Mastering Formylated Bicyclol Synthesis for High-Purity API Impurity Standards
The pharmaceutical industry's relentless pursuit of patient safety hinges on the rigorous control of impurities within Active Pharmaceutical Ingredients (APIs). A pivotal advancement in this domain is detailed in Chinese Patent CN112250658B, which discloses a robust and scalable method for the preparation of Formylated Bicyclol. Bicyclol, a renowned hepatoprotective agent developed for treating chronic viral hepatitis, requires stringent quality monitoring due to the potential toxicity of its degradation products and process-related impurities. This patent addresses a critical gap in the market by providing a directed synthetic route to generate Formylated Bicyclol, a specific impurity that naturally occurs during Bicyclol manufacturing but was previously difficult to obtain in sufficient quantities for analytical standardization. By establishing a reliable method to synthesize this compound with purity exceeding 99%, the technology empowers quality control laboratories to implement precise detection protocols, thereby safeguarding the therapeutic integrity of the final drug product.
For procurement specialists and supply chain managers, the implications of this technology extend far beyond mere compliance; it represents a strategic opportunity for cost reduction in API manufacturing and quality assurance workflows. Traditionally, acquiring reference standards for trace impurities involved laborious isolation from bulk crude materials, a process fraught with low yields and inconsistent supply. The methodology outlined in CN112250658B transforms this challenge into a streamlined chemical operation, utilizing readily available reagents like phosphorus oxychloride and N,N-dimethylformamide. As a reliable pharmaceutical intermediate supplier, understanding and leveraging such patented processes allows organizations to secure a stable supply of critical reference materials, reducing lead time for high-purity pharmaceutical intermediates and ensuring uninterrupted production schedules for the parent API.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovation described in this patent, the availability of Formylated Bicyclol was severely restricted, creating a significant bottleneck for pharmaceutical quality control departments. The conventional approach relied entirely on the incidental formation of this impurity during the bulk synthesis of Bicyclol, where it typically exists at trace levels, often around 0.25% in crude products. Isolating milligram quantities of this substance from tons of crude API required extensive preparative chromatography and repeated recrystallization, resulting in prohibitive costs and extremely low recovery rates. Furthermore, the lack of a dedicated synthetic route meant that there was no guarantee of batch-to-batch consistency for the reference standard, complicating the validation of analytical methods. This scarcity not only hindered the ability to accurately quantify the impurity in commercial batches but also posed a risk of non-compliance with increasingly strict international regulatory guidelines regarding impurity profiling and identification.
The Novel Approach
The novel approach presented in the patent circumvents these historical limitations by employing a targeted Vilsmeier-Haack type formylation strategy. Instead of scavenging for the impurity, the method constructs the molecule deliberately from the parent Bicyclol structure. By reacting Bicyclol with a pre-formed or in-situ generated Vilsmeier reagent—derived from the reaction of phosphorus oxychloride and DMF—the process selectively installs the formyloxy group onto the hydroxymethyl moiety of the biphenyl scaffold. This chemical transformation is highly efficient and controllable, allowing manufacturers to produce the target impurity standard in substantial quantities rather than trace amounts. The shift from passive isolation to active synthesis ensures a consistent, high-purity supply of Formylated Bicyclol, facilitating the commercial scale-up of complex organic molecules needed for comprehensive safety testing.
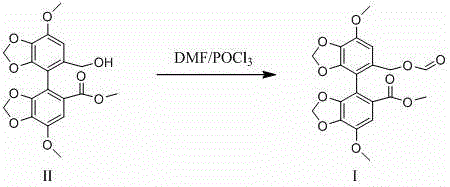
Mechanistic Insights into Vilsmeier-Haack Formylation
The core of this synthetic breakthrough lies in the mechanistic elegance of the Vilsmeier-Haack reaction conditions adapted for O-formylation. The process initiates with the interaction between N,N-dimethylformamide (DMF) and phosphorus oxychloride (POCl3) at low temperatures, typically between 0°C and 10°C. This exothermic step generates a highly reactive chloromethyleneiminium chloride species, commonly known as the Vilsmeier reagent. Strict temperature control during this phase is paramount to prevent thermal runaway and the decomposition of the active electrophile. Once formed, this electrophilic species attacks the nucleophilic oxygen of the hydroxymethyl group (-CH2OH) present on the Bicyclol substrate. Unlike traditional C-formylation of electron-rich aromatic rings, this specific application targets the alcohol functionality, converting it into a formate ester (-CH2-O-CHO). The reaction mixture is subsequently heated to a second temperature range of 80°C to 100°C, providing the necessary activation energy to drive the substitution to completion while minimizing the formation of side products.
Impurity control is intrinsic to the design of this mechanism, as the specificity of the Vilsmeier reagent under these controlled conditions limits over-reaction or degradation of the sensitive methylenedioxy bridges within the Bicyclol structure. The patent data indicates that maintaining the molar ratio of phosphorus oxychloride to Bicyclol between 1.0 and 3.0 is critical; excess reagent can lead to chlorination side reactions, while insufficient reagent results in incomplete conversion. Following the reaction, the workup involves a straightforward extraction with ethyl acetate and water, effectively separating the organic product from inorganic phosphorus byproducts. Final purification via column chromatography using a dichloromethane and petroleum ether system yields the target Formylated Bicyclol with a purity greater than 99%, demonstrating the method's capability to produce analytical-grade standards suitable for HPLC calibration and mass spectrometry confirmation.
