceutical intermediate supplier networks. This new technology addresses these challenges by introducing a concise, high-yielding route that leverages modern cross-coupling chemistry to streamline production.
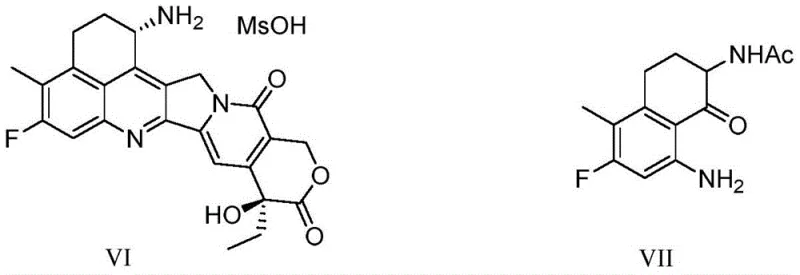
The strategic importance of this innovation cannot be overstated for global supply chains dependent on camptothecin derivatives. By optimizing the construction of the phenylbutyric acid scaffold, manufacturers can significantly reduce the environmental footprint and operational costs associated with API manufacturing. The patent details a method that not only improves chemical efficiency but also enhances the safety profile of the production process, making it an attractive option for large-scale industrial adoption. As we delve deeper into the technical specifics, it becomes clear that this approach represents a substantial leap forward in process chemistry for oncology drug precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing compound VII, the key intermediate for Exatecan, have relied on cumbersome and dangerous chemical transformations. For instance, earlier methodologies described in US005658920A utilized a sequence involving Friedel-Crafts acylation followed by carbonyl reduction and nitration. These routes often necessitated the use of polyphosphoric acid for cyclization steps, which poses severe safety hazards due to the requirement for high-temperature reactions and corrosive handling. Furthermore, the overall yield of such processes was notoriously low, with specific cyclization steps reporting yields as poor as 25%, leading to massive material loss and increased waste generation.
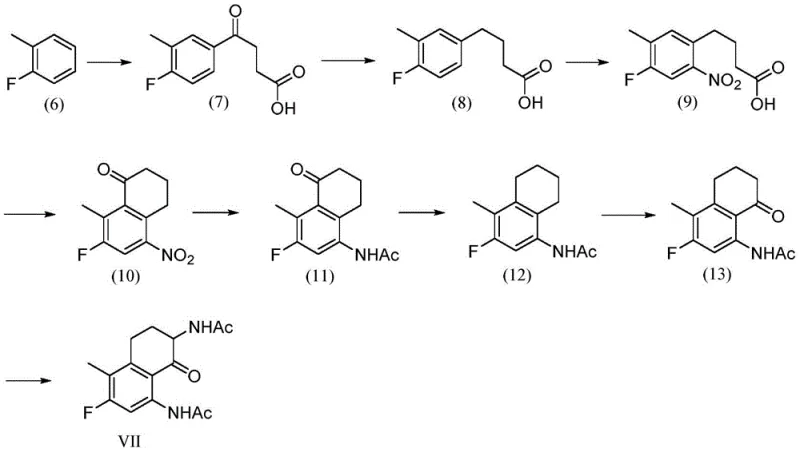
Another significant drawback of conventional methods, such as the 13-step route disclosed in WO96/26181, is the sheer complexity of the operation. While the individual reaction types might be simple, the cumulative effect of thirteen distinct steps results in a total yield of only roughly 17%. This attrition rate is economically unsustainable for commercial production. Additionally, these older routes frequently required multiple reduction steps, including alcohol formation, dehydration, and double bond reduction, which complicated purification and increased the risk of forming isomeric by-products. The reliance on low-temperature borane reductions further exacerbated safety concerns, requiring specialized equipment and rigorous hazard controls that drive up capital expenditure.
