Advancing Chiral Phthalide Production: A Copper-Catalyzed Breakthrough for Commercial Scale
Introduction to Next-Generation Chiral Phthalide Synthesis
The pharmaceutical industry's relentless pursuit of high-purity chiral intermediates has driven significant innovation in catalytic methodologies, particularly for bioactive scaffolds like phthalides. Patent CN113200948A introduces a transformative approach to synthesizing chiral phthalides, specifically addressing the limitations of traditional noble metal catalysis. This technology leverages a sophisticated copper salt-chiral phosphine ligand-silane system to achieve asymmetric reduction and lactonization in a single tandem operation. For R&D directors and procurement strategists, this represents a pivotal shift away from expensive, high-pressure hydrogenation protocols toward a more sustainable, cost-effective, and scalable manifold. The ability to generate high-value chiral building blocks, such as L-butylphthalide (a critical intermediate for ischemic cerebral apoplexy treatments), with exceptional enantioselectivity under mild conditions underscores the commercial viability of this invention. By replacing scarce precious metals with abundant copper, the process not only aligns with green chemistry principles but also offers substantial supply chain resilience against volatile noble metal markets.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of chiral phthalides has been plagued by inherent inefficiencies and safety concerns associated with established catalytic systems. Traditional routes often depend on ruthenium or iridium complexes, such as [RuCl2(p-cymene)]2 or [Ir(COD)Cl]2, which are not only prohibitively expensive but also subject to significant supply chain volatility. Furthermore, many of these legacy processes require high-pressure hydrogen gas (up to 10 atm) to drive the reduction, necessitating specialized, costly reactor infrastructure and rigorous safety protocols that increase operational overhead. Another critical bottleneck is the reliance on racemic resolution; when synthesizing compounds like L-butylphthalide via resolution, up to 50% of the produced material is the pharmacologically inactive D-enantiomer, representing a massive waste of raw materials and processing capacity. These factors combined create a manufacturing landscape characterized by high costs, low atom economy, and complex purification requirements that hinder the efficient commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes an earth-abundant copper catalyst paired with tailored chiral phosphine ligands to orchestrate a highly selective transformation. This novel approach employs silane as a mild, liquid reducing agent, effectively bypassing the hazards and infrastructure demands of high-pressure hydrogenation. The reaction proceeds through a tandem mechanism where the ketone group of the o-acyl benzoate is asymmetrically reduced, followed immediately by spontaneous lactonization to form the chiral phthalide ring system. This one-pot strategy eliminates the need to isolate unstable alcohol intermediates, streamlining the workflow and reducing solvent consumption. Crucially, the system achieves high enantiomeric excess (ee values up to 93%) without the need for downstream resolution, ensuring that nearly all starting material is converted into the desired bioactive isomer. This paradigm shift offers a reliable chiral phthalide supplier pathway that is both economically superior and environmentally friendlier than its predecessors.
Mechanistic Insights into Cu-Catalyzed Asymmetric Reduction/Lactonization
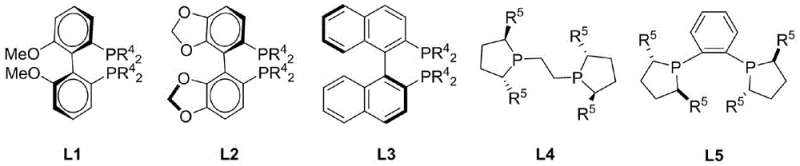
The core of this technological breakthrough lies in the precise coordination chemistry between the copper center and the chiral ligand environment. The catalytic cycle initiates with the formation of an active copper-hydride species generated in situ from the copper salt and the silane reductant. This reactive hydride complex then engages the prochiral ketone of the o-acyl benzoate substrate. The stereochemical outcome is rigorously controlled by the chiral pocket created by the phosphine ligand, which directs the hydride delivery to a specific face of the carbonyl group. The patent highlights a library of specialized ligands (L1-L5), including binaphthyl-derived and ferrocenyl-based phosphines, which can be tuned to optimize both reactivity and selectivity for different substrate profiles. Following the hydride transfer, the resulting chiral alkoxide intermediate undergoes an intramolecular nucleophilic attack on the adjacent ester carbonyl, closing the lactone ring and releasing the final chiral phthalide product while regenerating the active copper catalyst species for the next turnover.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-energy thermal processes. Because the reaction operates at relatively low temperatures (ranging from -20°C to 0°C), thermal degradation pathways and non-selective background reactions are significantly suppressed. The use of silane as a stoichiometric reductant ensures a clean reduction profile, minimizing the formation of over-reduced byproducts or side-reactions at the aromatic ring. Furthermore, the tandem nature of the reaction means that the intermediate alcohol is consumed as soon as it is formed, preventing its accumulation and potential racemization. This kinetic control is vital for maintaining high optical purity throughout the batch, ensuring that the final API intermediate meets the stringent purity specifications required for regulatory approval in pharmaceutical manufacturing.
