Scalable Production of 10α-Hydroxynalfurafine via Iridium-Catalyzed Reductive Amination
Scalable Production of 10α-Hydroxynalfurafine via Iridium-Catalyzed Reductive Amination
The pharmaceutical industry's relentless pursuit of purity and safety has placed impurity profiling at the forefront of drug development, particularly for potent opioid receptor agonists like nalfurafine. As detailed in patent CN109970747B, a groundbreaking methodology has been established for the preparation of 10α-hydroxynalfurafine, a critical related substance and potential metabolite that requires rigorous monitoring. This novel synthetic approach addresses the historical bottlenecks of reference standard production by leveraging commercially available naltrexone hydrochloride as the foundational starting material. By integrating selective oxidation strategies with advanced iridium-catalyzed chiral reductive amination, this process delivers high-purity intermediates essential for validating analytical methods and ensuring patient safety in the final dosage forms.
The structural complexity of 10α-hydroxynalfurafine, characterized by its morphinan backbone and specific stereochemistry at the C-10 position, presents significant challenges for traditional organic synthesis. 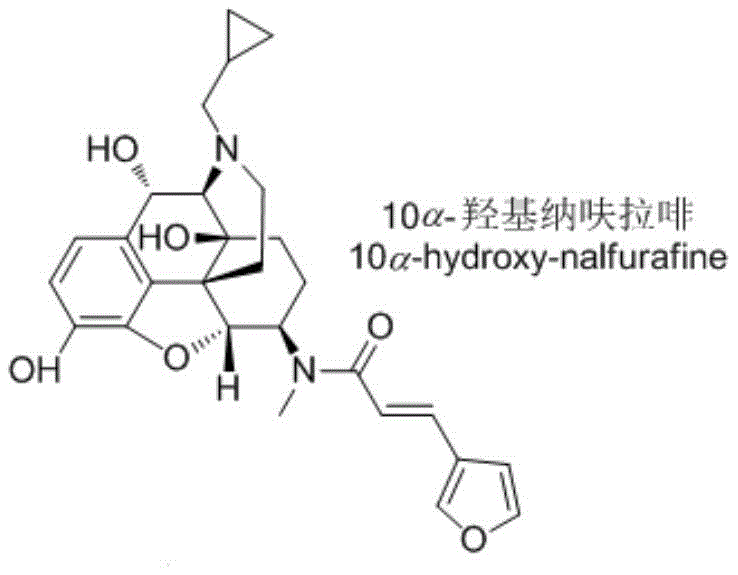 . The ability to synthesize this molecule efficiently is not merely an academic exercise but a regulatory necessity for any manufacturer aiming to bring nalfurafine-based therapies to market. The patent outlines a robust six-step sequence that maximizes atom economy and minimizes waste, reflecting a modern approach to green chemistry within the fine chemical sector. For R&D directors and procurement specialists, understanding the nuances of this pathway is vital for securing a reliable supply of high-quality reference materials.
