Advanced Catalytic Synthesis of Naloxone and Naltrexone for Commercial API Production
Advanced Catalytic Synthesis of Naloxone and Naltrexone for Commercial API Production
The pharmaceutical industry continuously seeks robust, scalable, and environmentally compliant synthetic routes for critical opioid antagonists like Naloxone and Naltrexone. Patent CN101955484A introduces a transformative methodology that addresses long-standing challenges in the production of these high-value active pharmaceutical ingredients (APIs). By leveraging a novel sequence starting from thebaine, this technology optimizes reaction conditions to minimize side reactions while maximizing throughput. The process is particularly notable for its strategic replacement of hazardous chlorinated solvents with greener alternatives, aligning with modern green chemistry principles without compromising yield or purity. For R&D directors and procurement specialists, this patent represents a viable pathway to secure a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials with reduced regulatory burden.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Naloxone from oxycodone or related precursors has been plagued by significant operational hazards and inefficiencies. Traditional routes, such as those reported by the Beijing Military Medical Science Institute, often rely heavily on toxic chlorinated solvents like chloroform and ethylene dichloride throughout the purification and reaction stages. These solvents pose severe health risks to synthetic staff and create substantial environmental liabilities due to their persistence and difficulty in waste treatment. Furthermore, the conventional conversion of key intermediates often suffers from difficult-to-control reaction conditions, leading to unpredictable side reactions and inconsistent yields. The total recovery rate in these legacy processes typically hovers around 40%, indicating substantial material loss and increased cost of goods sold (COGS). The reliance on such hazardous chemistry also complicates the commercial scale-up of complex opioid antagonists, as facilities require specialized containment and ventilation systems to meet safety regulations.
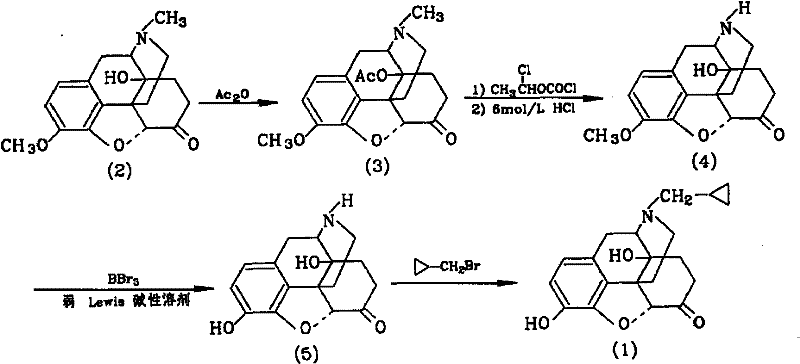
The Novel Approach
In stark contrast, the methodology outlined in CN101955484A offers a streamlined and safer alternative that fundamentally reengineers the synthetic pathway. This novel approach initiates with the direct oxidation of thebaine in formic acid, followed by a controlled hydrogenation step that utilizes inert gas protection to prevent degradation. A key innovation lies in the substitution of toxic halogenated solvents with benign organic solvents such as toluene, ethanol, and methanol during critical alkylation and purification steps. The process employs mild temperature ranges, typically between 20°C and 40°C for oxidation and 60°C to 100°C for acetylation, which enhances reaction controllability and minimizes thermal degradation of sensitive morphinan structures. By eliminating the use of chloroform and ethylene dichloride, this method drastically simplifies waste management protocols and reduces the overall environmental footprint. The result is a more stable process with obviously risen yields in individual steps, providing a solid foundation for cost reduction in API manufacturing.
Mechanistic Insights into Thebaine Oxidation and Catalytic Hydrogenation
The core of this synthetic strategy relies on a precise oxidative transformation of thebaine, followed by a selective catalytic hydrogenation. In the initial step, thebaine is dissolved in formic acid, and an oxidant such as m-chloroperbenzoic acid or hydrogen peroxide is added dropwise while maintaining a strict temperature window of 20°C to 40°C. This controlled oxidation is critical for generating the requisite epoxide or ketone functionality without over-oxidizing the sensitive aromatic rings. Following oxidation, the reaction vessel is purged with inert gas, and a metal framework catalyst, specifically Raney Nickel or Palladium on Carbon, is introduced. The system is then subjected to hydrogenation at 25°C to 45°C for 7 to 13 hours. This heterogeneous catalysis ensures high selectivity, reducing the double bond in the morphinan skeleton while preserving other functional groups. The use of solid catalysts allows for simple filtration, removing the need for complex metal scavenging resins often required with homogeneous catalysts, thereby streamlining the downstream processing workflow.
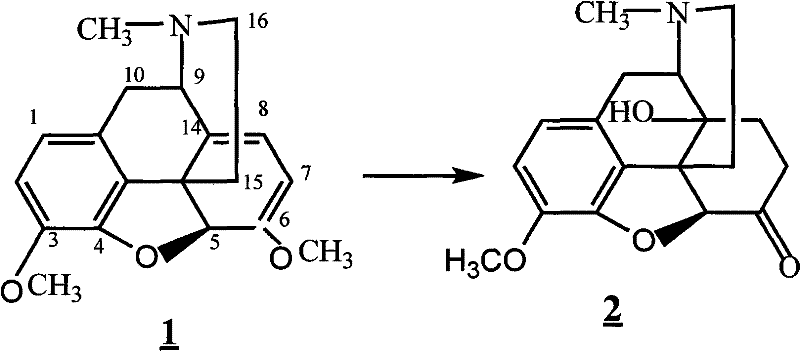
Subsequent steps involve acetylation and a unique cyclization-demethylation sequence that ensures high purity. The intermediate Compound 2 reacts with acetic anhydride at elevated temperatures (60-100°C) to protect hydroxyl groups, forming Compound 3 with yields exceeding 90%. The subsequent reaction with 1-chloroethyl chloroformate in toluene facilitates a Vilsmeier-Haack type formylation or cyclization, which is then hydrolyzed under acidic conditions to reveal the ketone functionality essential for biological activity. Finally, the demethylation step utilizes boron tribromide (BBr3) at temperatures ranging from -10°C to 40°C. This Lewis acid-mediated cleavage of the methyl ether is highly specific, avoiding the harsh conditions of traditional hydrobromic acid reflux which can lead to impurity formation. The careful control of stoichiometry and temperature in this final step is paramount for achieving the stringent purity specifications required for injectable opioid antagonists.
