Advanced Rhodium-Catalyzed Synthesis of Naphthol-Based Sulfones for Commercial Pharmaceutical Manufacturing
Introduction to Next-Generation Sulfone Synthesis Technology
The landscape of organic sulfur chemistry is undergoing a significant transformation, driven by the demand for more efficient and sustainable synthetic routes for high-value intermediates. Patent CN111302986B introduces a groundbreaking preparation method for naphthol-based sulfone compounds, addressing critical bottlenecks in the synthesis of sterically hindered molecular architectures. This technology leverages a sophisticated rhodium-catalyzed cyclization strategy to construct complex naphthyl sulfone scaffolds in a single operational step. For R&D directors and procurement specialists in the pharmaceutical sector, this represents a pivotal shift away from multi-step, low-yielding traditional processes towards a streamlined, high-controllability manufacturing paradigm. The ability to rapidly access these difficult-to-synthesize structures opens new avenues for drug discovery and process optimization, particularly for candidates requiring robust sulfone linkages within a naphthol framework.
The core innovation lies in the synergistic combination of a specific rhodium catalyst system with sulfur ylide chemistry. By utilizing bis(hexafluoroantimonic acid) triacetonitrile (pentamethylcyclopentadienyl) rhodium(III) alongside Lewis acid additives, the method achieves remarkable reaction controllability. This technical advancement ensures that the synthesis of large-steric-hindrance naphthyl sulfone compounds, which were previously accessible only through cumbersome pathways, can now be accomplished with enhanced precision. The patent data underscores the versatility of this approach, accommodating a wide range of substituents including halogens, alkyl groups, and alkoxy groups, thereby providing a robust platform for generating diverse libraries of pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of aryl cycloalkyl sulfones and related naphthol derivatives has relied heavily on the oxidation of thioethers or the dehydration of sodium benzene sulfinate with alcohols. These conventional pathways are fraught with significant operational challenges that hinder their applicability in modern, large-scale manufacturing. A primary drawback is the stringent requirement for specialized reaction substrates that often lack commercial availability, necessitating additional synthetic steps to prepare the starting materials. Furthermore, traditional methods such as direct sulfonylation of aromatic rings catalyzed by strong acids or reactions involving organolithium and Grignard reagents typically demand rigorous exclusion of air and moisture. This sensitivity not only complicates the operational procedure but also limits the compatibility with other functional groups present in complex drug molecules, often leading to side reactions and reduced overall purity.
Additionally, the use of strong oxidants in thioether oxidation routes poses safety hazards and environmental concerns due to the generation of hazardous waste streams. The lack of regioselectivity in some direct sulfonation methods can result in isomeric mixtures that are difficult and costly to separate, further eroding the economic viability of these processes. For supply chain managers, these inefficiencies translate into longer lead times, higher raw material costs, and increased risk of batch-to-batch variability. The inability to efficiently synthesize sterically congested sulfone structures has long been a barrier in the development of certain bioactive compounds, forcing chemists to settle for suboptimal analogs or invest excessive resources in process development.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN111302986B offers a direct, one-step catalytic cyclization route that fundamentally simplifies the synthesis of naphthol-based sulfones. This novel approach utilizes sulfonyl aryl acetylene compounds and sulfur ylide compounds as readily accessible building blocks, eliminating the need for pre-functionalized or sensitive precursors. 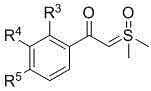
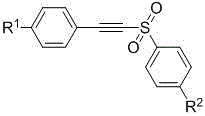 The reaction proceeds under mild thermal conditions, typically between 60°C and 100°C, in common organic solvents such as toluene, dioxane, or acetonitrile. This shift to milder conditions significantly enhances process safety and reduces energy consumption compared to high-temperature or cryogenic traditional methods.
The reaction proceeds under mild thermal conditions, typically between 60°C and 100°C, in common organic solvents such as toluene, dioxane, or acetonitrile. This shift to milder conditions significantly enhances process safety and reduces energy consumption compared to high-temperature or cryogenic traditional methods.