. The ability to synthesize this molecule efficiently is not merely an academic exercise but a regulatory necessity for any manufacturer aiming to bring nalfurafine-based therapies to market. The patent outlines a robust six-step sequence that maximizes atom economy and minimizes waste, reflecting a modern approach to green chemistry within the fine chemical sector. For R&D directors and procurement specialists, understanding the nuances of this pathway is vital for securing a reliable supply of high-quality reference materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations disclosed in CN109970747B, the synthesis of 10α-hydroxynalfurafine was hindered by significant logistical and chemical inefficiencies, primarily documented in earlier literature such as Chemical & Pharmaceutical Bulletin (2004). The conventional routes often relied on starting materials that were not commercially available, necessitating complex, multi-step pre-syntheses that drastically inflated costs and extended lead times. Furthermore, these legacy pathways typically involved seven or more reaction steps, each introducing potential yield losses and impurity profiles that complicated downstream purification. 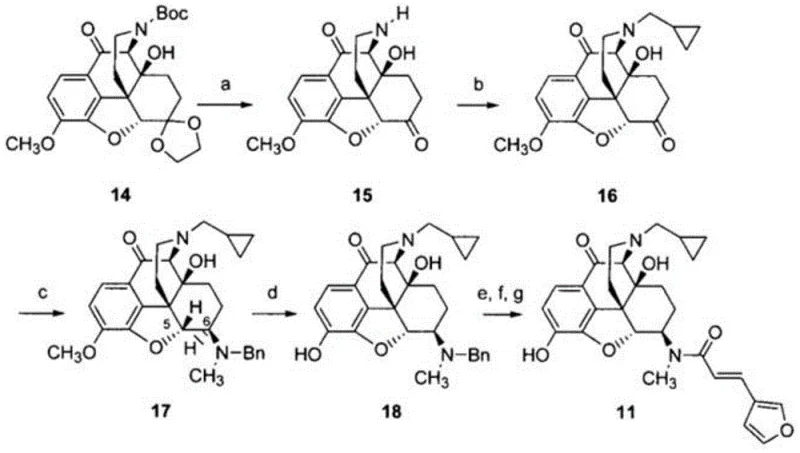 . The reliance on obscure precursors meant that supply chains were fragile, susceptible to disruption, and incapable of supporting the rapid scale-up required for clinical trial material or commercial QC testing. Additionally, the lack of stereochemical control in some older methods resulted in difficult-to-separate diastereomers, compromising the purity required for a certified reference standard.
. The reliance on obscure precursors meant that supply chains were fragile, susceptible to disruption, and incapable of supporting the rapid scale-up required for clinical trial material or commercial QC testing. Additionally, the lack of stereochemical control in some older methods resulted in difficult-to-separate diastereomers, compromising the purity required for a certified reference standard.
The Novel Approach
In stark contrast, the methodology presented in this patent revolutionizes the production landscape by utilizing naltrexone hydrochloride, a widely accessible and cost-effective bulk pharmaceutical chemical, as the sole starting point. This strategic shift eliminates the need for custom synthesis of precursors, immediately reducing the barrier to entry and stabilizing the supply chain. The new route condenses the synthesis into six highly efficient steps, each optimized for high conversion rates and minimal byproduct formation. By employing a protective group strategy that safeguards the ketone carbonyl and phenolic hydroxyl groups early in the sequence, the process ensures that subsequent reactions, such as the critical C-10 oxidation, proceed with exceptional regioselectivity. This streamlined approach not only accelerates the timeline from raw material to finished reference standard but also enhances the overall sustainability of the manufacturing process by reducing solvent consumption and waste generation associated with additional synthetic steps.
Mechanistic Insights into Selective Oxidation and Chiral Reductive Amination
The core chemical innovation of this synthesis lies in the precise manipulation of the morphinan skeleton, specifically the installation of the 10α-hydroxyl group and the subsequent establishment of the amine stereochemistry. The selective oxidation step utilizes Cerium Ammonium Nitrate (CAN) in acetonitrile at a controlled low temperature of 5°C. This specific condition is crucial for achieving the desired α-orientation of the hydroxyl group while preventing over-oxidation or degradation of the sensitive ether linkages within the molecule. The mechanism likely involves a single-electron transfer process where the cerium(IV) species abstracts a hydrogen atom from the C-10 position, followed by oxygen rebound, strictly governed by the steric constraints of the protected intermediate. This level of control is paramount for R&D teams, as it ensures that the resulting impurity profile matches the specific metabolic byproducts found in biological systems, thereby validating the analytical methods used for batch release.
