Advanced Synthesis of 2-Quinoxalinol Derivatives for Commercial Scale-up
Advanced Synthesis of 2-Quinoxalinol Derivatives for Commercial Scale-up
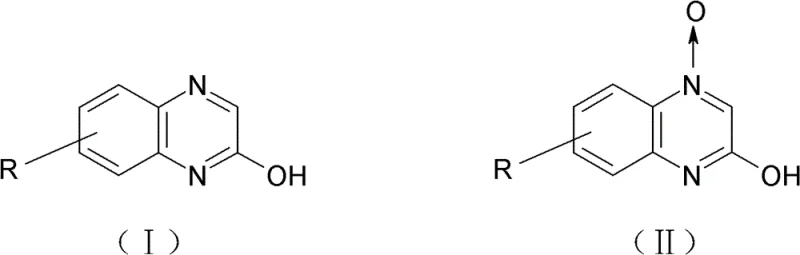
The pharmaceutical and agrochemical industries continuously seek robust synthetic routes for heterocyclic intermediates that balance safety, cost, and purity. Patent CN102108065B introduces a transformative method for preparing 2-quinoxalinol and its derivatives, specifically addressing the critical bottlenecks associated with traditional catalytic hydrogenation. This technology utilizes a selective chemical reduction strategy employing phosphorous acid or trimethyl phosphate to convert 2-quinoxalinol-4-oxide precursors into the desired final products. By shifting away from high-pressure hydrogen gas and heterogeneous metal catalysts, this innovation not only mitigates significant safety hazards but also streamlines the purification workflow. For R&D directors and procurement specialists, this represents a pivotal opportunity to optimize the supply chain for high-purity pharma intermediates while drastically reducing the operational risks inherent in legacy manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of quinoxaline N-oxides to their corresponding parent heterocycles has relied heavily on catalytic hydrogenation using Raney nickel or precious metals under hydrogen pressure. These conventional methods suffer from severe drawbacks, including the inherent explosion risks associated with handling high-pressure hydrogen gas in large-scale reactors. Furthermore, heterogeneous catalysts like Raney nickel are pyrophoric and require complex filtration and disposal protocols, adding substantial waste treatment costs. A major technical challenge is the lack of selectivity; metal-catalyzed hydrogenation often leads to over-reduction, generating difficult-to-remove impurities that compromise the purity profile required for sensitive API synthesis. Additionally, catalyst poisoning by sulfur or other heteroatoms present in the substrate can lead to inconsistent batch yields and unpredictable reaction kinetics, creating significant supply chain volatility for reliable agrochemical intermediates supplier networks.
The Novel Approach
The methodology disclosed in the patent circumvents these issues by employing homogeneous chemical reduction using phosphorous acid or trimethyl phosphate as the reducing agent. This approach operates under mild conditions, typically between 20°C and 120°C, eliminating the need for specialized high-pressure equipment and the associated safety infrastructure. The chemical selectivity of phosphorous acid towards the N-oxygen bond is exceptionally high, ensuring that the quinoxaline ring system remains intact without unwanted side reactions on other functional groups. This results in a cleaner reaction profile with fewer byproducts, directly translating to higher isolated yields and simplified downstream processing. By utilizing cheap and readily available commodity chemicals instead of expensive metal catalysts, the process achieves substantial cost reduction in pharma intermediates manufacturing while simultaneously enhancing the environmental profile by avoiding heavy metal contamination in the waste stream.
Mechanistic Insights into Phosphorous Acid-Mediated Deoxygenation
The core of this technological advancement lies in the specific mechanistic pathway of the deoxygenation reaction. In the presence of a polar aprotic solvent such as DMF or DMSO, the phosphorous acid or trimethyl phosphate acts as an oxygen acceptor. The reaction initiates with the nucleophilic attack or coordination of the phosphorus species to the oxygen atom of the N-oxide group. This interaction weakens the nitrogen-oxygen bond, facilitating its cleavage and the subsequent formation of a phosphorus-oxygen bond, which is thermodynamically very stable. This driving force ensures the reaction proceeds to completion with high efficiency. Unlike radical-based hydrogenation mechanisms which can be indiscriminate, this polar mechanism is highly specific to the N-oxide functionality, preserving other sensitive substituents on the aromatic ring. This specificity is crucial for synthesizing complex derivatives where maintaining the integrity of halogen or alkyl groups is essential for downstream biological activity.
Furthermore, the control of impurities is inherently built into this chemical mechanism. Since the reduction does not involve metal surfaces, there is no risk of metal leaching into the product, a critical parameter for meeting stringent regulatory limits in pharmaceutical applications. The byproduct of the reaction is typically a phosphate species, which is water-soluble and easily removed during the aqueous workup or recrystallization steps described in the patent embodiments. This ease of separation contrasts sharply with the removal of fine metal particles from viscous reaction mixtures. The ability to conduct the reaction in common industrial solvents like acetonitrile or DMF allows for excellent solubility of both the starting oxide and the final product, preventing premature precipitation that could trap impurities. Consequently, the final 2-quinoxalinol products exhibit superior purity profiles, making them ideal candidates for commercial scale-up of complex polymer additives or electronic materials where trace impurities can be detrimental.
