High-Efficiency Blue Fluoranthene OLED Materials: Scalable Synthesis & Commercial Supply
The landscape of organic light-emitting diodes (OLEDs) has been revolutionized by the demand for high-efficiency blue emitters, a critical component for full-color displays and solid-state lighting. Patent CN102924296B introduces a groundbreaking class of fluoranthene-containing organic compounds that address the longstanding challenges of efficiency roll-off and thermal instability in blue OLED materials. Unlike traditional spirofluorene derivatives which suffer from complex synthesis and poor solubility, these novel structures integrate a rigid fluoranthene core with electron-donating triphenylamine units, tunable via diverse substituents ranging from alkyl chains to cyano groups. This strategic molecular design not only enhances hole mobility but also drastically improves film-forming properties, making them ideal candidates for next-generation flat-panel displays. The versatility of the general formula, allowing for R1 and R2 modifications, enables precise tuning of electronic properties to match specific device architectures.
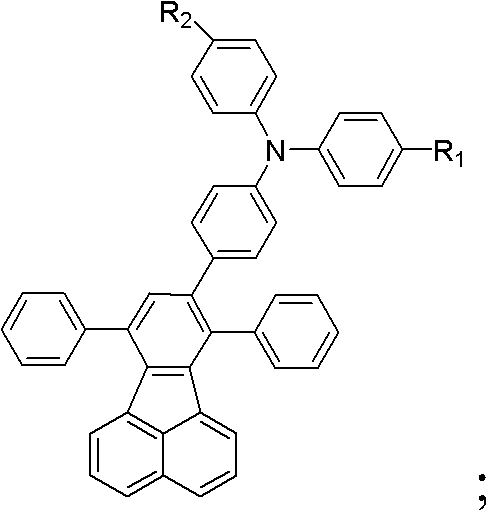
Historically, the development of blue OLED materials has been hindered by the trade-off between luminescence efficiency and processability. Conventional approaches often relied on unsubstituted fluoranthene derivatives like 7,8,10-triphenylfluoranthene (TPF), which, while exhibiting high quantum efficiency, suffered from poor solubility and a tendency to crystallize, leading to defective films and low device yields. Furthermore, the synthesis of alternative high-performance materials like spirofluorenes involved multi-step routes with low overall yields and difficult purification processes. The novel approach detailed in this patent overcomes these limitations by introducing flexible alkyl or alkoxy chains at the periphery of the triphenylamine moiety. This modification disrupts molecular aggregation, significantly enhancing solubility in common organic solvents without compromising the electronic integrity of the fluoranthene core. Consequently, this leads to smoother, more uniform thin films and higher synthesis yields, directly translating to cost-effective manufacturing.
Mechanistic Insights into the Three-Step Synthetic Pathway
The synthesis of these advanced fluoranthene derivatives relies on a robust three-step sequence that ensures high purity and structural fidelity. The process initiates with a palladium-copper co-catalyzed Sonogashira cross-coupling reaction, where a brominated triphenylamine precursor reacts with trimethylsilylacetylene in a triethylamine solvent system. This step is critical for installing the alkyne handle required for subsequent ring closure, and the use of a silyl protecting group prevents unwanted polymerization or side reactions of the terminal alkyne. Following this, a mild deprotection step using sodium hydroxide in a tetrahydrofuran and methanol mixture cleanly removes the trimethylsilyl group to reveal the reactive terminal ethynyl functionality. This two-step functionalization prepares the triphenylamine building block for the final, decisive transformation.
The culmination of the synthesis is a thermal cyclization reaction performed at elevated temperatures (170°C) in a high-boiling solvent such as xylene. In this step, the ethynyl-triphenylamine intermediate reacts with 7,9-diphenyl-cyclopentenonaphthalen-8-one to construct the fused fluoranthene ring system. This intramolecular cyclization is highly efficient, driven by the thermodynamic stability of the newly formed aromatic system. The reaction conditions are optimized to minimize side products, and the resulting crude material can be purified via standard column chromatography to achieve electronic-grade purity. This mechanistic pathway avoids the use of harsh reagents or unstable intermediates, ensuring a safe and reproducible process suitable for industrial scale-up.
