Advanced Synthetic Routes for High-Purity Bile Acids from Plant-Derived Steroids
The pharmaceutical and cosmetic industries are increasingly demanding high-purity active ingredients that are free from biological contaminants, driving a significant shift towards synthetic manufacturing processes. Patent CN111328332A introduces a groundbreaking methodology for the preparation of bile acids, specifically cholic acid, deoxycholic acid, and chenodeoxycholic acid, utilizing plant-derived steroids such as Dehydroepiandrosterone (DHEA) as the primary starting material. This innovation addresses critical safety concerns associated with traditional animal-extraction methods, which carry inherent risks of pathogen contamination including prions and bacterial toxins. By establishing a fully synthetic route, this technology enables the production of bile acids with superior purity profiles, meeting the rigorous standards required for injectable formulations and advanced cosmetic applications. The disclosed process not only enhances product safety but also stabilizes the supply chain by decoupling production from the volatility of animal agriculture markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, bile acids have been sourced primarily from bovine and ovine gallbladders, a practice that presents substantial regulatory and safety challenges for modern pharmaceutical manufacturing. The reliance on mammalian sources introduces the potential for contamination with animal-derived impurities, such as viruses, prions, and microbial metabolites, which are difficult to remove completely during downstream processing. Regulatory agencies globally have imposed stringent restrictions on animal-derived raw materials, requiring extensive testing and segregation protocols that significantly increase production costs and lead times. Furthermore, the supply of animal-sourced bile acids is subject to fluctuations in livestock populations and agricultural policies, creating uncertainty for long-term procurement planning. These limitations necessitate a transition to synthetic alternatives that can guarantee consistent quality and absolute freedom from biological hazards.
The Novel Approach
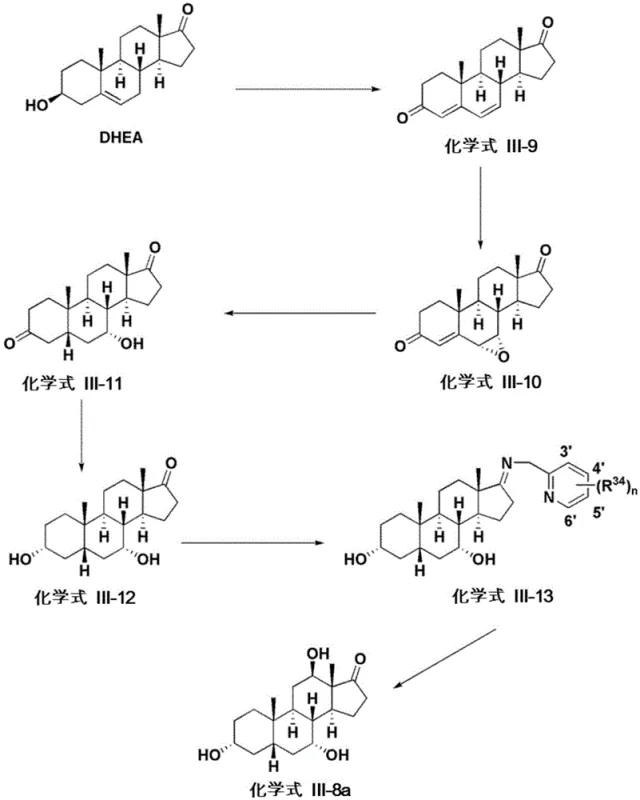
The novel synthetic approach detailed in the patent leverages the abundant availability of plant-derived steroids to construct the bile acid skeleton with precise stereochemical control. Starting from DHEA or acetyl-DHEA, the process employs a series of catalytic transformations to introduce the requisite hydroxyl groups at the 3, 7, and 12 positions of the steroid nucleus. This method bypasses the need for animal tissues entirely, resulting in a final product that is inherently free from animal-derived impurities. The route incorporates efficient oxidation and reduction steps, such as copper-mediated oxidation and selective hydrogenation, which allow for the scalable production of high-purity intermediates. By utilizing non-animal starting materials, manufacturers can achieve a more robust and compliant supply chain, suitable for sensitive applications like non-surgical fat dissolution treatments where purity is paramount.
