Revolutionizing Indole Sulfuration with Triphosgene for Commercial Scale Production
Revolutionizing Indole Sulfuration with Triphosgene for Commercial Scale Production
The introduction of fluorine-containing functional groups, particularly the trifluoromethylthio (-SCF3) moiety, has become a cornerstone strategy in modern medicinal chemistry and agrochemical design due to its profound impact on metabolic stability and lipophilicity. As highlighted in patent CN108586312B, a groundbreaking green sulfuration method has been developed that utilizes triphosgene as a reducing agent to efficiently functionalize indole compounds. This technology addresses critical bottlenecks in the synthesis of high-value intermediates used in blockbuster veterinary drugs like Toltrazuril and broad-spectrum insecticides such as Fipronil. 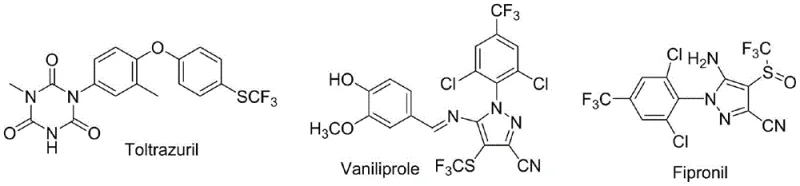 . For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, this patent represents a paradigm shift towards safer, more cost-effective manufacturing processes that eliminate the reliance on toxic reagents and expensive transition metal catalysts.
. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, this patent represents a paradigm shift towards safer, more cost-effective manufacturing processes that eliminate the reliance on toxic reagents and expensive transition metal catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct introduction of trifluoromethylthio radicals onto aromatic systems has been plagued by significant safety and economic challenges. Early methodologies relied heavily on electrophilic trifluoromethylsulfenyl chloride (CF3SCl), a reagent notorious for its extreme toxicity and low boiling point, which complicates handling and requires specialized containment infrastructure. Subsequent advancements, such as those by the Billard group using stable trifluoromethylthiolaniline, improved safety but often necessitated harsh conditions or exhibited limited substrate scope. Furthermore, strategies employing N-trifluoromethylthiophthalimide reagents, while effective, frequently require additives like NaCl whose precise mechanistic role remains ambiguous, introducing variability in batch-to-batch reproducibility. 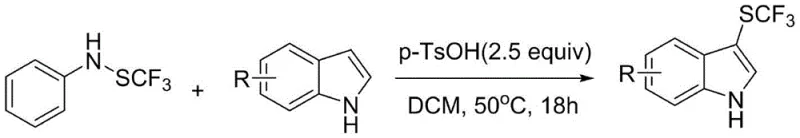 . Additionally, many existing protocols depend on phosphorus-based reducing agents or expensive electrophilic reagents that generate substantial hazardous waste, creating a heavy burden on environmental compliance and waste disposal costs for large-scale manufacturers.
. Additionally, many existing protocols depend on phosphorus-based reducing agents or expensive electrophilic reagents that generate substantial hazardous waste, creating a heavy burden on environmental compliance and waste disposal costs for large-scale manufacturers.