The outcome of this streamlined process is the direct formation of the target naphthol-based sulfone scaffold with high structural fidelity. 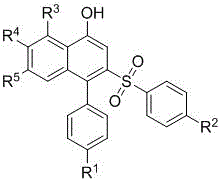 The method demonstrates exceptional tolerance for various substituents, allowing for the introduction of halogens, methyl, methoxy, and aryl groups without compromising the reaction efficiency. This broad substrate scope is crucial for medicinal chemists who need to explore structure-activity relationships (SAR) rapidly. By consolidating what was previously a multi-step sequence into a single catalytic event, this technology drastically reduces the number of unit operations, minimizes solvent usage, and accelerates the timeline from laboratory synthesis to pilot-scale production, offering a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing.
The method demonstrates exceptional tolerance for various substituents, allowing for the introduction of halogens, methyl, methoxy, and aryl groups without compromising the reaction efficiency. This broad substrate scope is crucial for medicinal chemists who need to explore structure-activity relationships (SAR) rapidly. By consolidating what was previously a multi-step sequence into a single catalytic event, this technology drastically reduces the number of unit operations, minimizes solvent usage, and accelerates the timeline from laboratory synthesis to pilot-scale production, offering a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Rhodium-Catalyzed Cyclization
The efficacy of this synthesis hinges on the unique reactivity of the pentamethylcyclopentadienyl rhodium(III) catalyst, specifically [RhCp*(CH3CN)3](SbF6)2. This catalyst is renowned for its ability to activate inert C-H and C-C bonds, a capability that is central to the cyclization mechanism described in the patent. In the presence of the sulfur ylide and the sulfonyl alkyne, the rhodium center facilitates a cascade of bond-forming events that construct the naphthalene core while simultaneously installing the sulfone moiety. The mechanism likely involves the initial coordination of the alkyne to the metal center, followed by C-H activation of the aromatic ring or insertion into the ylide carbon-sulfur bond. This precise orchestration of bond breaking and forming allows for the construction of complex polycyclic systems that are thermodynamically stable yet kinetically difficult to access via ionic pathways.
Furthermore, the inclusion of Lewis acids such as zinc trifluoromethanesulfonate, zinc acetate, or pivalic acid plays a pivotal role in modulating the reaction environment and enhancing yield. These additives are believed to activate the sulfur ylide or stabilize key intermediates within the catalytic cycle, thereby lowering the activation energy barrier for the cyclization step. The patent data indicates that zinc triflate, in particular, exhibits extremely high catalytic activity and offers the potential for recovery and regeneration, which is a significant advantage for sustainable chemistry. The careful optimization of the molar ratios between the sulfonyl aryl acetylene, sulfur ylide, Lewis acid, and rhodium catalyst ensures maximum atom economy. For instance, maintaining a specific ratio of 1.0:1.5:0.2 for the substrate, ylide, and Lewis acid respectively has been shown to maximize product yield while minimizing raw material waste, demonstrating a deep understanding of the underlying kinetic and thermodynamic parameters governing this transformation.
How to Synthesize Naphthol-Based Sulfone Compounds Efficiently
Implementing this advanced synthesis route requires adherence to specific operational protocols to ensure reproducibility and high purity. The process is designed to be robust, utilizing standard laboratory and plant equipment such as Schlenk tubes for small-scale optimization and stirred tank reactors for larger batches. The reaction is conducted under an inert nitrogen atmosphere to protect the catalyst and reagents from oxidative degradation, although the tolerance to minor impurities is higher than in traditional organometallic chemistry. Following the reaction period of 13 to 24 hours, the workup procedure is straightforward, involving solvent removal and purification via silica gel chromatography. This simplicity in downstream processing is a key factor in the technology's commercial appeal, as it reduces the burden on purification teams and shortens the overall production cycle time. Detailed standardized synthesis steps are provided in the guide below.
- Combine sulfonyl aryl acetylene, sulfur ylide, rhodium catalyst, Lewis acid, and organic solvent in a Schlenk tube under nitrogen.
- Heat the mixture in a preheated oil bath at 60-100°C for 13-24 hours to facilitate catalytic cyclization.
