Scalable Synthesis of Pyrazolopiperidine Derivatives for Next-Generation Antiparasitic Therapeutics
Introduction to Advanced Pyrazolopiperidine Synthesis
The pharmaceutical industry is constantly seeking more efficient pathways to access complex heterocyclic scaffolds, particularly those showing promise in treating neglected tropical diseases. Patent CN113264931B introduces a groundbreaking preparation method for 1,4,6,7-tetrahydro-5H-pyrazolo[4,3-c]pyridine derivatives, a core structure identified as a potent inhibitor of the PEX14-PEX5 protein-protein interaction. This interaction is critical for glucose transport in trypanosomes, making these compounds potential therapeutics for sleeping sickness. The disclosed methodology represents a significant leap forward in process chemistry, replacing traditional, wasteful coupling strategies with a streamlined, high-yielding sequence that begins with readily available N-Boc-4-piperidone. By leveraging modern organic synthesis techniques, this route not only enhances the purity profile of the final intermediate but also drastically simplifies the supply chain logistics for manufacturers aiming to produce these vital antiparasitic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of pyrazolopiperidine skeletons has relied heavily on the condensation of phenylhydrazines with piperidone derivatives, followed by harsh N-arylation steps. A prevalent technique involves the copper-catalyzed Ullmann reaction, which necessitates the use of iodoarenes as coupling partners. From a process development perspective, this conventional approach presents severe drawbacks, including the high cost and low atom economy associated with iodine-containing substrates. Furthermore, Ullmann couplings typically require elevated temperatures and stoichiometric amounts of copper catalysts, leading to significant challenges in removing heavy metal residues to meet stringent pharmaceutical purity standards. The requirement to pre-synthesize unstable phenylhydrazine intermediates also introduces safety hazards and additional unit operations, inflating both the production timeline and the overall cost of goods sold for the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the methodology outlined in patent CN113264931B circumvents these bottlenecks by employing a direct aromatic nucleophilic substitution (SNAr) strategy. Instead of relying on expensive iodoarenes and toxic copper catalysts, the process utilizes 2-fluorobenzonitrile, a commercially abundant and cost-effective electrophile. The synthesis initiates with a controlled Claisen condensation to install the necessary carbon framework, followed by a facile cyclization with hydrazine hydrate to forge the pyrazole ring. This strategic shift eliminates the need for transition metal catalysis in the C-N bond formation step, thereby reducing the impurity burden related to metal leaching. The result is a cleaner reaction profile that facilitates downstream purification, offering a distinct competitive advantage for contract development and manufacturing organizations (CDMOs) focused on delivering high-quality pharmaceutical intermediates.
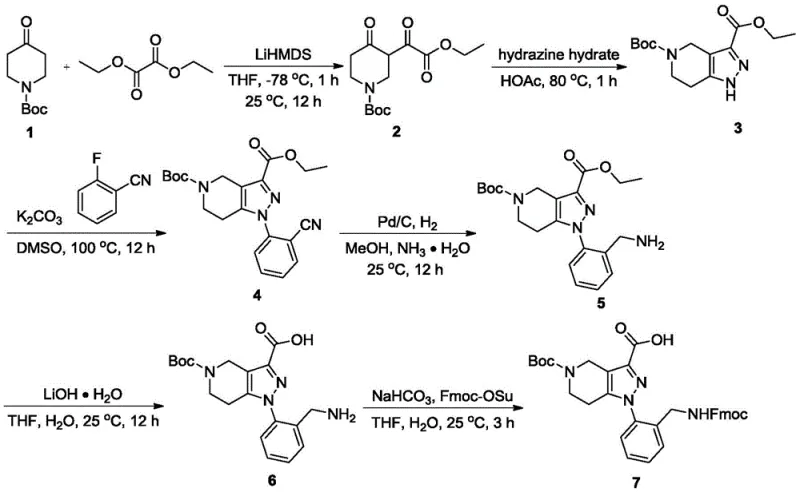
Mechanistic Insights into LiHMDS-Mediated Cyclization and SNAr
The success of this synthetic route hinges on the precise execution of the initial enolate chemistry and the subsequent heterocycle formation. The process begins with the deprotonation of N-Boc-4-piperidone using lithium hexamethyldisilazide (LiHMDS) at a cryogenic temperature of -78°C. This low-temperature condition is critical for generating the kinetic enolate selectively, preventing self-condensation or polymerization side reactions that could compromise the yield. Upon addition of diethyl oxalate, a Claisen-type condensation occurs, yielding a beta-keto ester intermediate with high fidelity. This intermediate then undergoes cyclocondensation with hydrazine hydrate in acetic acid at 80°C. The nucleophilic attack of hydrazine on the ketone and ester functionalities drives the formation of the pyrazolo[4,3-c]pyridine core, a rigid scaffold essential for the biological activity of the target molecule.