How to Synthesize Chiral Phthalide Efficiently
Implementing this synthesis route requires careful attention to the preparation of the catalytic species and the order of reagent addition to maximize turnover numbers. The process begins with the activation of the copper precursor in an anhydrous solvent under an inert atmosphere, followed by the introduction of the silane to generate the active hydride. Once the catalyst is activated, the substrate is introduced at controlled temperatures to ensure high enantioselectivity. The detailed standardized synthetic steps, including specific molar ratios, solvent choices, and workup procedures derived from the patent examples, are outlined below to facilitate technology transfer and process validation.
- Prepare the catalyst system by adding a copper compound (e.g., CuF(PPh3)3·2MeOH) and a chiral phosphine ligand (e.g., L4) into a dry reaction vessel under inert atmosphere.
- Add the reaction solvent (e.g., toluene or THF) and stir, followed by the addition of the silane reducing agent (e.g., PMHS) to activate the catalyst species.
- Introduce the o-acyl benzoate substrate to the mixture at controlled temperatures (e.g., 0°C to -20°C), allow the tandem reduction/lactonization to proceed, and perform aqueous workup to isolate the chiral phthalide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this copper-catalyzed protocol offers immediate and tangible strategic benefits that extend beyond simple yield improvements. The elimination of noble metals removes a major cost driver and supply risk, as copper salts are commoditized and widely available compared to the geopolitically sensitive markets for ruthenium and iridium. Additionally, the replacement of high-pressure hydrogen gas with liquid silanes simplifies logistics, removing the need for specialized gas cylinder handling and high-pressure reactor certification, which drastically reduces capital expenditure for new production lines. The mild reaction conditions also translate to lower energy consumption for heating and cooling, contributing to a reduced carbon footprint and lower utility costs per kilogram of product. These factors collectively enable a more robust and flexible supply chain capable of responding rapidly to market demand fluctuations without the bottlenecks associated with complex high-pressure infrastructure.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts with inexpensive copper salts results in a drastic reduction in direct material costs. Since the catalyst loading is low and the metal is abundant, the overall cost of goods sold (COGS) for the chiral phthalide intermediate is significantly optimized. Furthermore, by avoiding the 50% material loss inherent in racemic resolution processes, the effective yield of the desired enantiomer is theoretically doubled relative to resolution methods, maximizing the value extracted from every kilogram of starting material purchased.
- Enhanced Supply Chain Reliability: Relying on earth-abundant copper and commercially available silanes insulates the production process from the supply shocks often seen with precious metals. The use of standard laboratory glassware or stainless steel reactors at atmospheric pressure means that manufacturing can be easily outsourced to a wider range of CDMOs without requiring specialized high-pressure facilities. This flexibility ensures continuous supply continuity even if specific high-tech manufacturing slots are constrained, providing a reliable chiral phthalide supplier advantage.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional methods that may require heavy metal scavengers or high-energy inputs. The workup involves simple aqueous quenching and extraction, which are well-understood unit operations easily scaled from pilot plant to multi-ton production. This simplicity facilitates rapid commercial scale-up of complex pharmaceutical intermediates while adhering to increasingly strict environmental regulations regarding heavy metal discharge and energy usage.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on yield expectations, safety profiles, and scalability potential for decision-makers evaluating this technology for their pipeline.
Q: What are the primary advantages of using copper catalysis over ruthenium or iridium for chiral phthalide synthesis?
A: The copper-catalyzed method described in patent CN113200948A eliminates the need for expensive noble metals like Ruthenium or Iridium, significantly reducing raw material costs. Furthermore, it operates under mild conditions without the requirement for high-pressure hydrogen gas, enhancing operational safety and simplifying equipment requirements for large-scale manufacturing.
Q: How does this method address the issue of low atom economy in traditional resolution processes?
A: Traditional methods often rely on resolving racemic mixtures, which theoretically wastes 50% of the material (the unwanted enantiomer). This asymmetric synthesis directly produces the desired chiral configuration with high enantiomeric excess (up to 93% ee), thereby maximizing raw material utilization and eliminating the need for complex separation steps to remove the ineffective isomer.
Q: Is the silane reducing agent safe for industrial scale-up?
A: Yes, the process utilizes silanes such as polymethylhydrosiloxane (PMHS) or phenylsilane, which are generally safer and easier to handle than high-pressure hydrogen gas. The reaction proceeds at atmospheric pressure and moderate temperatures, reducing the risks associated with high-pressure hydrogenation reactors commonly found in older methodologies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phthalide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced catalytic technologies to maintain competitiveness in the global pharmaceutical market. Our technical team has thoroughly analyzed the copper-catalyzed route for chiral phthalides and is fully prepared to support your development needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications and enantiomeric excess levels, guaranteeing that every batch meets the highest international standards for API intermediates.
We invite you to collaborate with us to leverage this cost-effective synthesis for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this innovative copper-catalyzed method can enhance your supply chain efficiency and reduce overall manufacturing costs.