- Remove solvent via rotary evaporation and purify the crude product using silica gel column chromatography with ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rhodium-catalyzed technology translates into tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the supply chain for complex naphthol sulfone intermediates. By replacing multi-step sequences with a single catalytic step, manufacturers can significantly reduce the inventory of intermediate materials required, lower the working capital tied up in work-in-progress, and minimize the logistical complexity associated with managing multiple reaction stages. The use of commercially available and relatively inexpensive starting materials, such as substituted aryl acetylenes and sulfur ylides, ensures a stable and reliable supply base, mitigating the risk of raw material shortages that often plague specialty chemical manufacturing.
- Cost Reduction in Manufacturing: The elimination of multiple synthetic steps inherently drives down manufacturing costs by reducing labor, energy, and solvent consumption. Traditional routes often require distinct reaction vessels, separate purification stages, and extensive quality control checks for each intermediate, all of which accumulate significant overhead. In contrast, this one-pot cyclization consolidates these operations, leading to substantial cost savings. Moreover, the potential for recycling the Lewis acid additive, particularly zinc triflate, further enhances the economic profile of the process by reducing the consumption of auxiliary reagents. The mild reaction conditions also allow for the use of standard glass-lined or stainless steel reactors without the need for specialized cryogenic or high-pressure equipment, resulting in lower capital expenditure and maintenance costs.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system contributes to a more resilient supply chain. The reaction's tolerance to a variety of functional groups means that the same core process can be adapted to produce a wide array of derivatives without extensive re-optimization. This flexibility allows suppliers to respond quickly to changing customer demands for different analogs, reducing lead times for custom synthesis projects. Additionally, the use of common organic solvents like toluene and acetonitrile ensures that solvent supply is not a bottleneck, unlike processes relying on exotic or highly regulated solvents. The high controllability of the reaction minimizes the risk of batch failures, ensuring consistent delivery schedules and fostering stronger partnerships between suppliers and pharmaceutical clients.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns well with green chemistry principles. The atom economy is improved by the direct construction of the core structure, reducing the generation of by-products and waste streams associated with protecting group manipulations or oxidation steps found in older methods. The ability to scale this reaction from milligram to kilogram scales with minimal modification demonstrates its readiness for commercial production. The reduced waste profile simplifies effluent treatment and lowers the environmental compliance burden, which is increasingly critical for chemical manufacturers operating under strict regulatory frameworks. This sustainability angle not only reduces disposal costs but also enhances the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this naphthol sulfone synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic value of the method for industry stakeholders. Understanding these nuances is essential for R&D teams evaluating the technology for pipeline integration and for procurement professionals assessing supplier capabilities. The answers reflect the balance between high-performance chemistry and practical manufacturing constraints.
Q: What are the key advantages of this rhodium-catalyzed method over traditional sulfone synthesis?
A: Unlike traditional methods requiring harsh oxidation or sensitive organometallic reagents, this patent describes a direct one-step cyclization. It offers superior functional group tolerance, milder reaction conditions (60-100°C), and avoids the need for air-sensitive reagents, significantly simplifying the operational workflow.
Q: How does the choice of Lewis acid impact the reaction yield and efficiency?
A: The patent highlights that Lewis acids such as zinc trifluoromethanesulfonate or zinc acetate play a critical role in enhancing catalytic activity and yield. Specifically, zinc triflate is noted for its high activity and potential for recycling, which contributes to both economic efficiency and process sustainability.
Q: Is this synthesis method scalable for industrial production of API intermediates?
A: Yes, the method utilizes commercially available solvents like toluene and acetonitrile and operates at moderate temperatures compatible with standard industrial reactors. The one-step nature reduces unit operations, making it highly suitable for scaling up complex naphthol sulfone intermediates from kilogram to tonnage levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthol-Based Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the rhodium-catalyzed synthesis described in CN111302986B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory methodology into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale operation is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and potency.
We invite global pharmaceutical and agrochemical companies to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Whether you require custom synthesis of novel naphthol sulfone derivatives or scale-up of existing intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize your supply chain and accelerate your time to market. Partner with us to secure a reliable, cost-effective, and high-quality supply of complex sulfone intermediates for your next-generation therapeutics.