Mechanistic Insights into Copper-Catalyzed Oxidation and Stereoselective Reduction
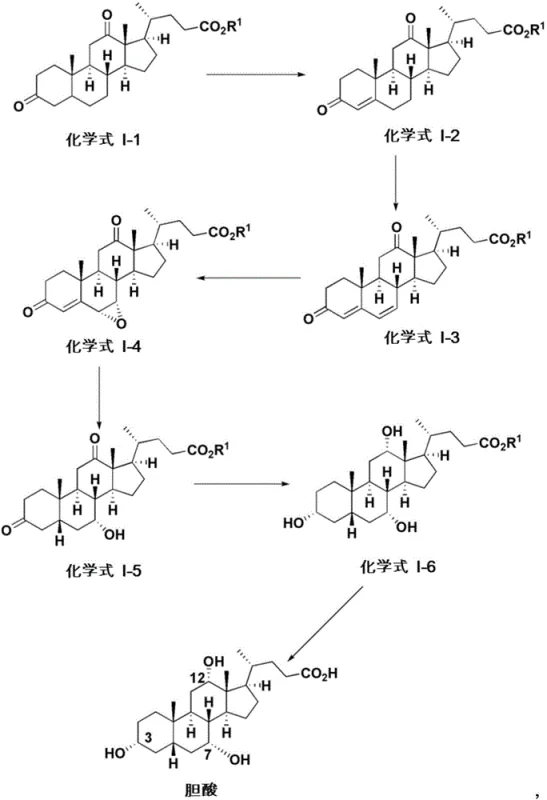
A critical component of this synthetic strategy involves the precise installation of oxygenated functional groups on the steroid core, particularly the introduction of the 12-alpha-hydroxyl group which distinguishes cholic acid from other bile acids. The patent describes a sophisticated copper-mediated oxidation process where an imine intermediate, formed from the reaction of a ketone with an amine such as 2-pyridylmethylamine, is oxidized in the presence of oxygen and a copper salt. This mechanism allows for the selective oxidation at the C-12 position without affecting other sensitive functionalities on the molecule. The use of copper catalysts, such as Cu(MeCN)4PF6 or Cu(OTf)2, facilitates a radical or coordination-based pathway that ensures high regioselectivity. Following oxidation, the process utilizes stereoselective reduction agents, including lithium tri-tert-butoxyaluminum hydride, to establish the correct alpha-configuration of the hydroxyl groups. This level of mechanistic control is essential for producing biologically active isomers that meet pharmacopeial specifications.
Furthermore, the synthesis employs Horner-Wadsworth-Emmons (HWE) olefination reactions to extend the side chain at the C-17 position, converting aldehyde intermediates into alpha,beta-unsaturated esters. This step is crucial for building the carboxylic acid tail characteristic of bile acids. The subsequent hydrogenation of the double bond is carefully managed to avoid epimerization at the C-20 position, preserving the natural stereochemistry of the side chain. The combination of these catalytic cycles creates a robust pathway that minimizes the formation of diastereomeric impurities. By understanding these mechanistic nuances, process chemists can optimize reaction conditions to maximize yield and purity, ensuring that the final API intermediate is suitable for direct formulation or further derivatization into pharmaceutically acceptable salts.
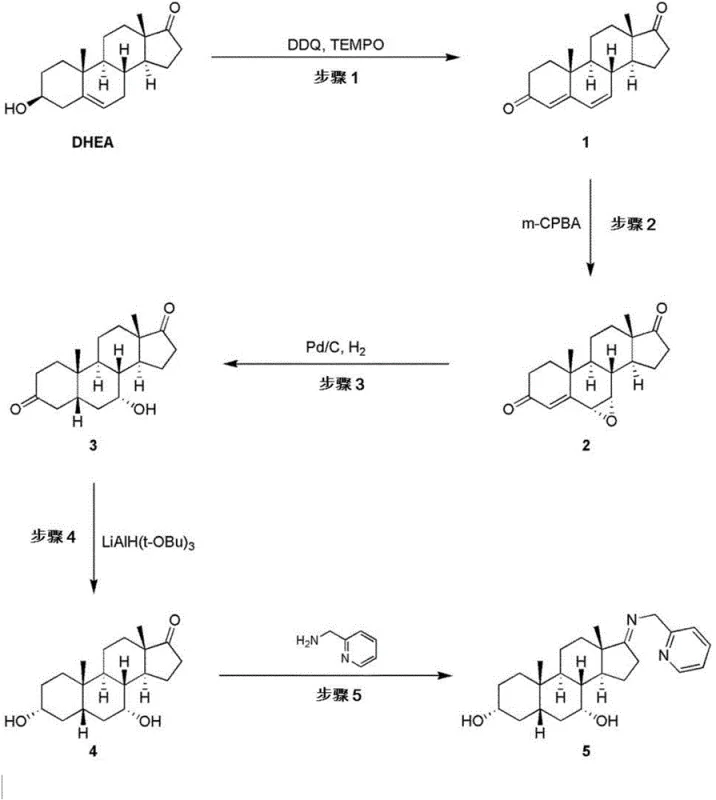
How to Synthesize Cholic Acid Efficiently
The synthesis of cholic acid from DHEA involves a multi-step sequence that requires careful control of reaction parameters to ensure high yield and stereochemical integrity. The process begins with the oxidation of the starting steroid to introduce unsaturation and oxygen functionality, followed by side-chain manipulation and final deprotection steps. Each stage of the synthesis is designed to be scalable, utilizing reagents and conditions that are compatible with large-scale manufacturing environments. The detailed procedural steps involve specific stoichiometric ratios, temperature controls, and purification techniques such as column chromatography and crystallization to isolate intermediates of sufficient purity for subsequent reactions. Implementing this route requires a thorough understanding of steroid chemistry and the ability to manage complex reaction mixtures effectively.
