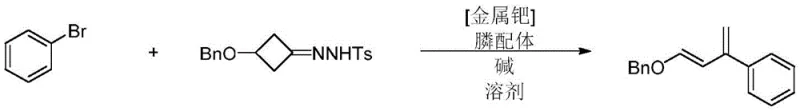
Advanced Synthesis of Electron-Rich Conjugated Dienes for Commercial Scale-Up and High-Purity Applications
The pharmaceutical and fine chemical industries constantly seek robust methodologies for constructing complex carbon skeletons, particularly six-membered rings which are ubiquitous in bioactive molecules. Patent CN113683493B introduces a significant advancement in this domain by disclosing a novel preparation method for electron-rich conjugated diene compounds. These compounds serve as critical precursors for [4+2] cyclization reactions, enabling the efficient synthesis of anthraquinone antibiotics and other high-value scaffolds. Unlike traditional approaches that struggle with stereoselectivity and cumbersome purification, this invention leverages a palladium-catalyzed system to deliver E-type 1,3-butadienes with exceptional structural uniformity. The technology addresses long-standing challenges in organic synthesis by utilizing accessible starting materials like aryl halides and cyclobutanone derivatives, thereby streamlining the production workflow. For R&D directors and procurement specialists, this represents a pivotal shift towards more predictable and scalable manufacturing processes for complex intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of electron-rich conjugated dienes has relied heavily on ruthenium-catalyzed intermolecular metathesis of enol ethers and alkynes or phosphorus ylide reactions. The ruthenium-catalyzed pathway often suffers from poor stereoselectivity, resulting in mixtures of Z and E isomers that require energy-intensive separation techniques to isolate the desired product. Furthermore, the ylide reaction route necessitates the generation of phosphorus ylides as intermediates, which subsequently produce stoichiometric amounts of triphenylphosphine oxide during the reaction with enones. This byproduct is notoriously difficult to remove completely, leading to complicated post-processing steps that increase both operational time and waste disposal costs. Additionally, achieving high stereo-purity in these conventional methods frequently demands cryogenic conditions, which escalates energy consumption and limits the feasibility of large-scale commercial production in standard reactor setups.
The Novel Approach
In stark contrast, the methodology outlined in CN113683493B utilizes a palladium-catalyzed cross-coupling strategy that operates under significantly milder conditions, typically around 90°C in a nitrogen atmosphere. This novel approach allows for the direct coupling of N-(3-(benzyloxy)cyclobutyl)-4-methylbenzenesulfonylhydrazone with various aryl halides, or alternatively, a one-pot synthesis starting from 3-(benzyloxy)cyclobutan-1-one. The process eliminates the formation of hard-to-remove phosphine oxides and avoids the use of expensive ruthenium catalysts, thereby simplifying the purification protocol to standard silica gel chromatography. By employing specific phosphine ligands such as diphenylcyclohexylphosphine, the reaction achieves high yields and excellent functional group tolerance, accommodating substrates with electron-withdrawing or electron-donating groups alike. This operational simplicity translates directly into reduced manufacturing complexity and enhanced safety profiles for industrial applications.
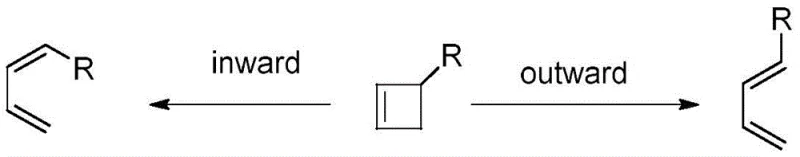
The mechanistic superiority of this new route lies in its ability to control the stereochemical outcome through the thermal ring-opening isomerization of a four-membered ring intermediate. As illustrated in the patent data, the cyclobutane derivative undergoes a conrotatory ring opening where the electronic nature of the substituent at the 3-position dictates the rotational preference. Specifically, the presence of electron-donating groups increases the preference for outward rotation, which kinetically favors the formation of the thermodynamically stable E-isomer over the Z-isomer. This intrinsic stereoselectivity removes the burden of downstream isomer separation, ensuring that the final product stream consists of a single, well-defined chemical entity. For process chemists, understanding this electronic effect is crucial for optimizing reaction parameters and predicting outcomes when scaling up to multi-kilogram batches for clinical or commercial supply.
Beyond the primary synthesis, the utility of these electron-rich dienes extends to their role as highly reactive dienophiles in Diels-Alder cycloadditions. The patent demonstrates that the generated E-type 1,3-butadienes react efficiently with alkenes and alkynes to construct six-membered ring systems with yields exceeding 85% in model reactions. This high reactivity is essential for building complex molecular architectures found in natural products and pharmaceutical agents, such as anthraquinone antibiotics. The ability to generate these dienes with high purity ensures that subsequent cyclization steps proceed without interference from impurities that could poison catalysts or lead to side reactions. Consequently, this technology not only solves the synthesis problem of the diene itself but also enhances the efficiency of the entire synthetic sequence leading to the final active pharmaceutical ingredient.