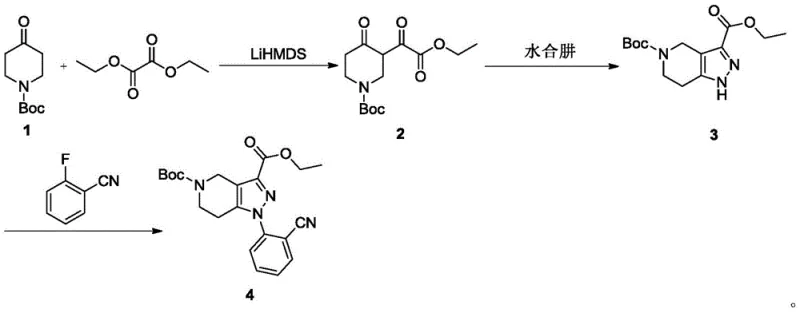
Following the core construction, the installation of the aryl side chain is achieved through a classic SNAr mechanism. The electron-deficient nature of the pyrazole nitrogen, combined with the activating effect of the adjacent nitrile group on the fluorobenzene ring, facilitates the displacement of the fluoride ion under mild basic conditions using potassium carbonate in DMSO. This step proceeds with exceptional efficiency, reportedly achieving yields as high as 96%. The absence of transition metals in this key bond-forming step ensures that the impurity profile remains simple, primarily consisting of unreacted starting materials which are easily removed during workup. For R&D directors, this mechanistic clarity translates to a robust process that is easier to validate and scale, minimizing the risk of batch failures due to catalyst poisoning or unpredictable side reactions.
How to Synthesize Pyrazolopiperidine Intermediate Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly temperature control during the lithiation step and solvent selection for the arylation. The patent provides a detailed roadmap that balances reaction kinetics with operational simplicity, making it highly suitable for technology transfer. The initial steps establish the heterocyclic core with high purity, while the subsequent functionalization steps introduce the necessary handles for further drug development. Operators should note the specific stoichiometry of LiHMDS (1.3 equivalents) to ensure complete conversion of the piperidone starting material. The following guide summarizes the critical operational phases derived from the patent examples, ensuring reproducibility and safety during scale-up.
- Perform Claisen condensation of N-Boc-4-piperidone with diethyl oxalate using LiHMDS at -78°C to form the beta-keto ester intermediate.
- Cyclize the beta-keto ester with hydrazine hydrate in acetic acid at 80°C to construct the pyrazolo[4,3-c]pyridine core.
- Execute aromatic nucleophilic substitution with 2-fluorobenzonitrile in DMSO at 100°C, followed by nitrile reduction and Fmoc protection.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this novel synthetic route are profound. By shifting away from precious metal catalysis and expensive halogenated substrates, the process inherently lowers the raw material costs associated with producing this key pharmaceutical intermediate. The reliance on commodity chemicals such as diethyl oxalate, hydrazine hydrate, and 2-fluorobenzonitrile ensures a stable and resilient supply chain, less susceptible to the volatility often seen with specialized organometallic reagents. Furthermore, the high yields observed across multiple steps—such as the 99% yield in the catalytic hydrogenation step and 96% in the SNAr reaction—significantly reduce the amount of waste generated per kilogram of product. This efficiency directly correlates to lower disposal costs and a reduced environmental footprint, aligning with modern green chemistry mandates.
