Advanced Sterically Hindered Iridium Complexes for High-Efficiency Red OLED Manufacturing
The landscape of organic light-emitting diode (OLED) technology is constantly evolving, with a persistent demand for materials that offer higher efficiency and stability, particularly in the red spectrum where concentration quenching has historically been a bottleneck. Patent CN113461739B introduces a groundbreaking class of red light organic electrophosphorescent materials based on tricyclic metal iridium complexes featuring phthalazine derivatives. This innovation specifically targets the limitation of existing materials that suffer from severe efficiency roll-off due to intermolecular interactions at high doping concentrations. By integrating rigid steric hindrance groups directly into the ligand structure, the invention achieves a significant reduction in non-radiative transitions while maintaining high luminous efficiency. For R&D directors and procurement specialists seeking a reliable OLED material supplier, this technology represents a pivotal shift towards more robust and commercially viable red emitters that can withstand the rigorous demands of next-generation display manufacturing.
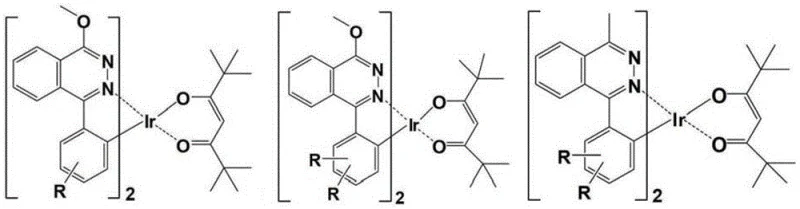
The core innovation lies in the molecular architecture depicted in the general formulas, where specific substituents are engineered to provide massive steric bulk without compromising the electronic properties necessary for phosphorescence. Unlike previous generations of bicyclic complexes or those with insufficient steric protection, these tricyclic homoleptic complexes utilize groups such as triptycene, substituted carbazoles, and bulky alkyl chains to physically isolate the emissive centers. This strategic design ensures that the electroluminescent wavelength and efficiency remain stable even as concentration and driving voltage fluctuate, solving a critical pain point in device engineering. The ability to tune these properties through the selection of R1, R2, R3, and R4 groups offers a versatile platform for customizing emission profiles, making these materials highly attractive for specialized electronic chemical manufacturing applications where precision and consistency are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional red phosphorescent materials, particularly those based on simpler phthalazine or pyridazine ligands, have struggled with the phenomenon of concentration quenching, where the proximity of emitter molecules leads to energy loss rather than light emission. Prior art often relied on bicyclic metal iridium complexes which, while functional, exhibited lower stability and luminous efficiency compared to their tricyclic counterparts. Furthermore, earlier attempts to introduce steric hindrance often involved complex multi-step syntheses like Diels-Alder reactions to attach bicycloalkyl groups, which significantly increased production costs and reduced overall yield. These conventional methods also frequently lacked comprehensive steric protection, leaving certain coordination sites vulnerable to intermolecular interactions that degrade device performance over time. Consequently, manufacturers faced challenges in achieving high brightness and long operational lifetimes, limiting the commercial viability of these materials in high-end display panels.
The Novel Approach
The methodology outlined in this patent overcomes these hurdles by employing a streamlined synthetic route that incorporates rigid, bulky groups directly onto the phthalazine core through efficient coupling reactions. Instead of relying on fragile or difficult-to-synthesize moieties, the new approach utilizes robust structures like triptycene and substituted diphenylamines that provide immense steric density in the direction of the N-Ir and C-Ir bonds. This comprehensive shielding effect drastically reduces the probability of triplet exciton self-quenching, allowing the material to maintain high quantum efficiency even in pure solid forms or at high doping levels. Additionally, the electron-donating or withdrawing nature of these substituents can be fine-tuned to balance carrier transport properties, enhancing the overall performance of the OLED device. This novel strategy not only improves the photophysical properties but also simplifies the supply chain by using readily available starting materials, offering a clear path for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Steric Hindrance and Cyclometalation
The enhanced performance of these materials is rooted in the precise manipulation of molecular geometry and electronic distribution during the synthesis and operation of the complex. The introduction of electron-withdrawing groups at the R1 position increases the acidity of the hydrogen atoms on the cyclometalated benzene ring, facilitating the deprotonation and subsequent coordination with the iridium atom to form the stable tricyclic structure. Simultaneously, the symmetrical arrangement of bulky groups on the phthalazine ring prevents the formation of isomers and provides a physical barrier that inhibits the close approach of neighboring molecules. This rigidity is crucial as it suppresses vibrational modes that typically lead to non-radiative energy dissipation, thereby preserving the excited state energy for photon emission. The result is a material with a high internal quantum efficiency that approaches theoretical limits, driven by a molecular design that prioritizes both electronic optimization and steric isolation.