How to Synthesize Naloxone Efficiently
The synthesis of Naloxone via this patented route requires meticulous attention to reaction parameters, particularly regarding temperature control and gas atmosphere management. The process begins with the dissolution of thebaine in formic acid, followed by the dropwise addition of oxidants under constant stirring. It is imperative to maintain the reaction temperature between 20°C and 40°C to prevent exothermic runaway and ensure selective oxidation. Following the oxidation phase, the system must be rigorously purged with nitrogen or argon before introducing hydrogen gas and the metal catalyst. The detailed standardized synthesis steps, including specific reagent ratios, workup procedures, and purification techniques, are outlined in the guide below to ensure reproducibility and safety in a GMP environment.
- Dissolve thebaine in formic acid and oxidize at 20-40°C, followed by hydrogenation using Raney Nickel or Pd/C catalyst to obtain Compound 2.
- React Compound 2 with acetic anhydride at 60-100°C to form the acetylated intermediate Compound 3.
- Perform cyclization using 1-chloroethyl chloroformate in toluene at 75-100°C, followed by acid hydrolysis to yield Compound 4.
- Alkylate Compound 4 with allyl chloride or cyclopropylmethyl bromide in the presence of base to form Compound 5.
- Demethylate Compound 5 using boron tribromide in an organic solvent at -10 to 40°C to finalize Naloxone or Naltrexone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the solvent supply chain and waste disposal logistics. By eliminating the need for chloroform and ethylene dichloride, manufacturers can avoid the volatile pricing and strict regulatory quotas associated with these controlled substances. This shift not only stabilizes raw material costs but also accelerates the procurement cycle, as safer solvents like toluene and ethanol are commodity chemicals with robust global availability. Furthermore, the reduced toxicity profile of the process lowers the barrier for contract manufacturing organizations (CMOs) to accept production campaigns, as fewer specialized safety certifications are required. This flexibility enhances supply chain reliability, ensuring continuous availability of high-purity naloxone even during market fluctuations.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous chlorinated solvents leads to significant cost savings in both raw material procurement and waste treatment. Traditional methods require costly incineration or specialized recycling for chlorinated waste, whereas the solvents used in this novel process (toluene, ethanol) can often be recovered and reused through standard distillation columns. Additionally, the use of heterogeneous catalysts like Raney Nickel allows for catalyst recovery and reuse, further driving down the cost per kilogram of the final API. The higher step yields observed in this process, particularly in the acetylation and alkylation stages where yields exceed 90%, reduce the amount of starting material required, directly impacting the variable cost of production.
- Enhanced Supply Chain Reliability: The reliance on commodity reagents and solvents ensures that the supply chain is resilient against disruptions. Unlike specialized reagents that may have single-source suppliers, chemicals like formic acid, acetic anhydride, and sodium bicarbonate are produced globally in massive quantities. This diversity of supply sources mitigates the risk of shortages that could halt production lines. Moreover, the milder reaction conditions (20-40°C) reduce the energy load on manufacturing facilities compared to high-temperature reflux processes, decreasing the dependency on high-capacity utility systems and allowing for production in a wider range of facilities. This operational flexibility is crucial for maintaining reducing lead time for high-purity pharmaceutical intermediates during peak demand periods.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is facilitated by the absence of highly toxic intermediates and solvents. The process generates less hazardous waste, simplifying the environmental impact assessment (EIA) required for new production lines. The use of solid catalysts that can be filtered off eliminates the need for complex aqueous workups to remove metal residues, reducing water consumption and wastewater volume. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the corporate social responsibility (CSR) profile of the brand. The robust nature of the reaction conditions ensures that quality remains consistent regardless of batch size, supporting the commercial scale-up of complex opioid antagonists.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and beneficial effects described in patent CN101955484A, providing clarity on process feasibility and advantages. Understanding these details is essential for technical teams evaluating the transfer of this technology to their own manufacturing sites.
Q: How does this synthesis method improve environmental safety compared to traditional routes?
A: This method replaces highly toxic chlorinated solvents like chloroform and ethylene dichloride with safer alternatives such as toluene, ethanol, and methanol, significantly reducing environmental pollution and operator health risks.
Q: What are the expected yields for the key intermediate steps?
A: The process demonstrates robust step yields, with the initial oxidation-hydrogenation step achieving 80-90%, acetylation reaching over 90%, and the final demethylation step yielding approximately 55-60%, resulting in a competitive total recovery rate.
Q: Which catalysts are utilized in the hydrogenation steps?
A: The process utilizes heterogeneous catalysts such as Raney Nickel or Palladium on Carbon (Pd/C), which allows for easy filtration and recovery of the catalyst, enhancing process efficiency and cost-effectiveness.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naloxone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant synthesis routes for life-saving medications like Naloxone. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped to handle the specific requirements of this patented process, including rigorous temperature control and inert atmosphere handling. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Naloxone or Naltrexone meets the highest international pharmacopoeia standards, providing you with a dependable source for your pharmaceutical formulations.
We invite you to collaborate with us to leverage this advanced technology for your product portfolio. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this greener synthetic route can optimize your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply chain for your opioid antagonist programs.