Following deprotection, the synthesis employs a sophisticated iridium-catalyzed asymmetric reductive amination to introduce the N-methyl group with high fidelity. Using a catalyst system composed of [Ir(COD)Cl]2 and the chiral ligand (R)-(S)-Xyliphos, the reaction proceeds via an iminium ion intermediate formed from the ketone and methylamine. The chiral environment created by the Xyliphos ligand directs the hydride delivery from formic acid to a specific face of the iminium ion, ensuring the formation of the correct stereoisomer at the C-7 position (using morphinan numbering). This step is a masterclass in modern catalysis, replacing stoichiometric reducing agents with a catalytic cycle that is both atom-economical and highly selective. For quality control laboratories, this means the final product exhibits a clean chromatographic profile, free from the diastereomeric impurities that often plague non-catalytic reductive aminations, thus simplifying the validation of HPLC methods.
How to Synthesize 10α-Hydroxynalfurafine Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly temperature control during the oxidation phase and the exclusion of moisture during the Lewis acid-mediated deprotection. The process begins with the formation of a cyclic acetal to protect the C-6 ketone, followed by methylation of the phenol to prevent oxidative coupling. 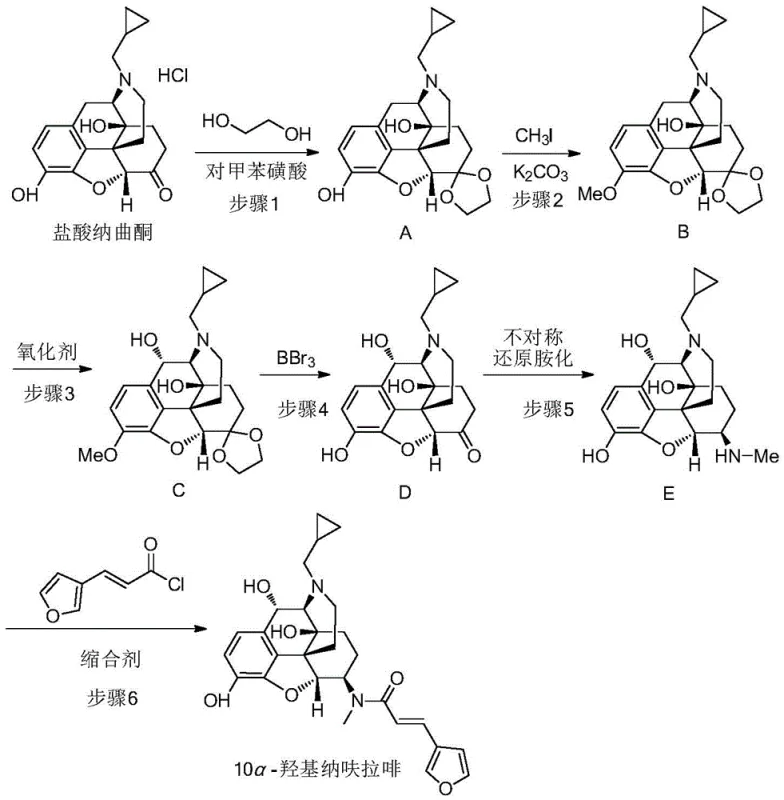 . The subsequent oxidation and deprotection steps reveal the reactive ketone at C-6, which is then subjected to the chiral amination. The final step involves coupling the amine intermediate with (E)-3-(furan-3-yl)-acryloyl chloride using PYBOP as a coupling agent. Detailed standardized operating procedures for scaling this pathway from gram to kilogram quantities are provided in the structured guide below, ensuring reproducibility across different manufacturing sites.
. The subsequent oxidation and deprotection steps reveal the reactive ketone at C-6, which is then subjected to the chiral amination. The final step involves coupling the amine intermediate with (E)-3-(furan-3-yl)-acryloyl chloride using PYBOP as a coupling agent. Detailed standardized operating procedures for scaling this pathway from gram to kilogram quantities are provided in the structured guide below, ensuring reproducibility across different manufacturing sites.
- Protect the ketone carbonyl of naltrexone hydrochloride using ethylene glycol and p-toluenesulfonic acid to form the acetal intermediate.
- Perform phenolic hydroxyl methylation using methyl iodide and potassium carbonate, followed by selective oxidation at the C-10 position using CAN.
