Scalable Synthesis of Stable Isotope Labeled Tritoquinol Hydrochloride for High-Precision Analytics
Scalable Synthesis of Stable Isotope Labeled Tritoquinol Hydrochloride for High-Precision Analytics
The pharmaceutical and analytical chemistry sectors are increasingly demanding high-fidelity reference standards to support rigorous regulatory compliance and advanced metabolic research. Patent CN112358446B discloses a robust and innovative synthetic methodology for producing stable isotope labeled tritoquinol hydrochloride, a compound of significant interest due to its classification as a beta-2 agonist and its presence on the World Anti-Doping Agency (WADA) prohibited list. This technology addresses the critical need for internal standards in Isotope Dilution Mass Spectrometry (IDMS), offering a pathway to generate materials with exceptional isotopic abundance and chemical purity. By leveraging a linear synthetic strategy starting from readily available commodity chemicals, this process ensures that the resulting labeled compounds can serve as authoritative benchmarks for athlete drug testing, food safety analysis, and pharmacokinetic studies. The ability to produce such specialized intermediates with undiluted isotope signatures represents a substantial advancement in the field of analytical reference materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for tritoquinol and its analogues often rely on early-stage introduction of functional groups or non-specific labeling strategies that can lead to significant isotopic dilution during downstream processing. Conventional methods may involve complex protection and deprotection sequences that increase the step count and reduce overall yield, thereby inflating the cost of goods for these high-value specialty chemicals. Furthermore, existing literature often lacks detailed protocols for introducing stable isotopes such as deuterium or Carbon-13 without compromising the structural integrity of the sensitive isoquinoline core. The reliance on harsh conditions in older methodologies can result in the scrambling of isotopic labels or the formation of difficult-to-remove impurities, which renders the final product unsuitable for high-precision mass spectrometry applications where signal-to-noise ratios are paramount.
The Novel Approach
The methodology outlined in patent CN112358446B introduces a streamlined and highly efficient route that overcomes these historical bottlenecks by utilizing a late-stage labeling strategy combined with robust cyclization techniques. This novel approach begins with the demethylation of 2-(3,4,5-trimethoxyphenyl)acetic acid to generate a reactive trihydroxy intermediate, which is then coupled with a protected phenethylamine derivative. Crucially, the stable isotope label is introduced via alkylation of the phenolic hydroxyl groups using labeled methyl iodide (such as CD3I or 13CH3I) at an intermediate stage, ensuring maximum retention of the isotopic signature throughout the subsequent ring-closing and reduction steps. This strategic placement of the labeling step minimizes waste and maximizes the economic efficiency of using expensive isotopic reagents.
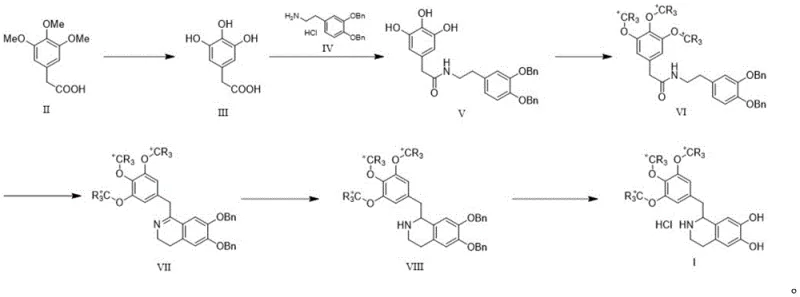
As illustrated in the reaction scheme, the process flows logically from simple precursors to the complex target molecule through a series of high-yielding transformations including amide formation, etherification, Bischler-Napieralski cyclization, and catalytic hydrogenolysis. The use of phosphorus oxychloride for cyclization and sodium borohydride for reduction provides a reliable and scalable framework that avoids the need for exotic catalysts or extreme reaction conditions. This clarity in synthetic design not only facilitates easier scale-up but also simplifies the purification profile, allowing manufacturers to achieve the stringent purity specifications required for certified reference materials without excessive chromatographic burden.
Mechanistic Insights into Isotope Incorporation and Cyclization
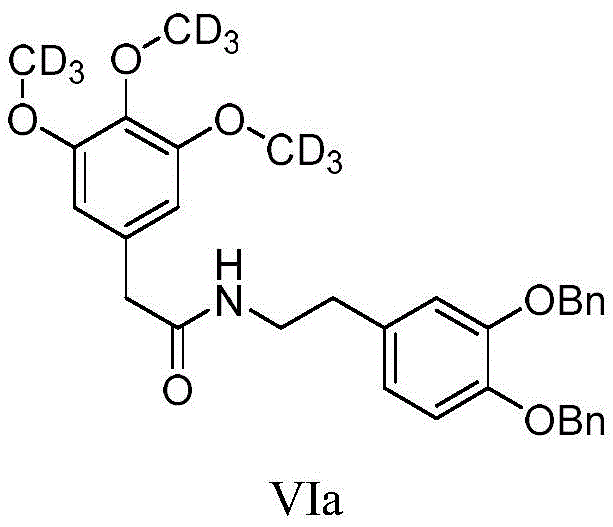
The core of this synthetic innovation lies in the precise control of isotopic incorporation during the etherification of the phenolic intermediate. In the transformation of Compound V to Compound VI, the reaction utilizes a strong base such as potassium carbonate in a polar aprotic solvent like DMF to deprotonate the phenolic hydroxyl groups, generating nucleophilic phenoxide ions. These ions then attack the electrophilic carbon of the stable isotope-labeled methyl iodide (e.g., CD3I) in an SN2 mechanism, effectively installing the deuterated methoxy groups onto the aromatic ring. This step is critical because it locks the isotopic label into the molecular framework prior to the formation of the heterocyclic ring, ensuring that the label remains intact during the subsequent acidic cyclization conditions.