- Oxidize DHEA using DDQ and TEMPO to form the dienone intermediate, followed by epoxidation with m-CPBA.
- Perform hydrogenation to open the epoxide and reduce double bonds, establishing the 5-beta stereochemistry.
- Execute copper-mediated oxidation at C-12, followed by side-chain extension via Horner-Wadsworth-Emmons reaction and final reduction.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this synthetic manufacturing process offers significant strategic benefits for procurement and supply chain management within the pharmaceutical and cosmetic sectors. By shifting away from animal-derived sources, companies can mitigate the risks associated with biological contamination and regulatory non-compliance, which often lead to costly product recalls or market access delays. The use of plant-based starting materials like DHEA ensures a more stable and predictable raw material supply, independent of livestock disease outbreaks or agricultural trade restrictions. This stability translates into enhanced supply chain reliability, allowing manufacturers to maintain consistent production schedules and meet global demand without interruption. Additionally, the synthetic route facilitates better control over the impurity profile, reducing the burden on quality control laboratories and streamlining the release testing process for finished goods.
- Cost Reduction in Manufacturing: The elimination of expensive purification steps required to remove animal-derived pathogens significantly lowers the overall cost of goods sold. Synthetic routes allow for the optimization of reaction yields and the recycling of solvents and catalysts, further driving down production expenses. By avoiding the volatility of animal by-product markets, manufacturers can secure more favorable long-term pricing agreements for raw materials. The streamlined process flow reduces the number of unit operations required, leading to lower energy consumption and reduced waste disposal costs. These efficiencies collectively contribute to a more competitive cost structure for high-purity bile acid intermediates.
- Enhanced Supply Chain Reliability: Sourcing starting materials from plant-based feedstocks provides a resilient supply chain that is less susceptible to external shocks compared to animal-based sourcing. The availability of DHEA from multiple global suppliers ensures that production is not bottlenecked by a single source of origin. This diversification of supply reduces the risk of shortages and enables manufacturers to scale production volumes rapidly in response to market demand. Furthermore, the synthetic nature of the product simplifies logistics and storage requirements, as there is no need for specialized cold chain handling often associated with biological extracts. This reliability is crucial for maintaining uninterrupted supply to downstream formulation partners.
- Scalability and Environmental Compliance: The described synthetic methods are designed with scalability in mind, utilizing standard chemical engineering principles that facilitate easy transfer from laboratory to commercial scale. The process avoids the use of hazardous biological waste, simplifying environmental compliance and waste management protocols. Synthetic manufacturing allows for the implementation of green chemistry practices, such as solvent recovery and catalyst reuse, which align with corporate sustainability goals. The ability to produce large quantities of high-purity material without the ethical and environmental concerns of animal slaughter enhances the brand reputation of companies adopting this technology. This alignment with environmental, social, and governance (ESG) criteria is increasingly important for securing investment and market share.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthetic production of bile acids as described in the patent documentation. These insights are derived from the specific chemical transformations and process advantages outlined in the disclosure, providing clarity on the feasibility and benefits of this manufacturing approach. Understanding these details helps stakeholders evaluate the potential for integrating this technology into their existing production portfolios. The answers reflect the current state of the art in steroid synthesis and regulatory expectations for pharmaceutical ingredients.
Q: Why is synthetic bile acid preferred over animal-extracted sources?
A: Synthetic routes eliminate the risk of animal-derived impurities such as prions and viral pathogens, ensuring compliance with stringent regulatory standards for pharmaceutical and cosmetic use.
Q: What is the key advantage of using DHEA as a starting material?
A: DHEA is a readily available plant-derived steroid that provides a cost-effective and sustainable foundation for building the complex bile acid skeleton without relying on livestock supply chains.
Q: How does this process ensure high stereochemical purity?
A: The process utilizes specific catalytic conditions, such as copper-mediated oxidation and selective hydrogenation, to control stereochemistry at critical positions like C-3, C-7, and C-12.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cholic Acid Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract development, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the complexities of steroid chemistry and is equipped to implement the advanced synthetic routes described in patent CN111328332A with precision and efficiency. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of cholic acid or deoxycholic acid meets the highest international standards. Our commitment to quality and safety makes us an ideal partner for pharmaceutical companies seeking to transition to synthetic, animal-free supply chains for their bile acid requirements.
We invite you to contact our technical procurement team to discuss your specific project needs and request a Customized Cost-Saving Analysis tailored to your volume requirements. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your product development goals. By collaborating with NINGBO INNO PHARMCHEM, you gain access to a reliable supply of high-purity pharmaceutical intermediates that drive innovation and ensure patient safety. Let us help you navigate the complexities of synthetic steroid manufacturing and secure a competitive advantage in the global market.