The Novel Approach
The innovative process disclosed in the patent data circumvents these legacy issues by employing triphosgene, a solid and manageable substitute for phosgene, as both a chlorinating and reducing agent. This method enables the in-situ generation of highly reactive electrophilic species from inexpensive sodium sulfinates, thereby avoiding the storage and handling of volatile, toxic sulfenyl chlorides. 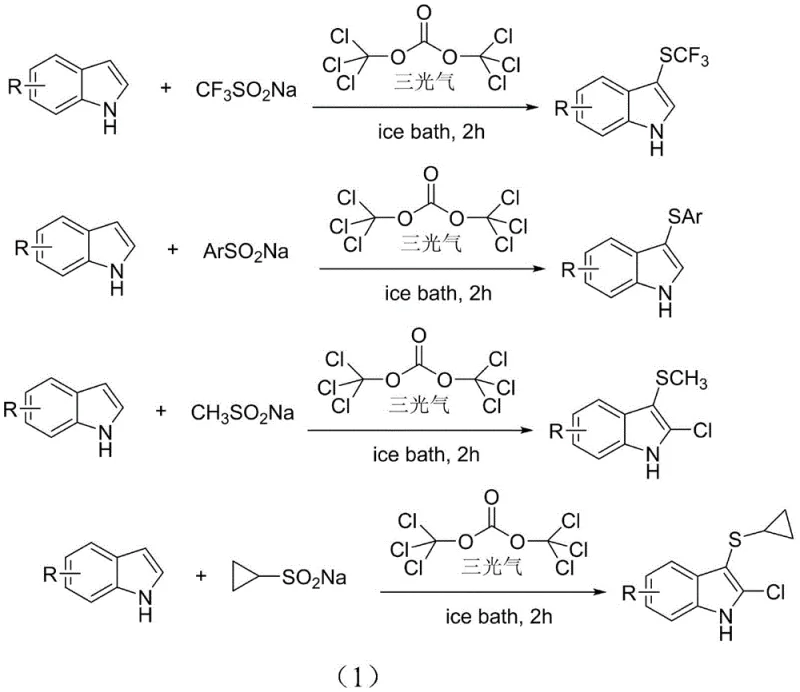 . By operating under mild ice-bath conditions in acetonitrile, this transition-metal-free protocol achieves remarkable regioselectivity at the C3 position of the indole ring. The elimination of transition metal catalysts not only simplifies the purification workflow by removing the need for rigorous metal scavenging steps but also ensures the final product meets stringent purity specifications required for pharmaceutical and agrochemical applications without the risk of heavy metal contamination.
. By operating under mild ice-bath conditions in acetonitrile, this transition-metal-free protocol achieves remarkable regioselectivity at the C3 position of the indole ring. The elimination of transition metal catalysts not only simplifies the purification workflow by removing the need for rigorous metal scavenging steps but also ensures the final product meets stringent purity specifications required for pharmaceutical and agrochemical applications without the risk of heavy metal contamination.
Mechanistic Insights into Triphosgene-Mediated Electrophilic Sulfuration
The core mechanistic advantage of this technology lies in the unique ability of triphosgene to activate sodium sulfinate salts under mild conditions. In the reaction system, triphosgene likely facilitates the conversion of sodium trifluoromethanesulfonate (CF3SO2Na) into an electrophilic trifluoromethylsulfenyl chloride (CF3SCl) intermediate in situ. This reactive species then undergoes electrophilic substitution with the electron-rich indole nucleus, preferentially attacking the C3 position due to its high nucleophilicity. Unlike radical-based approaches that often suffer from poor selectivity and side reactions, this electrophilic pathway ensures clean conversion with minimal byproduct formation. The absence of external oxidants or metal promoters further streamlines the reaction profile, reducing the complexity of the impurity spectrum and facilitating easier downstream processing for quality control teams.
From an impurity control perspective, the use of triphosgene offers distinct advantages over phosphine-based reduction systems. Traditional methods utilizing phosphites or triphenylphosphine generate stoichiometric amounts of phosphine oxides, which are notoriously difficult to separate from the target organic molecules and often require extensive chromatographic purification. In contrast, the byproducts of the triphosgene-mediated reaction are primarily gaseous or water-soluble species that can be easily removed during the aqueous workup phase. This inherent cleanliness of the reaction profile translates directly into higher isolated yields, reported between 61% and 94% across diverse substrates, and significantly reduces the consumption of silica gel and solvents during purification, aligning perfectly with green chemistry principles and cost-reduction goals.
How to Synthesize 3-Trifluoromethylthioindole Efficiently
The practical implementation of this synthesis route is designed for operational simplicity and scalability, making it ideal for contract development and manufacturing organizations. The standard procedure involves combining the indole substrate with sodium trifluoromethanesulfonate and triphosgene in acetonitrile, followed by stirring under controlled low-temperature conditions. 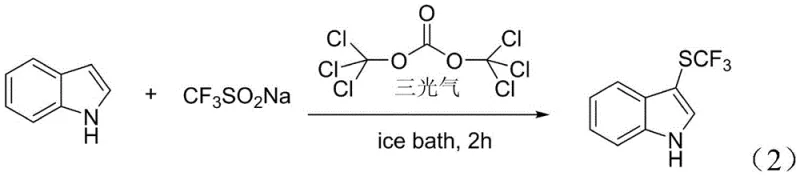 . Detailed standard operating procedures, including precise molar ratios and quenching protocols, are critical for maintaining safety and maximizing yield. The following guide outlines the generalized steps derived from the patent examples to assist process chemists in replicating this high-efficiency transformation in a pilot or production setting.