How to Synthesize Electron-Rich Conjugated Dienes Efficiently
To implement this advanced synthesis route in a laboratory or pilot plant setting, operators must adhere to a precise sequence of reagent addition and environmental controls to maximize yield and safety. The process begins with the careful preparation of the reaction mixture under an inert atmosphere to prevent catalyst deactivation by oxygen or moisture. Detailed standardized operating procedures regarding temperature ramping, stirring rates, and quenching protocols are essential to reproduce the high stereoselectivity reported in the patent documentation consistently. The following guide outlines the critical phases of the synthesis, emphasizing the importance of reagent quality and process monitoring to ensure the production of high-purity E-type dienes suitable for sensitive downstream applications.
- Mix N-(3-(benzyloxy)cyclobutyl)-4-methylbenzenesulfonylhydrazone with a base such as cesium carbonate and a palladium catalyst in a suitable vessel.
- Add phosphine ligand, aryl halide, and solvent like 1,4-dioxane, then react under nitrogen at 90°C with magnetic stirring.
- Cool the mixture, filter through celite, and purify via silica gel chromatography to isolate the E-type electron-rich conjugated diene.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, adopting this palladium-catalyzed methodology offers substantial benefits in terms of cost structure and supply chain resilience compared to legacy technologies. The elimination of ruthenium catalysts, which are subject to significant price volatility and supply constraints due to their status as precious metals, immediately reduces the raw material cost baseline for the synthesis. Furthermore, the simplified workup procedure, which avoids the generation of massive amounts of triphenylphosphine oxide waste, lowers the operational expenditure associated with solvent usage, waste treatment, and labor hours required for purification. These efficiencies compound when scaling from gram-scale laboratory experiments to tonnage-level commercial production, where even minor improvements in step economy translate into significant financial savings and reduced environmental footprint.
- Cost Reduction in Manufacturing: The transition away from expensive ruthenium-based catalysts to more abundant palladium systems, combined with the avoidance of stoichiometric phosphine oxide byproducts, drastically simplifies the material balance of the process. This reduction in hazardous waste generation lowers the cost of waste disposal and solvent recovery, while the milder reaction conditions reduce energy consumption for heating and cooling. Additionally, the high selectivity minimizes the loss of valuable starting materials into unwanted isomeric byproducts, thereby improving the overall atom economy and reducing the cost per kilogram of the final active intermediate.
- Enhanced Supply Chain Reliability: The reliance on readily available aryl halides and cyclobutanone derivatives as starting materials ensures a robust and diversified supply base that is less susceptible to single-source bottlenecks. Unlike specialized reagents required for ylide chemistry, these commoditized chemicals are produced by multiple global suppliers, providing procurement teams with greater negotiating power and security of supply. The operational simplicity of the reaction, which does not require extreme cryogenic infrastructure, also means that production can be flexibly shifted between different manufacturing sites without extensive capital investment in specialized equipment.
- Scalability and Environmental Compliance: The process operates at moderate temperatures around 90°C in standard solvents like 1,4-dioxane, making it highly amenable to scale-up in existing stainless steel reactors without the need for exotic metallurgy or pressure vessels. The reduced generation of solid waste and the absence of heavy metal contaminants in the final product streamline the regulatory compliance process for pharmaceutical filings. This environmental compatibility aligns with modern green chemistry principles, facilitating easier approval from environmental health and safety departments and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. They are derived from a detailed analysis of the experimental data and beneficial effects reported in CN113683493B, focusing on practical aspects that impact process development and sourcing decisions. Understanding these nuances is vital for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary stereoselectivity advantage of this new diene synthesis method?
A: The method described in patent CN113683493B offers excellent Z/E selectivity, specifically yielding single-structure E-type 1,3-butadienes. This is achieved through the thermal ring-opening isomerization of a four-membered ring intermediate, where electronic properties of substituents favor outward rotation, eliminating the need for difficult separation of isomers common in ruthenium-catalyzed metathesis.
Q: How does this process improve upon traditional ylide-based synthesis routes?
A: Traditional ylide reactions often generate triphenylphosphine oxide as a byproduct, which complicates post-reaction processing and purification significantly. In contrast, this palladium-catalyzed approach utilizes readily available aryl halides and cyclobutanone derivatives, simplifying the workup procedure to standard filtration and chromatography while operating under milder conditions without the need for extremely low temperatures.
Q: Can this synthesis method be adapted for diverse aromatic substrates?
A: Yes, the protocol demonstrates broad substrate universality and functional group compatibility. It successfully accommodates aryl halides substituted with electron-deficient groups, electron-rich groups, heterocyclic rings, and naphthalene rings. This flexibility allows for the synthesis of a wide library of diene intermediates suitable for subsequent Diels-Alder cyclizations in drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Electron-Rich Conjugated Diene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating drug development timelines and ensuring commercial success. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of electron-rich conjugated dienes meets the exacting standards required for GMP manufacturing. Our commitment to technical excellence allows us to navigate the complexities of palladium catalysis and stereoselective ring-opening reactions with precision, delivering consistent results for our global partners.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this advanced synthetic technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your long-term production goals and enhance your competitive advantage in the marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