How to Synthesize Formylated Bicyclol Efficiently
The synthesis of Formylated Bicyclol described in patent CN112250658B offers a reproducible pathway for generating high-quality reference standards essential for pharmaceutical analysis. The procedure balances reaction kinetics with safety considerations, particularly regarding the handling of phosphorus oxychloride. By adhering to the specified temperature gradients and stoichiometric ratios, chemists can achieve optimal yields while maintaining the structural integrity of the complex biphenyl backbone. The following guide outlines the standardized operational protocol derived from the patent examples, serving as a foundational reference for laboratory execution.
- Dissolve Bicyclol in N,N-Dimethylformamide (DMF) at room temperature and cool the solution to 0-10°C.
- Dropwise add phosphorus oxychloride (POCl3) while maintaining the temperature below 10°C to generate the active Vilsmeier reagent in situ.
- Heat the reaction mixture to 80-100°C to complete the formylation, followed by aqueous workup and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology translates into tangible operational efficiencies and risk mitigation strategies. The ability to internally produce or source Formylated Bicyclol through a defined chemical route eliminates the dependency on unpredictable isolation from crude API batches. This shift fundamentally alters the cost structure associated with quality control, moving from a high-cost, low-yield recovery process to a predictable manufacturing model. Consequently, organizations can achieve significant cost savings by reducing the man-hours and solvent volumes previously required for impurity isolation. Furthermore, securing a dedicated supply line for this critical reference standard enhances the overall resilience of the supply chain, ensuring that regulatory audits and batch release testing are never delayed due to the unavailability of comparison standards.
- Cost Reduction in Manufacturing: The transition from isolation to directed synthesis drastically reduces the resource intensity associated with obtaining Formylated Bicyclol. Traditional isolation required processing massive volumes of crude Bicyclol to recover minute quantities of the impurity, involving extensive chromatographic separation and solvent consumption. In contrast, the new method utilizes commodity chemicals like DMF and phosphorus oxychloride to convert the parent drug directly into the impurity standard. This efficiency eliminates the need for expensive preparative HPLC runs on crude mixtures, thereby lowering the cost per milligram of the reference standard significantly. Additionally, the simplified workup procedure reduces waste disposal costs and minimizes the consumption of high-purity solvents, contributing to a leaner and more economical quality control budget.
- Enhanced Supply Chain Reliability: Relying on the incidental presence of impurities in crude API batches introduces significant volatility into the supply of reference standards. Variations in the manufacturing process of the parent API could lead to fluctuations in impurity profiles, potentially resulting in batches with insufficient levels of Formylated Bicyclol for isolation. The patented synthetic route decouples the supply of the reference standard from the variability of the main API production. By establishing an independent synthesis channel, companies ensure a continuous and reliable availability of the standard, regardless of the impurity content in commercial Bicyclol batches. This reliability is crucial for maintaining uninterrupted quality control operations and meeting strict regulatory submission timelines without the risk of material shortages.
- Scalability and Environmental Compliance: The process described is inherently scalable, utilizing standard unit operations such as cooled addition, heating, liquid-liquid extraction, and column chromatography. These are well-understood processes in the fine chemical industry, allowing for seamless translation from laboratory bench scale to pilot and commercial production. From an environmental perspective, the method avoids the use of exotic or highly toxic catalysts, relying instead on common reagents that can be managed through established waste treatment protocols. The high selectivity of the reaction minimizes the generation of complex byproduct mixtures, simplifying downstream purification and reducing the environmental footprint associated with solvent-intensive purification steps. This alignment with green chemistry principles supports corporate sustainability goals while maintaining high production efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of Formylated Bicyclol. These insights are derived directly from the experimental data and technical specifications provided in patent CN112250658B, offering clarity on reaction parameters, purity profiles, and regulatory relevance. Understanding these details is essential for R&D teams planning to implement this standard in their analytical workflows.
Q: Why is synthesizing Formylated Bicyclol critical for Bicyclol API manufacturing?
A: Formylated Bicyclol is a specific process-related impurity found in Bicyclol crude products. Having a synthesized, high-purity standard allows manufacturers to accurately quantify this impurity using HPLC, ensuring the final API meets strict pharmacopoeia safety limits.
Q: What are the key reaction conditions for this Vilsmeier-Haack formylation?
A: The process requires strict temperature control: the addition of phosphorus oxychloride must be kept between 0-10°C to prevent exothermic runaway, while the subsequent reaction phase is heated to 80-100°C to drive the conversion to completion.
Q: How does this synthetic route improve supply chain reliability for reference standards?
A: Previously, this impurity could only be isolated in trace amounts from crude API batches. This directed synthesis allows for the production of gram-to-kilogram quantities on demand, eliminating the bottleneck of sourcing rare natural impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Formylated Bicyclol Supplier
The successful implementation of advanced impurity control strategies requires a partner with deep technical expertise and robust manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of the fine chemical industry, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which ensure that every batch of chemical intermediate meets the exacting standards required by global pharmaceutical regulators. We understand the critical nature of reference standards in ensuring drug safety and are equipped to deliver high-purity Formylated Bicyclol that aligns perfectly with the synthetic routes described in recent intellectual property.
We invite procurement leaders and technical directors to collaborate with us to optimize their supply chains for hepatoprotective drug manufacturing. By leveraging our specialized synthesis capabilities, your organization can secure a stable source of critical impurity standards while achieving substantial cost savings. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our solutions can enhance your quality control infrastructure and support your long-term commercial goals.
