Advanced Catalytic Hydrogenation for 1-Cyclohexene-1,2-Dicarboximide Production and Commercial Scale-Up
Advanced Catalytic Hydrogenation for 1-Cyclohexene-1,2-Dicarboximide Production and Commercial Scale-Up
The chemical landscape for high-value intermediates is constantly evolving, driven by the need for cleaner, more efficient synthetic pathways that can meet the rigorous demands of the pharmaceutical and advanced materials sectors. A significant breakthrough in this domain is detailed in patent CN108689910B, which discloses a novel synthesis method for 1-cyclohexene-1,2-dicarboximide, also known as tetrahydrophthalimide. This compound serves as a critical building block in organic synthesis, finding extensive applications in biomedicine, the modification of composite materials, and the production of new unsaturated resin materials. The traditional manufacturing bottlenecks associated with this molecule have long plagued supply chains, but this new catalytic approach offers a transformative solution by utilizing phthalimide and hydrogen as raw materials under specific palladium-carbon catalysis.
The core innovation lies in the ability to achieve high selectivity and high conversion rates while effectively avoiding the notorious side reactions that typically plague benzene ring hydrogenation. By optimizing process conditions, specifically through the use of a 5% palladium-carbon catalyst with a 60% dry basis mass fraction, the invention realizes a reaction conversion rate exceeding 93%. Furthermore, the downstream processing is remarkably simplified; the reaction solution requires only simple distillation and crystallization filtration to yield a product with a mass fraction of greater than 98%. For global procurement teams seeking a reliable pharmaceutical intermediate supplier, this technological leap represents a shift towards more robust and cost-effective sourcing strategies.
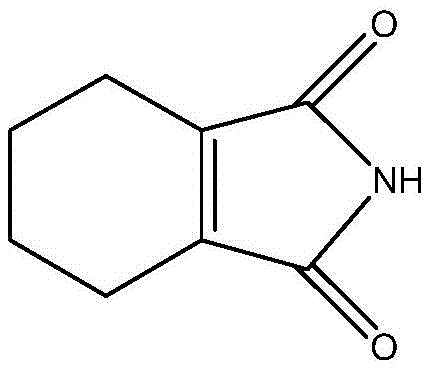
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 1-cyclohexene-1,2-dicarboximide has been fraught with inefficiencies and technical hurdles that drive up costs and compromise quality. The established literature describes a convoluted multi-step pathway beginning with the conjugate addition of maleic anhydride and 1,4-butadiene to form 1,2,3,6-tetrahydrophthalic anhydride. This intermediate must then undergo a harsh isomerization reaction at high temperatures (often around 190°C for 8 hours) catalyzed by phosphorus pentoxide to yield 3,4,5,6-tetrahydrophthalic anhydride. Finally, an amidation reaction with ammonia water or urea is required to produce the target imide. This legacy route is not only operationally complex but also suffers from inherently low yields at each stage, necessitating purification treatments between steps which further erodes overall productivity.
Moreover, the high-temperature conditions required for isomerization and amidation create severe quality control issues. The extreme thermal stress inevitably leads to the generation of substantial amounts of polymeric byproducts, often described as 'jelly,' resulting from double bond disconnection and repolymerization. These impurities are notoriously difficult to separate, leading to final products with compromised quality that are unsuitable for sensitive applications in biology and materials science. The total yield of this traditional process is low, and the production efficiency is insufficient to meet the growing demand for high-purity intermediates in the unsaturated resin and biomedical industries.
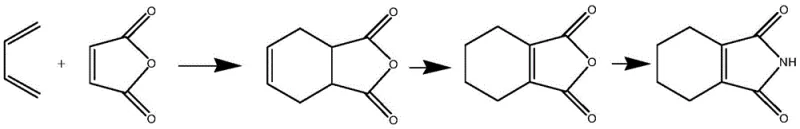
The Novel Approach
In stark contrast to the cumbersome traditional methods, the novel approach disclosed in the patent streamlines the entire synthesis into a single, elegant catalytic hydrogenation step. By starting directly from phthalimide and reacting it with hydrogen in the presence of a specialized catalyst, the process bypasses the need for Diels-Alder adducts and high-temperature isomerization entirely. This direct route fundamentally alters the economic and technical feasibility of production, offering a pathway that is both safer and more controllable. The reaction is conducted in N,N-dimethylformamide (DMF), a solvent choice that plays a critical role in moderating hydrogen solubility and stabilizing the reaction intermediate.