The Novel Approach
In stark contrast, the methodology presented in CN112300019A offers a radically simplified pathway that condenses the synthesis into just four primary stages. The core innovation lies in the utilization of a Negishi coupling reaction to construct the carbon-carbon bond between the aromatic ring and the butyric acid side chain. This transition metal-catalyzed approach bypasses the need for multiple Friedel-Crafts acylations and the subsequent tedious reduction sequences. By directly coupling an aryl halide with an organozinc reagent, the process achieves high regioselectivity and efficiency, effectively eliminating the formation of difficult-to-separate isomers that plagued previous nitration-heavy routes.
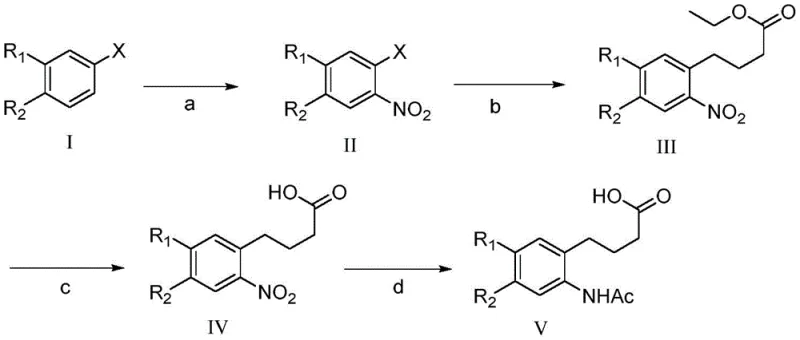
The new route begins with a controlled nitration of a substituted benzene derivative, followed immediately by the pivotal Negishi coupling. This is succeeded by a straightforward ester hydrolysis and a final tandem reduction-acetylation step. The elimination of polyphosphoric acid and borane reagents not only improves the safety profile but also simplifies the downstream processing and waste treatment protocols. The result is a robust, scalable process that delivers the target o-acetamidophenylbutyric acid compounds with superior purity and yield, positioning it as a preferred method for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Negishi Coupling and Tandem Reduction
The heart of this technological advancement is the palladium-catalyzed Negishi coupling, a powerful tool for forming C-C bonds between sp2 and sp3 hybridized carbons. In this specific application, an aryl bromide or iodide (Compound II) reacts with an alkylzinc reagent derived from ethyl 4-bromobutyrate. The mechanism involves the oxidative addition of the aryl halide to the Pd(0) catalyst, followed by transmetallation with the organozinc species. This step is crucial as it transfers the four-carbon chain to the palladium center with high fidelity. The subsequent reductive elimination releases the coupled product (Compound III) and regenerates the active catalyst. The use of specific ligands like S-PHOS or X-PHOS enhances the stability of the catalytic cycle and prevents beta-hydride elimination, a common side reaction in alkyl-alkyl couplings that leads to olefin by-products.
Following the coupling, the process employs a highly efficient tandem reduction and protection strategy. The nitro group on the aromatic ring is reduced to an amine using catalytic hydrogenation (Pd/C), a clean method that produces water as the only by-product. Without isolating the sensitive amine intermediate, acetic anhydride is introduced directly into the reaction mixture in the presence of a base like pyridine. This one-pot transformation converts the nascent amine into the stable acetamide (Compound V), preventing oxidation or polymerization of the free amine. This mechanistic elegance ensures that the final product is obtained with minimal impurities, reducing the burden on purification units and enhancing the overall throughput of the manufacturing line.
How to Synthesize O-Acetamidophenylbutyric Acid Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize the benefits of the new route. The process initiates with the nitration of the starting material at low temperatures (-10 to 0°C) to ensure mono-nitration and prevent over-reaction. The subsequent Negishi coupling demands strictly anhydrous conditions, typically using THF or DMF as solvents, to maintain the integrity of the organozinc reagent. Careful monitoring of the catalyst loading (2-10% mol) is essential to balance cost and reaction rate. Finally, the hydrolysis and reduction steps are conducted under mild conditions, utilizing aqueous base and atmospheric hydrogen pressure respectively, which facilitates easy scale-up in standard stainless steel reactors.