Furthermore, the synthesis mechanism leverages well-established catalytic cycles to ensure high purity and reproducibility, which is essential for commercial scale-up of complex OLED materials. The initial formation of the ligand involves either a palladium-catalyzed Suzuki coupling or a nucleophilic aromatic substitution, both of which are highly selective and scalable processes. Following ligand purification, the cyclometalation step proceeds through a thermal reaction with iridium precursors in high-boiling solvents, driving the equilibrium towards the formation of the facial tris-cyclometalated isomer. This specific isomer is favored for its superior luminescent properties and stability. The entire process is designed to minimize impurities that could act as quenching sites, ensuring that the final product meets the stringent purity specifications required for high-performance optoelectronic applications.
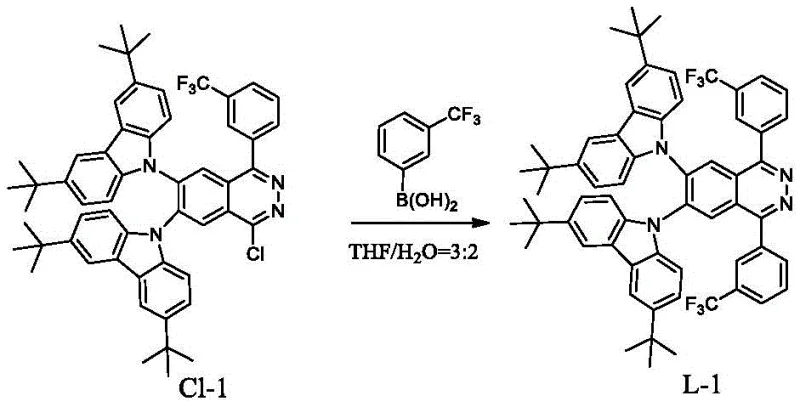
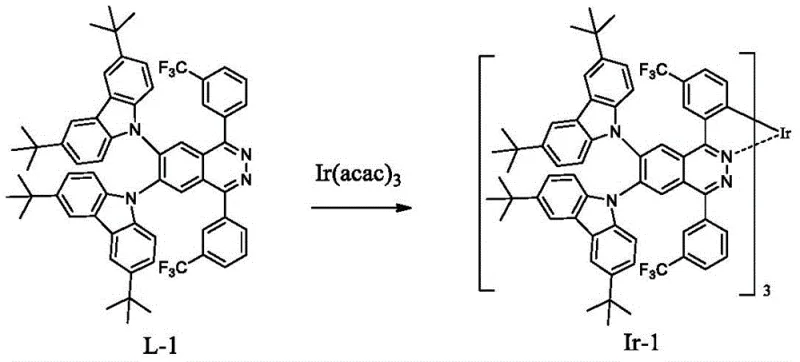
How to Synthesize Sterically Hindered Phthalazine Iridium Complexes Efficiently
The synthesis of these advanced phosphorescent materials follows a logical two-stage protocol that balances chemical complexity with operational simplicity, making it accessible for pilot and commercial production. The process begins with the construction of the functionalized phthalazine ligand, where specific steric groups are attached to the core structure using optimized reaction conditions that maximize yield and minimize byproducts. Once the ligand is isolated and purified to remove any residual catalysts or unreacted starting materials, it undergoes a cyclometalation reaction with an iridium source. Detailed standardized synthesis steps are provided in the guide below to ensure consistent quality and performance across different batches.
- Synthesize the phthalazine ligand via Suzuki coupling of 1-chlorophthalazine derivatives with substituted phenylboronic acids or via nucleophilic substitution with diphenylamines.