. Detailed standard operating procedures, including precise molar ratios and quenching protocols, are critical for maintaining safety and maximizing yield. The following guide outlines the generalized steps derived from the patent examples to assist process chemists in replicating this high-efficiency transformation in a pilot or production setting.
- Mix indole substrate, sodium trifluoromethanesulfonate, and triphosgene in acetonitrile solvent under an ice bath.
- Stir the reaction mixture for 2 hours while monitoring progress via TLC until completion.
- Quench with aqueous NaOH, extract with ethyl acetate, and purify the crude product using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this triphosgene-mediated sulfuration technology offers transformative benefits regarding cost structure and supply continuity. By replacing expensive, specialized fluorinating reagents with commodity chemicals like triphosgene and sodium sulfinates, the raw material cost base is drastically lowered. Furthermore, the elimination of transition metal catalysts removes a significant variable from the supply chain, mitigating risks associated with the price volatility of precious metals like palladium or copper. The simplified workup procedure, which avoids complex metal scavenging and extensive chromatography, leads to faster batch turnover times and reduced solvent consumption, directly impacting the overall cost of goods sold (COGS) for high-purity indole derivatives.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the substitution of costly electrophilic trifluoromethylthio reagents with inexpensive sodium sulfinate salts. Since the reaction does not require expensive ligands or transition metal catalysts, the input material costs are significantly minimized. Additionally, the avoidance of phosphorus-containing reducing agents eliminates the downstream costs associated with treating phosphorus-laden waste streams, resulting in substantial operational savings. The high yields achieved across a broad range of substrates further enhance the atom economy, ensuring that raw material investment is maximized in the final product output.
- Enhanced Supply Chain Reliability: Utilizing stable, solid reagents like triphosgene and sodium sulfinates enhances supply chain robustness compared to relying on volatile liquids or air-sensitive organometallic complexes. These starting materials are widely available from bulk chemical suppliers, reducing the risk of procurement bottlenecks. The mild reaction conditions, specifically the use of an ice bath rather than cryogenic cooling or high-pressure equipment, allow the process to be executed in standard glass-lined reactors found in most multipurpose chemical plants, ensuring that production capacity can be easily scaled up without requiring specialized infrastructure investments.
- Scalability and Environmental Compliance: The green nature of this synthesis aligns with increasingly stringent global environmental regulations. By avoiding the generation of heavy metal waste and toxic phosphine byproducts, the process simplifies environmental permitting and waste disposal logistics. The use of acetonitrile, a common and recoverable solvent, facilitates solvent recycling programs. This environmental compatibility not only reduces regulatory compliance costs but also strengthens the sustainability profile of the supply chain, a key metric for multinational corporations evaluating their tier-one chemical suppliers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green sulfuration technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on substrate scope, safety profiles, and purification requirements for potential partners evaluating this methodology for their own production pipelines.
Q: What are the advantages of using triphosgene over traditional phosphine reducing agents?
A: Triphosgene eliminates the environmental hazards associated with phosphorus-containing waste and avoids the use of expensive, odor-intensive thiophenols, significantly simplifying downstream purification and waste treatment.
Q: Can this method tolerate various functional groups on the indole ring?
A: Yes, the protocol demonstrates excellent compatibility with electron-donating groups like methoxy and methyl, as well as electron-withdrawing halogens such as chlorine and bromine, yielding products with 61-94% efficiency.
Q: Is transition metal catalysis required for this sulfuration process?
A: No, this novel approach operates under transition-metal-free conditions, which removes the risk of heavy metal contamination in the final API or agrochemical active ingredient.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethylthioindole Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced synthetic methodologies to deliver superior chemical solutions. Our technical team has extensively evaluated the triphosgene-mediated sulfuration pathway and confirmed its potential for robust commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of indole derivative meets the exacting standards required for agrochemical and pharmaceutical applications.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce your overall manufacturing expenditures.