How to Synthesize Fluoranthene Derivatives Efficiently
The patented methodology offers a streamlined route to high-performance OLED emitters, balancing chemical complexity with operational simplicity. By leveraging well-established cross-coupling and cyclization chemistries, the process minimizes the need for exotic reagents while maximizing yield and purity. The modular nature of the synthesis allows for the easy incorporation of various functional groups, enabling the customization of material properties for specific applications. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below which outlines the critical control points for each stage of the reaction.
- Perform Sonogashira coupling of bromo-triphenylamine with trimethylsilylacetylene using Pd/Cu catalyst in triethylamine.
- Execute deprotection of the trimethylsilyl group using NaOH in a THF/methanol mixture to reveal the terminal alkyne.
- Conduct thermal cyclization with 7,9-diphenyl-cyclopentenonaphthalen-8-one in xylene at 170°C to form the fluoranthene core.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the adoption of this fluoranthene-based technology represents a significant opportunity to optimize material costs and secure long-term supply stability. The synthetic route utilizes commodity chemicals and widely available catalysts, reducing dependency on scarce or expensive proprietary reagents. Furthermore, the improved solubility of the final products simplifies downstream processing, reducing solvent consumption and waste generation during device fabrication. This efficiency gain is crucial for maintaining competitive pricing in the volatile display materials market while ensuring consistent quality across large production batches.
- Cost Reduction in Manufacturing: The elimination of complex multi-step syntheses associated with traditional spiro-compounds leads to substantial cost savings. By utilizing a concise three-step pathway with high-yielding reactions, the overall material cost is significantly reduced. Additionally, the enhanced solubility reduces the need for specialized high-boiling solvents or extensive heating during film deposition, further lowering the operational expenditure for display manufacturers.
- Enhanced Supply Chain Reliability: The starting materials, such as brominated triphenylamines and cyclopentenonaphthalenones, are commercially accessible and can be sourced from multiple suppliers, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions ensures high batch-to-batch consistency, which is vital for maintaining uninterrupted production lines in high-volume display manufacturing facilities.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram to multi-gram scales in the patent examples. The use of standard organic solvents like triethylamine and xylene facilitates straightforward waste management and recycling protocols. This aligns with increasingly stringent environmental regulations, allowing manufacturers to maintain compliance without sacrificing production throughput or material performance.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of these new materials is essential for integrating them into existing device architectures. The following questions address common inquiries regarding the stability, processing, and performance characteristics of these fluoranthene derivatives. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering a clear picture of what to expect during implementation.
Q: How does the substituent strategy improve material processability?
A: By introducing alkyl or alkoxy chains (C1-C20) at the R1 and R2 positions, the molecular symmetry is disrupted and intermolecular stacking is reduced. This significantly enhances solubility in common organic solvents, facilitating solution-processing techniques like spin-coating and inkjet printing for uniform film formation.
Q: What are the thermal stability characteristics of these fluoranthene derivatives?
A: The rigid fluoranthene backbone combined with the triphenylamine moiety provides exceptional thermal stability. This structural rigidity prevents crystallization during device operation, ensuring long-term morphological stability of the emissive layer and consistent blue light output over extended periods.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the three-step pathway utilizes robust and scalable reactions such as Sonogashira coupling and thermal cyclization. The use of standard solvents like triethylamine and xylene, along with readily available catalysts, allows for straightforward scale-up from gram-scale laboratory synthesis to multi-kilogram commercial manufacturing without complex purification bottlenecks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoranthene Derivatives Supplier
As the demand for high-efficiency blue OLED materials continues to surge, partnering with an experienced chemical manufacturer is paramount for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of fluoranthene derivative meets the exacting standards required for premium display applications. We understand the critical nature of material consistency in OLED manufacturing and are committed to delivering products that enhance device longevity and performance.
We invite you to collaborate with our technical team to explore how these advanced materials can optimize your product lineup. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and production planning, ensuring a seamless transition to next-generation blue emitter technology.
