Scalable Biocatalytic Production of Optically Active Phenyl Glycidol Intermediates for Pharma
Scalable Biocatalytic Production of Optically Active Phenyl Glycidol Intermediates for Pharma
Introduction to Advanced Biocatalytic Epoxidation Technology
The pharmaceutical and fine chemical industries are continuously seeking robust manufacturing pathways that align with stringent regulatory standards while maximizing operational efficiency and environmental sustainability. Patent CN102337307A introduces a groundbreaking biocatalytic methodology for the preparation of optically active phenyl glycidol and its derivatives, which serve as critical building blocks for beta-blocker drugs and other bioactive molecules. This innovative approach utilizes genetically engineered Escherichia coli whole resting cells containing the styrene monooxygenase gene to drive the asymmetric epoxidation of allylic alcohol substrates with exceptional precision. By leveraging biological catalysis, this technology effectively circumvents the traditional reliance on hazardous chemical oxidants and toxic transition metal catalysts that have long plagued conventional synthetic routes. The result is a streamlined process that delivers high-purity chiral intermediates suitable for direct use in sensitive pharmaceutical applications without extensive purification burdens. Furthermore, the scalability of this fermentation-based system offers a reliable pharmaceutical intermediate supplier solution for companies aiming to secure their supply chains against volatile chemical markets. Implementing this green chemistry protocol allows manufacturers to significantly reduce their environmental footprint while maintaining the rigorous quality specifications demanded by global health authorities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The conventional chemical synthesis of optically active glycidol derivatives frequently relies on the Sharpless asymmetric epoxidation methodology, which, while effective, necessitates the use of stoichiometric amounts of titanium tetraisopropoxide and tert-butyl hydroperoxide. This dependency introduces significant challenges regarding the removal of toxic heavy metal residues from the final active pharmaceutical ingredients, thereby complicating the purification workflow and escalating the overall environmental footprint of the manufacturing process. Furthermore, alternative enzymatic resolution strategies utilizing lipases often suffer from inherent theoretical yield limitations, capping the maximum achievable output at fifty percent of the starting racemic material. These processes generate substantial quantities of unwanted enantiomeric waste that must be disposed of or recycled through energy-intensive methods, ultimately undermining the economic viability and sustainability goals of modern fine chemical production facilities. Consequently, the industry faces a persistent demand for innovative catalytic systems that can overcome these atom-economy barriers while maintaining rigorous stereochemical control over the formation of multiple chiral centers. Addressing these critical bottlenecks requires a paradigm shift towards whole-cell biocatalytic platforms that integrate enzyme stability with streamlined downstream processing capabilities for large-scale industrial applications. The accumulation of metal waste and organic solvent usage in traditional methods also poses increasing regulatory compliance risks for manufacturers operating under tightening environmental protection laws.
The Novel Approach
In stark contrast to legacy chemical methods, the novel biocatalytic route disclosed in the patent employs whole resting cells of E. coli expressing styrene monooxygenase to selectively epoxidize specific enantiomers of racemic allylic alcohols. This highly selective biological transformation allows for the conversion of exactly half of the input racemic substrate into the desired optically pure epoxy alcohol while leaving the complementary enantiomer intact as a recoverable chiral alcohol. The preservation of the unreacted isomer represents a significant advancement in atom economy, as this residual material can be isolated and utilized as a valuable synthetic intermediate rather than being discarded as waste. Moreover, the reaction proceeds in aqueous buffer systems under mild temperature and pressure conditions, eliminating the need for cryogenic cooling or anhydrous organic solvents that drive up energy consumption and safety risks. This gentle reaction environment ensures the integrity of sensitive functional groups on the substrate, reducing the formation of side products and simplifying the isolation of the target compound. The ability to produce two distinct high-value chiral intermediates from a single racemic starting material fundamentally transforms the cost structure of the synthesis, offering substantial cost savings in API manufacturing. Ultimately, this technology provides a sustainable and economically superior alternative for the commercial scale-up of complex chiral intermediates required by the global pharmaceutical sector.