- Purify the resulting ligand using silica gel column chromatography and recrystallization to ensure high purity before metal coordination.
- React the purified ligand with an iridium source such as Ir(acac)3 or IrCl3 in high-boiling solvents like glycerol or ethylene glycol ethers at elevated temperatures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers significant strategic benefits beyond mere performance metrics, primarily driven by the simplification of the manufacturing workflow and the robustness of the raw material supply. The elimination of complex, multi-step synthetic routes for introducing steric groups translates directly into reduced processing time and lower consumption of expensive reagents and catalysts. By utilizing common industrial solvents and standard reaction vessels, the capital expenditure required for setting up production lines is minimized, while the reliance on exotic or hard-to-source intermediates is greatly diminished. This streamlining of the process enhances supply chain reliability, ensuring that production schedules can be met consistently without the bottlenecks often associated with specialty chemical synthesis.
- Cost Reduction in Manufacturing: The synthetic route described avoids the need for costly and low-yielding Diels-Alder reactions previously used to introduce steric bulk, replacing them with high-yielding Suzuki couplings and nucleophilic substitutions. This shift significantly lowers the cost of goods sold by reducing the number of unit operations and improving the overall material throughput. Furthermore, the use of abundant iridium sources and common organic solvents helps stabilize input costs, protecting margins against market volatility. The high purity of the final product also reduces the need for extensive downstream purification, saving both time and resources in the final stages of production.
- Enhanced Supply Chain Reliability: The starting materials, such as chlorophthalazine derivatives and substituted boronic acids, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent mixtures, allows for flexibility in sourcing and inventory management. This resilience ensures that production can continue uninterrupted even if specific reagent grades vary slightly, providing a buffer against supply chain disruptions. Additionally, the scalability of the process means that volume requirements can be ramped up quickly to meet surging demand in the display sector.
- Scalability and Environmental Compliance: The process operates under relatively mild conditions compared to other organometallic syntheses, with temperatures generally staying below 230°C and pressures remaining near atmospheric levels. This reduces the energy footprint of the manufacturing process and simplifies the engineering controls needed for safe operation. The solvents used, such as glycerol and ethylene glycol ethers, are easier to recover and recycle than many halogenated alternatives, supporting sustainability goals and regulatory compliance. The high efficiency of the reaction also minimizes waste generation, aligning with modern green chemistry principles and reducing disposal costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and performance of these novel iridium complexes in OLED applications. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating these materials into existing product lines and for assessing their long-term value proposition.
Q: How do these new complexes address concentration quenching in red OLEDs?
A: The patent introduces rigid steric hindrance groups such as triptycene and bulky carbazoles directly onto the phthalazine ligand framework. This structural modification physically separates the iridium centers, minimizing intermolecular interactions that typically lead to triplet-triplet annihilation and efficiency roll-off at high doping concentrations.
Q: What is the expected emission wavelength range for these materials?
A: According to the technical specifications in the patent, the synthesized phosphorescent materials exhibit emission wavelengths between 590 nanometers and 650 nanometers, covering the critical orange to deep-red spectrum required for full-color display applications.
Q: Are the synthesis conditions suitable for large-scale production?
A: Yes, the methodology utilizes standard industrial reactions such as Suzuki-Miyaura coupling and thermal cyclometalation. The processes operate at manageable temperatures ranging from 100°C to 230°C using common solvents like THF, glycerol, and ethylene glycol ethers, facilitating straightforward scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phthalazine Iridium Complex Supplier
As the demand for high-efficiency red emitters continues to grow, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge technology and seamless production capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our rigorous QC labs to guarantee stringent purity specifications for every batch. Our team of experts is dedicated to optimizing these complex synthetic routes to meet your specific volume and timeline requirements, ensuring that you receive materials that perform consistently in your final devices. With a focus on quality and reliability, we are committed to supporting your innovation in the competitive field of organic optoelectronics.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. By engaging with us early in your development cycle, you can gain valuable insights into route feasibility assessments and obtain specific COA data that demonstrates the superior quality of our materials. Let us help you accelerate your time-to-market with a supply partner that understands the intricacies of advanced electronic chemical manufacturing and is ready to support your growth.