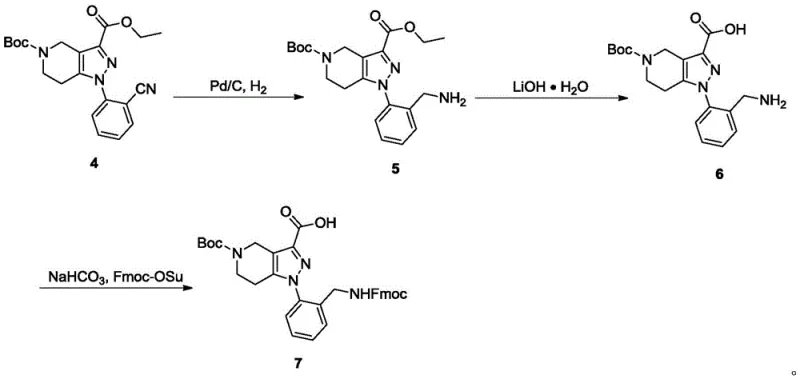
- Cost Reduction in Manufacturing: The elimination of copper catalysts and iodoarenes removes the need for expensive scavenging resins and complex purification protocols typically required to meet residual metal specifications. This simplification of the downstream processing workflow leads to substantial cost savings in both consumables and labor hours. Additionally, the use of common solvents like THF, methanol, and DMSO allows for efficient solvent recovery and recycling, further driving down the operational expenditure. The overall atom economy of the route ensures that a higher percentage of the input mass is converted into the desired product, maximizing the return on investment for raw material procurement.
- Enhanced Supply Chain Reliability: The starting materials identified in this pathway are bulk chemicals with established global supply networks, mitigating the risk of shortages that can plague niche reagent markets. The robustness of the reaction conditions, which tolerate standard industrial equipment without requiring specialized high-pressure or cryogenic infrastructure beyond the initial step, enhances the flexibility of manufacturing sites. This adaptability allows for multi-vendor sourcing strategies, ensuring continuity of supply even in the face of regional disruptions. The simplified process flow also shortens the manufacturing lead time, enabling faster response to market demand fluctuations.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, avoiding exothermic runaways and hazardous intermediates that complicate large-scale production. The aqueous workups and standard extraction procedures described are easily adaptable to existing plant infrastructure, facilitating a smooth transition from pilot plant to commercial tonnage. From an environmental perspective, the avoidance of heavy metals and the use of less toxic fluorine sources instead of iodine contribute to a cleaner waste stream. This compliance with stringent environmental regulations reduces the regulatory burden and potential liabilities associated with hazardous waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of pyrazolopiperidine derivatives. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the technology's capabilities. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this intermediate into their broader drug development pipelines.
Q: How does this novel route improve atom economy compared to traditional Ullmann coupling?
A: Traditional methods often rely on copper-catalyzed Ullmann coupling using iodoarenes, which generates stoichiometric metal waste and requires expensive iodine substrates. This patent utilizes aromatic nucleophilic substitution (SNAr) with 2-fluorobenzonitrile, a cheaper and more atom-economical reagent, eliminating the need for heavy metal catalysts and improving overall process sustainability.
Q: What are the critical temperature controls required for the pyrazole ring formation?
A: The initial enolate formation requires strict cryogenic control at -78°C to prevent side reactions during the addition of LiHMDS. However, the subsequent cyclization with hydrazine hydrate proceeds efficiently at 80°C in acetic acid, balancing reaction kinetics with thermal stability to ensure high purity of the pyrazolo[4,3-c]pyridine core.
Q: Is this process suitable for large-scale commercial production of API intermediates?
A: Yes, the process is designed for industrial scalability. It avoids hazardous reagents like phenylhydrazine and uses common solvents such as THF, DMSO, and methanol. The high yields reported in the patent examples (up to 99% for reduction steps) indicate a robust pathway amenable to kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazolopiperidine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our team of expert chemists has extensively analyzed the route disclosed in CN113264931B and possesses the technical capability to execute this synthesis with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of pyrazolopiperidine intermediate meets the exacting standards required for clinical and commercial applications.
We invite you to collaborate with us to optimize this synthesis for your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how our efficient manufacturing processes can enhance your project's economics. Please contact us today to request specific COA data and route feasibility assessments, and let us support your journey from discovery to market with superior chemical solutions.