Following the labeling step, the synthesis proceeds through a classic Bischler-Napieralski cyclization mediated by phosphorus oxychloride (POCl3). This reaction involves the activation of the amide carbonyl oxygen by the phosphorus species, followed by intramolecular electrophilic aromatic substitution to close the dihydroisoquinoline ring (Compound VII). The mechanism is highly efficient and tolerates the electron-rich nature of the labeled trimethoxyphenyl ring. Subsequent reduction of the imine double bond in Compound VII using sodium borohydride yields the tetrahydroisoquinoline scaffold (Compound VIII) with high stereoselectivity and chemoselectivity. The final step involves catalytic debenzylation using palladium on carbon and ammonium formate as a hydrogen source, which cleanly removes the benzyl protecting groups to reveal the active catechol moiety, followed by salt formation with hydrochloric acid to yield the final stable isotope labeled tritoquinol hydrochloride.
How to Synthesize Stable Isotope Labeled Tritoquinol Hydrochloride Efficiently
The synthesis of this high-value intermediate requires careful attention to reaction stoichiometry and temperature control to maintain isotopic integrity. The process begins with the preparation of the trihydroxy precursor, followed by coupling and labeling, and concludes with ring closure and deprotection. Each step has been optimized in the patent examples to balance reaction rate with product stability. For detailed operational parameters, reagent grades, and workup procedures necessary to replicate this high-purity synthesis in a GMP environment, please refer to the standardized technical guide below.
- Demethylation of 2-(3,4,5-trimethoxyphenyl)acetic acid using boron tribromide to form the trihydroxy intermediate.
- Condensation with 2-(3,4-bis(benzyloxy)phenyl)ethylamine followed by methylation with stable isotope reagents like CD3I.
- Cyclization using phosphorus oxychloride, reduction with sodium borohydride, and final catalytic debenzylation to yield the target hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers distinct advantages in terms of cost stability and raw material security. The starting materials, such as 2-(3,4,5-trimethoxyphenyl)acetic acid and protected phenethylamines, are commodity chemicals that are widely available from multiple global suppliers, reducing the risk of single-source dependency. By utilizing a linear synthesis that avoids complex chiral resolution steps or rare transition metal catalysts, the manufacturing process significantly lowers the barrier to entry for production, which translates into more competitive pricing structures for the final isotope-labeled product. This accessibility is crucial for laboratories and pharmaceutical companies that require consistent access to internal standards for long-term regulatory monitoring programs.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive chiral catalysts or enzymatic resolutions, relying instead on robust chemical transformations that utilize standard industrial reagents. By introducing the expensive isotopic label at a mid-stage rather than the very beginning, the process minimizes the loss of valuable labeled atoms during early synthetic steps, thereby optimizing the utilization of high-cost isotopic reagents. Furthermore, the high chemical purity achieved directly from the synthesis reduces the need for extensive and costly downstream purification processes such as preparative HPLC, leading to substantial operational savings.
- Enhanced Supply Chain Reliability: The reliance on common solvents like dichloromethane, DMF, and ethanol, along with standard reagents like thionyl chloride and sodium borohydride, ensures that the supply chain is resilient to market fluctuations. The synthetic route is designed to be scalable from gram to kilogram quantities without requiring specialized equipment, allowing manufacturers to respond quickly to surges in demand driven by regulatory changes or new doping control requirements. This flexibility ensures a continuous supply of critical reference materials, preventing bottlenecks in the quality control workflows of major testing laboratories.
- Scalability and Environmental Compliance: The synthetic steps described are amenable to large-scale batch processing, with reaction conditions that can be easily managed in standard stainless steel reactors. The use of catalytic hydrogenolysis for the final deprotection step generates benign byproducts, aligning with modern green chemistry principles and simplifying waste treatment protocols. The overall atom economy of the route is favorable, particularly in the labeling step where the stoichiometry is tightly controlled to maximize yield. This environmental efficiency not only reduces disposal costs but also supports the sustainability goals of modern pharmaceutical supply chains.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of isotope-labeled synthesis is essential for stakeholders involved in analytical method development and regulatory compliance. The following questions address common inquiries regarding the stability, application, and production of these specialized compounds. The answers provided are derived directly from the technical disclosures and experimental data within the patent documentation to ensure accuracy and relevance.
Q: What is the primary application of stable isotope labeled tritoquinol hydrochloride?
A: It serves as a critical internal standard for Isotope Dilution Mass Spectrometry (IDMS) in anti-doping tests (WADA compliant) and pharmaceutical quality control, ensuring high accuracy in trace analysis.
Q: How does this synthesis method prevent isotope dilution?
A: The route introduces the stable isotope label (e.g., deuterium or Carbon-13) at a mid-stage intermediate (Compound V to VI) using labeled methyl iodide, ensuring the label is not lost during subsequent harsh cyclization or reduction steps.
Q: What represents the key purity advantage of this process?
A: The process achieves chemical purity exceeding 99% and isotopic abundance greater than 99%, which is essential for meeting the rigorous standards of reference material certification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Stable Isotope Labeled Tritoquinol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality reference standards play in ensuring public safety and regulatory compliance in the pharmaceutical and sports industries. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which are equipped with state-of-the-art analytical instrumentation to verify isotopic abundance and chemical identity. Our dedication to quality assurance means that every batch of stable isotope labeled tritoquinol hydrochloride is thoroughly characterized to support your most demanding analytical applications.
We invite you to collaborate with us to optimize your supply chain for these essential analytical reagents. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and delivery schedules. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and quotations that reflect our commitment to being your trusted partner in fine chemical manufacturing.