How to Synthesize 2-Quinoxalinol Efficiently
The synthesis protocol outlined in the patent provides a clear, two-step pathway that is amenable to industrial adaptation. The process begins with the cyclization of chloro-m-nitroacetoacetanilide in the presence of a hydroxide base to form the key 2-quinoxalinol-4-oxide intermediate. This oxide is then subjected to the novel reduction conditions. The detailed operational parameters, including specific molar ratios, temperature gradients, and workup procedures, are critical for maximizing yield and ensuring reproducibility. For process chemists looking to implement this route, understanding the precise stoichiometry of the phosphorous reducing agent relative to the oxide substrate is vital to prevent excess reagent carryover. The following guide summarizes the standardized synthesis steps derived from the patent data to assist in rapid process validation.
- Cyclize chloro-m-nitroacetoacetanilide with alkali metal hydroxide to form the 2-quinoxalinol-4-oxide intermediate.
- Dissolve the oxide intermediate in a polar aprotic solvent such as DMF or DMSO.
- Add phosphorous acid or trimethyl phosphate and heat to 110°C to effect selective reduction to the final 2-quinoxalinol product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this phosphorous acid-mediated reduction technology offers profound advantages for supply chain stability and cost management. The elimination of high-pressure hydrogenation removes a major bottleneck in production scheduling, as facilities no longer need to queue for limited high-pressure reactor capacity. This flexibility allows for faster turnaround times and more responsive inventory management. Moreover, the raw materials involved—phosphorous acid and trimethyl phosphate—are bulk commodity chemicals with stable pricing and widespread availability, insulating the supply chain from the volatility often seen with precious metal catalysts. This reliability is essential for maintaining continuous production lines for high-purity OLED material or specialty chemical manufacturing where interruptions can be costly.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive catalytic systems with low-cost chemical reductants. By removing the need for Raney nickel or palladium catalysts, manufacturers eliminate the capital expenditure associated with catalyst recovery systems and the ongoing cost of catalyst replenishment. Additionally, the simplified workup procedure reduces labor hours and solvent consumption during purification. The high selectivity of the reaction minimizes the loss of valuable starting materials to byproducts, effectively increasing the overall mass balance efficiency. These factors combine to deliver substantial cost savings without compromising the quality of the final 2-quinoxalinol intermediate, making it a highly competitive option for cost reduction in electronic chemical manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by decoupling production from specialized hydrogenation infrastructure. Facilities that lack high-pressure capabilities can now produce these intermediates using standard glass-lined or stainless steel reactors. The non-pyrophoric nature of the reagents simplifies storage and handling logistics, reducing the regulatory burden and insurance costs associated with hazardous materials. This accessibility broadens the base of potential suppliers, reducing dependency on single-source vendors and mitigating the risk of supply disruptions. For procurement managers, this means securing a more robust supply of reliable pharma intermediates supplier sources that can scale up quickly to meet market demand fluctuations.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower than traditional methods, aligning with modern green chemistry principles. The absence of heavy metals in the waste stream simplifies effluent treatment and reduces the cost of hazardous waste disposal. The reaction conditions are mild enough to be safely scaled from pilot plant to multi-ton production without significant engineering redesigns. This scalability ensures that the process can grow alongside market demand for reducing lead time for high-purity pharmaceutical intermediates. Furthermore, the use of less toxic reagents contributes to a safer working environment, enhancing the overall sustainability profile of the manufacturing operation and ensuring compliance with increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation. They are designed to provide clarity on the operational feasibility and strategic value of adopting this technology for your specific application needs. Understanding these details is crucial for making informed decisions about process integration and supplier qualification.
Q: What are the safety advantages of using phosphorous acid over catalytic hydrogenation?
A: Using phosphorous acid eliminates the need for high-pressure hydrogen gas and pyrophoric Raney nickel catalysts, significantly reducing explosion risks and simplifying reactor safety requirements.
Q: How does this method improve product purity compared to traditional routes?
A: The chemical reduction using phosphorous acid or trimethyl phosphate offers high selectivity for the N-oxide bond, minimizing over-reduction byproducts often seen with metal-catalyzed hydrogenation.
Q: Is this process scalable for industrial production of quinoxaline intermediates?
A: Yes, the use of common commodity chemicals like phosphorous acid and standard polar aprotic solvents makes the process highly scalable without requiring specialized high-pressure hydrogenation infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Quinoxalinol Supplier
The technical superiority of the phosphorous acid reduction method positions 2-quinoxalinol as a highly viable candidate for large-scale commercial production. NINGBO INNO PHARMCHEM leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this innovative chemistry to the global market. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal requirements of this process, ensuring consistent batch-to-batch quality. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every shipment meets the exacting standards required for downstream API synthesis and advanced material applications.
We invite you to collaborate with us to optimize your supply chain for quinoxaline derivatives. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this greener, safer route can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your next project, ensuring you secure a competitive edge in the global marketplace.