The brilliance of this new method lies in its ability to suppress the polymerization side reactions that characterize the old process. By carefully controlling the reaction temperature between 70°C and 150°C and utilizing a specific palladium-carbon catalyst formulation, the synthesis achieves high selectivity for the desired 1-cyclohexene structure. The result is a clean reaction profile that allows for simple downstream processing—evaporating a portion of the solvent, cooling, and filtering yields a high-quality product without the need for complex chromatographic separations. This represents a paradigm shift for cost reduction in fine chemical manufacturing, as it drastically reduces energy consumption, waste generation, and operational time.
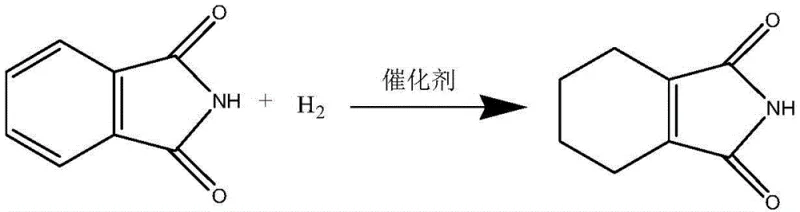
Mechanistic Insights into Pd/C-Catalyzed Selective Hydrogenation
To fully appreciate the technical sophistication of this synthesis, one must delve into the mechanistic nuances of selective benzene ring hydrogenation. The transformation of phthalimide to 1-cyclohexene-1,2-dicarboximide is a classic example of partial hydrogenation, where the goal is to stop the reaction at the cyclohexene stage rather than proceeding to the fully saturated cyclohexane derivative. Classical organic chemistry theory posits that benzene ring hydrogenation proceeds through adsorption, chemical conversion, and desorption stages. The challenge is to promote the desorption of the cyclohexene-structured intermediate before it can undergo further hydrogenation or isomerization. In this specific system, two primary side reactions compete with the desired pathway: hydroisomerization to cis-1,2,3,6-tetrahydrophthalimide and over-hydrogenation to cis-cyclohexyl-1,2-dicarboximide.
The selection of the catalyst is paramount in navigating these competing pathways. While noble metals like rhodium and ruthenium are active, they are often too expensive or difficult to source for large-scale applications. Raney nickel, though cheaper, suffers from low activity, requires large loading amounts, and poses significant safety risks due to its pyrophoric nature. The patent identifies 5% palladium-on-carbon (with 60% dry basis) as the optimal catalyst. This specific formulation provides a hydrogenation capacity that is sufficiently active to reduce the aromatic ring but weak enough to prevent immediate over-reduction. Furthermore, the choice of DMF as a solvent is strategic; its low solubility for hydrogen helps to limit the availability of hydrogen at the catalyst surface, effectively 'starving' the reaction just enough to favor the accumulation of the cyclohexene intermediate over the fully saturated product.
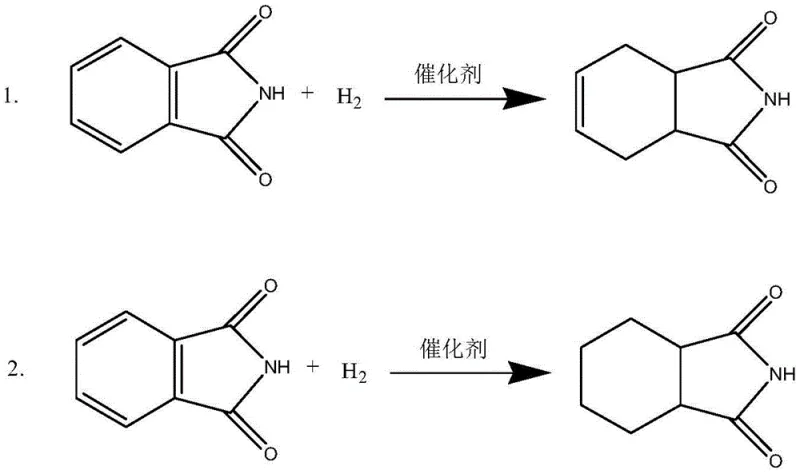
Controlling the reaction parameters is equally critical for managing the impurity profile. The patent data indicates that the reaction temperature and time dictate the ratio of the 1,2,3,6-isomer versus the desired 3,4,5,6-isomer (1-cyclohexene-1,2-dicarboximide), as these two forms are interconvertible under hydrogenation conditions. By maintaining the temperature between 70°C and 150°C and extending the reaction time to 70-80 hours, the process allows for thermodynamic equilibration that favors the desired product configuration. The mechanism relies on suppressing the re-adsorption of the cyclohexene product onto the catalyst surface. By optimizing the catalyst model and loading (3%-10% of phthalimide mass), the process ensures that once the cyclohexene structure is formed, it desorbs rapidly into the bulk solution, thereby escaping further reduction. This precise control over the catalytic cycle is what enables the achievement of >98% purity, a specification that is vital for high-purity API intermediate applications.