- Execute deprotection with boron tribromide, followed by iridium-catalyzed chiral reductive amination and final amidation with furan-acryloyl chloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers profound strategic benefits that extend beyond simple unit cost calculations. The primary advantage stems from the utilization of naltrexone hydrochloride, a commodity chemical with a mature, global supply network, which effectively decouples the production of this reference standard from the volatility of custom intermediate markets. By eliminating the dependency on proprietary or hard-to-source starting materials, manufacturers can secure long-term supply contracts with greater confidence, mitigating the risk of production halts due to raw material shortages. Furthermore, the reduction in total step count from seven to six, combined with the high yields reported in the patent examples, translates directly into a substantial reduction in operational expenditures related to labor, reactor occupancy time, and solvent procurement. This efficiency gain allows for a more competitive pricing structure without compromising the stringent purity specifications required for pharmaceutical reference standards.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive, custom-synthesized precursors and the optimization of reaction yields at every stage. By starting with a commercially available bulk drug substance, the initial material cost is significantly lower than that of specialized organic building blocks. Additionally, the high efficiency of the iridium-catalyzed step reduces the need for extensive chromatographic purification, which is often the most costly unit operation in fine chemical manufacturing. The cumulative effect of these improvements results in a drastically simplified cost structure, enabling the production of high-purity 10α-hydroxynalfurafine at a fraction of the cost associated with legacy methods.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved by the reliance on off-the-shelf reagents such as ethylene glycol, methyl iodide, and standard coupling agents like PYBOP. These materials are produced by multiple vendors globally, ensuring that no single point of failure can disrupt the manufacturing schedule. The robustness of the reaction conditions, which tolerate standard industrial solvents like dichloromethane and acetonitrile, further facilitates technology transfer between different contract manufacturing organizations (CMOs). This flexibility empowers supply chain heads to diversify their manufacturing base, negotiating better terms and ensuring continuous availability of critical quality control materials even during periods of market fluctuation.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process is designed to minimize hazardous waste generation. The use of catalytic amounts of iridium, rather than stoichiometric metal hydrides, aligns with green chemistry principles and reduces the burden on waste treatment facilities. The reaction conditions are generally mild, avoiding extreme temperatures or pressures that would require specialized, high-cost equipment for scale-up. This accessibility means that the process can be readily transferred to larger reactors (100 L to 1000 L scale) with minimal re-engineering, ensuring that production capacity can be rapidly expanded to meet increasing demand for nalfurafine impurity standards as the parent drug gains market traction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis protocol. These insights are derived directly from the experimental data and claims within patent CN109970747B, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of in-house production versus outsourcing to a specialized CDMO partner.
Q: What is the primary advantage of this synthesis route over prior art?
A: Unlike previous methods requiring unavailable starting materials and lengthy 7-step sequences, this patent utilizes commercially available naltrexone hydrochloride, significantly shortening the synthesis to 6 high-yielding steps.
Q: How is stereochemical control achieved at the 10-position?
A: Stereocontrol is managed through selective oxidation using Cerium Ammonium Nitrate (CAN) at low temperatures (5°C) to install the 10α-hydroxyl group, followed by asymmetric reductive amination using an Iridium catalyst with (R)-(S)-Xyliphos ligand.
Q: Is this process suitable for large-scale manufacturing of reference standards?
A: Yes, the process employs mild reaction conditions, such as room temperature amidation and standard solvent systems like dichloromethane and acetonitrile, making it highly amenable to commercial scale-up for impurity profiling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 10α-Hydroxynalfurafine Supplier
As the global demand for high-purity opioid receptor agonists continues to rise, the need for certified reference standards and impurity profiles becomes increasingly critical for regulatory compliance. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in complex morphinan chemistry to deliver superior pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of multinational pharmaceutical clients. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify the identity and potency of every batch of 10α-hydroxynalfurafine we produce.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for nalfurafine-related projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume needs and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply chain security.