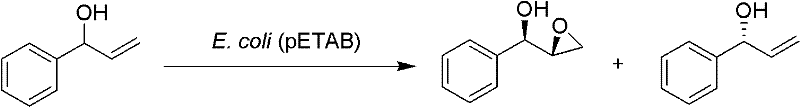
Mechanistic Insights into Styrene Monooxygenase Catalyzed Asymmetric Epoxidation
The core of this transformative technology lies in the exquisite stereoselectivity of the styrene monooxygenase enzyme system expressed within the recombinant bacterial host cells. This biocatalyst exhibits profound enantioselectivity towards the carbon-carbon double bond of the allylic alcohol substrate, distinguishing between the (S) and (R) enantiomers with high fidelity during the oxygen transfer process. The enzyme preferentially oxidizes the (S)-enantiomer of the starting material to form the (1R,2R)-epoxy product while exhibiting negligible catalytic activity towards the (R)-enantiomer, which remains largely unconverted in the reaction mixture. This kinetic resolution mechanism is further enhanced by the enzyme's diastereoselectivity, ensuring that the newly formed epoxide ring possesses the correct relative stereochemistry required for downstream drug synthesis. The whole-cell format provides a protective microenvironment for the enzyme, stabilizing its active conformation and extending its operational lifespan compared to isolated enzyme preparations. Understanding this mechanistic precision is crucial for R&D directors focused on purity and impurity profiles, as it guarantees a consistent stereochemical outcome batch after batch. The high specificity minimizes the generation of diastereomeric impurities that are notoriously difficult to separate, thereby enhancing the overall quality of the high-purity phenyl glycidol produced.
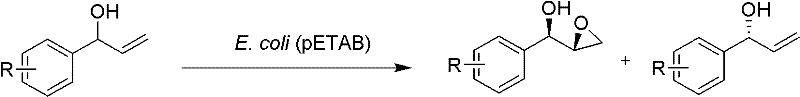
Impurity control is inherently built into the biological nature of this catalytic system, as the absence of transition metals precludes the formation of metal-complexed byproducts that often contaminate chemically catalyzed reactions. The aqueous reaction medium further suppresses non-enzymatic background reactions that could lead to racemization or polymerization of the sensitive epoxide product. By avoiding harsh chemical oxidants, the process prevents over-oxidation side reactions that typically degrade the yield and purity of the target molecule in traditional synthetic routes. The resulting product stream is therefore cleaner, requiring fewer purification steps such as chromatography or recrystallization to meet pharmacopeial standards. This inherent purity advantage translates directly into reduced processing time and lower solvent consumption during the workup phase, contributing to a more efficient manufacturing cycle. For quality assurance teams, this means a more robust and predictable impurity profile that simplifies regulatory filings and validation protocols. The combination of high enantiomeric excess and minimal chemical impurities makes this biocatalytic route particularly attractive for the synthesis of chiral drugs where stereochemical purity is a critical safety parameter.
How to Synthesize Optically Active Phenyl Glycidol Efficiently
Implementing this synthesis route begins with the cultivation of the engineered E. coli (pETAB) strain in a nutrient-rich medium supplemented with kanamycin to maintain plasmid stability throughout the fermentation process. Once the cell density reaches the optimal level, the expression of the styrene monooxygenase gene is induced by lowering the temperature, allowing the bacteria to accumulate high levels of the active biocatalyst before harvesting. The harvested wet cells are then resuspended in a buffered aqueous solution where the pH is carefully controlled to maximize enzyme activity and stability during the transformation phase. The racemic substrate is introduced into this suspension, either directly or dissolved in a minimal amount of co-solvent, initiating the asymmetric epoxidation reaction under mild agitation. Following the completion of the bioconversion, the cells are removed via centrifugation, and the product is extracted from the aqueous supernatant using organic solvents followed by standard drying and concentration techniques. Detailed standardized synthesis steps are provided below to ensure reproducibility and adherence to the patented protocol for optimal yields.
- Cultivate E. coli (pETAB) strains in TB medium with kanamycin, induce styAB gene expression at 20°C, and harvest wet cells via centrifugation.
- Suspend harvested cells in phosphate buffer (pH 5.5-8.0) and add racemic 1-phenyl-2-allyl alcohol substrate to initiate asymmetric epoxidation.
- After reaction completion, separate cells by centrifugation, extract product with ether, dry over sodium sulfate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this biocatalytic technology presents a compelling value proposition centered on cost optimization and supply security. The elimination of expensive transition metal catalysts and the associated scavenging agents significantly reduces the raw material costs and consumables budget required for each production batch. Additionally, the simplified workup procedure resulting from the cleaner reaction profile decreases the consumption of extraction solvents and silica gel, leading to further reductions in operational expenditures. The ability to recover and utilize the unreacted chiral alcohol isomer adds an additional revenue stream or cost offset, effectively improving the overall material balance of the process. From a supply chain perspective, the reliance on fermentation-derived catalysts ensures a consistent and renewable source of catalytic activity that is not subject to the geopolitical volatility often associated with mined metal resources. This stability enhances supply chain reliability and reduces the risk of production delays caused by raw material shortages or quality fluctuations. Furthermore, the green nature of the process aligns with corporate sustainability mandates, potentially qualifying the manufacturing site for environmental incentives and improving brand reputation among eco-conscious stakeholders.