How to Synthesize 1-Cyclohexene-1,2-Dicarboximide Efficiently
Implementing this synthesis on an industrial scale requires strict adherence to the optimized parameters regarding pressure cycling and temperature control. The process involves charging an autoclave with phthalimide, DMF, and the palladium catalyst, followed by a series of hydrogen pressurization and depressurization cycles to ensure complete conversion while managing heat release. The reaction is maintained for an extended period (70-80 hours) to ensure high conversion, after which the catalyst is filtered off and the product is isolated via solvent distillation and crystallization. This standardized approach minimizes variability and ensures consistent batch-to-batch quality.
- Charge a stainless steel autoclave with phthalimide, N,N-dimethylformamide (DMF) solvent, and a 5% palladium-on-carbon catalyst (60% dry basis).
- Replace air with nitrogen, heat to 70-150°C, and introduce hydrogen gas in cycles, maintaining pressure until uptake ceases over a period of 70-80 hours.
- Filter the catalyst, concentrate the filtrate under reduced pressure, and crystallize the product to obtain white flaky crystals with >98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis route offers compelling strategic advantages that extend far beyond simple chemistry. The elimination of the high-temperature isomerization step and the removal of the Diels-Alder precursor requirement fundamentally reshape the cost structure of production. By simplifying the workflow from a multi-step sequence to a single catalytic reaction, manufacturers can significantly reduce capital expenditure on complex reactor setups and lower the operational overhead associated with intermediate handling and purification. This streamlining translates directly into a more competitive pricing structure for the final material, making it an attractive option for cost-sensitive projects in the polymer and pharmaceutical sectors.
- Cost Reduction in Manufacturing: The new process eliminates the need for expensive and hazardous reagents like phosphorus pentoxide and avoids the energy-intensive heating cycles required for isomerization. By removing these costly inputs and the associated waste disposal fees, the overall manufacturing cost is substantially decreased. Furthermore, the ability to recycle the palladium-carbon catalyst, as demonstrated in the patent examples, adds another layer of economic efficiency, reducing the dependency on fresh precious metal inputs for every batch.
- Enhanced Supply Chain Reliability: Sourcing raw materials is a critical vulnerability in any supply chain. This method relies on phthalimide and hydrogen, which are commodity chemicals available from a wide range of global suppliers, unlike the specialized anhydrides required in the traditional route. This diversification of the raw material base mitigates the risk of supply disruptions and price volatility. Additionally, the simplified process flow reduces the lead time for production, allowing for faster response to market demand fluctuations and ensuring a more consistent supply of high-purity 1-cyclohexene-1,2-dicarboximide.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is vastly superior. It operates at lower temperatures and avoids the generation of difficult-to-separate polymeric 'jelly' byproducts, which simplifies wastewater treatment and solid waste management. The process produces a cleaner effluent stream and reduces the carbon footprint associated with high-temperature processing. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, making the commercial scale-up of complex heterocycles more viable in regions with stringent environmental regulations.
Frequently Asked Questions (FAQ)
Understanding the technical specifics of this synthesis is crucial for stakeholders evaluating its adoption. The following questions address common concerns regarding catalyst life, product specifications, and process safety, drawing directly from the experimental data provided in the patent documentation. These insights are intended to clarify the operational realities of scaling this technology.
Q: How does the new hydrogenation method improve upon traditional synthesis routes?
A: The novel method utilizes direct selective hydrogenation of phthalimide, bypassing the complex multi-step Diels-Alder and high-temperature isomerization processes required in conventional synthesis. This eliminates the formation of polymeric byproducts ('jelly') and significantly simplifies purification.
Q: What is the expected purity and yield of the final product?
A: According to patent data, the process achieves a reaction conversion rate of over 93% and yields a final product with a mass fraction exceeding 98% after simple distillation and crystallization, with melting points ranging between 170°C and 172°C.
Q: Can the palladium-carbon catalyst be recycled in this process?
A: Yes, the patent demonstrates that the filtered catalyst cake can be dried and reused for subsequent batches. However, efficiency may decrease after multiple cycles, requiring monitoring of product content and melting range.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Cyclohexene-1,2-Dicarboximide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We understand the critical nature of stringent purity specifications in the pharmaceutical and electronic materials sectors, and our rigorous QC labs are equipped to verify every batch against the highest international standards, guaranteeing the consistency required for your downstream applications.
We invite you to explore how this innovative hydrogenation technology can optimize your supply chain and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemical solution available.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