- Perform nitration on a substituted benzene derivative using potassium nitrate in sulfuric acid at low temperature (-10 to 0°C) to introduce the nitro group ortho to the halogen.
- Execute a Negishi coupling reaction between the nitrated aryl halide and an organic zinc reagent (derived from ethyl 4-bromobutyrate) using a palladium catalyst to install the four-carbon side chain.
- Hydrolyze the ester group of the coupled product using sodium hydroxide in methanol to obtain the free carboxylic acid, followed by catalytic hydrogenation and acetylation to yield the final o-acetamido product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic benefits beyond mere chemical curiosity. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the need for specialized and hazardous reagents like polyphosphoric acid and borane complexes, the process reduces dependency on niche chemical suppliers and mitigates the risks associated with transporting dangerous goods. This streamlining of the input material list enhances supply chain resilience, ensuring that production schedules are less likely to be disrupted by regulatory hurdles or logistics bottlenecks associated with hazardous cargo.
- Cost Reduction in Manufacturing: The economic impact of shortening the synthetic route from 13 steps to 4 steps is profound. Each eliminated step removes associated costs for solvents, energy, labor, and waste disposal. Furthermore, the high yields reported in the patent, such as 97% for the nitration step and 91% for the final acetylation, mean that less starting material is required to produce the same amount of final product. The avoidance of expensive and difficult-to-handle reagents like polyphosphoric acid also lowers the operational expenditure related to equipment maintenance and corrosion control, leading to substantial long-term savings.
- Enhanced Supply Chain Reliability: The robustness of the Negishi coupling and the subsequent one-pot reduction/acetylation creates a more predictable manufacturing timeline. Traditional routes with low-yielding cyclization steps often required re-processing or extensive chromatography, introducing variability into lead times. With this new method, the high purity of intermediates allows for direct use in subsequent steps without rigorous purification, accelerating the overall production cycle. This reliability is critical for maintaining consistent inventory levels of high-purity pharmaceutical intermediates, enabling downstream API manufacturers to plan their production with greater confidence.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is vastly superior. The removal of polyphosphoric acid eliminates a major source of acidic waste, while the switch to catalytic hydrogenation reduces the generation of stoichiometric metal waste typical of hydride reductions. The process operates at near-ambient temperatures for most steps, reducing energy consumption for heating and cooling. These factors collectively lower the environmental footprint of the manufacturing process, facilitating easier compliance with increasingly stringent global environmental regulations and reducing the costs associated with waste treatment and disposal.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new preparation method is essential for stakeholders evaluating its potential for integration into existing production lines. The following questions address common inquiries regarding the practical implementation and benefits of this technology. The answers are derived directly from the experimental data and process descriptions provided in the patent literature, ensuring accuracy and relevance for technical decision-makers.
Q: How does the new Negishi coupling route compare to traditional Friedel-Crafts acylation methods?
A: The new route significantly reduces the number of synthetic steps from over 13 in conventional methods to just 4 key steps. It eliminates the need for hazardous polyphosphoric acid cyclization and low-temperature borane reductions, thereby improving overall process safety and yield.
Q: What are the typical yields and purity levels achieved in this process?
A: The patent data indicates high efficiency, with the initial nitration step achieving yields up to 97% and purity of 98%. The final acetylation step demonstrates a yield of approximately 91%, ensuring a robust supply of high-purity intermediates suitable for downstream API synthesis.
Q: Is this method scalable for industrial production of Exatecan intermediates?
A: Yes, the process utilizes standard industrial reagents like palladium catalysts and avoids extreme conditions such as high-temperature polyphosphoric acid reactions. The simplified workup procedures and high regioselectivity of the Negishi coupling make it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable O-Acetamidophenylbutyric Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN112300019A for the production of Exatecan intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to rapidly adapt this innovative chemistry for commercial deployment. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cost-effective and environmentally friendly synthesis method. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can optimize your supply chain for next-generation oncology therapeutics.