- Cost Reduction in Manufacturing: The removal of heavy metal catalysts from the process flow eliminates the need for specialized resin columns or complex chemical treatments designed to scavenge trace metals from the final product. This simplification of the downstream processing stage results in significant labor and material savings, as fewer unit operations are required to achieve the necessary purity levels. The reduced solvent usage during extraction and purification further lowers the utility costs associated with solvent recovery and waste disposal systems. By maximizing the utility of the starting material through the recovery of the complementary enantiomer, the effective cost per kilogram of the target intermediate is drastically reduced compared to traditional resolution methods. These cumulative efficiencies contribute to a leaner manufacturing model that can withstand price pressures in the competitive pharmaceutical intermediate market. Ultimately, the process economics favor large-scale production where the fixed costs of fermentation infrastructure are amortized over high volumes of output.
- Enhanced Supply Chain Reliability: Biocatalytic processes rely on renewable feedstocks and genetically stable strains that can be stored and propagated indefinitely, ensuring a long-term supply of the critical catalytic component. Unlike chemical catalysts that may suffer from batch-to-batch variability or supply disruptions from single-source vendors, the biological catalyst can be produced in-house or sourced from multiple qualified fermentation facilities. The robustness of the whole-cell system against minor fluctuations in reaction conditions also reduces the likelihood of batch failures, thereby improving the predictability of delivery schedules. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, allowing downstream drug manufacturers to maintain tighter inventory controls and respond faster to market demand. The decentralized nature of fermentation technology also mitigates the risk of regional supply chain interruptions, providing a more resilient network for global chemical distribution. Procurement teams can thus negotiate contracts with greater confidence knowing that the production technology is inherently stable and scalable.
- Scalability and Environmental Compliance: The transition from laboratory shake flasks to industrial fermenters is a well-established practice in the biotechnology sector, making the scale-up of this epoxidation process straightforward and low-risk. The aqueous nature of the reaction mixture simplifies heat and mass transfer management in large vessels, preventing the hot spots and runaway reactions that can occur in exothermic chemical oxidations. Waste streams generated from this process are primarily biological and aqueous, which are easier and cheaper to treat in standard wastewater treatment plants compared to heavy metal-laden chemical waste. This environmental compatibility ensures compliance with increasingly stringent discharge regulations, avoiding potential fines and shutdowns that could disrupt supply continuity. The reduced carbon footprint of the biocatalytic route also supports corporate goals for carbon neutrality, making it a preferred choice for companies with aggressive sustainability targets. Overall, the process offers a scalable pathway to multi-ton production capacities without compromising on safety or environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented biocatalytic epoxidation technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to provide accurate guidance for potential adopters. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The information covers aspects ranging from stereochemical outcomes to process scalability and regulatory implications. We encourage technical teams to review these insights thoroughly to assess the alignment with their specific project requirements. Further detailed discussions can be arranged with our scientific experts to explore customization options for specific substrate derivatives.
Q: How does this biocatalytic method improve atom economy compared to lipase resolution?
A: Unlike lipase resolution which theoretically wastes 50% of the starting material, this styrene monooxygenase process converts one enantiomer to the epoxy product while leaving the other enantiomer intact and recoverable as a valuable chiral alcohol intermediate.
Q: Does this process eliminate the need for heavy metal scavengers?
A: Yes, the use of whole-cell E. coli biocatalysts completely avoids transition metal catalysts like titanium, thereby removing the costly and complex downstream steps required to scavenge metal residues from the final API intermediate.
Q: What is the enantiomeric excess achievable with this technology?
A: The patented method demonstrates exceptional stereocontrol, consistently achieving enantiomeric excess (ee) values greater than 99% for the target (1R,2R)-1-phenyl-2,3-glycidol derivatives across various substituted substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenyl Glycidol Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale innovation to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of chiral intermediates we produce. We understand the critical importance of consistency in the pharmaceutical supply chain and are committed to delivering high-purity phenyl glycidol that meets the exacting standards of global regulatory bodies. Our team of process chemists is ready to optimize the biocatalytic parameters to suit your specific throughput needs while maintaining the highest levels of safety and quality. Partnering with us means gaining access to a robust supply network that prioritizes reliability and technical excellence above all else. We are dedicated to supporting your drug development timelines with responsive service and transparent communication throughout the engagement.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project's unique requirements. Let us provide you with a Customized Cost-Saving Analysis that demonstrates the tangible economic benefits of switching to this green biocatalytic manufacturing route. Our experts are standing by to discuss how we can collaborate to enhance your supply chain resilience and reduce your overall production costs. Reach out today to initiate a conversation about securing a sustainable and high-quality supply of these critical chiral building blocks for your future success.
