Advanced Synthesis of 6-(2-Methoxyethoxy)-N-Hydroxynaphthalimide Trifluoromethanesulfonic Acid for High-Performance Lithography
Advanced Synthesis of 6-(2-Methoxyethoxy)-N-Hydroxynaphthalimide Trifluoromethanesulfonic Acid for High-Performance Lithography
The semiconductor industry's relentless drive toward smaller node sizes has placed unprecedented demands on the performance of photoresist materials, specifically requiring photoacid generators (PAGs) with exceptional purity and thermal stability. Patent CN109384720B introduces a robust and scalable methodology for synthesizing 6-(2-methoxyethoxy)-N-hydroxynaphthalimide trifluoromethanesulfonic acid, a critical component in next-generation lithography processes. This technical disclosure addresses long-standing challenges in the organic synthesis of naphthalimide derivatives, offering a pathway that mitigates the risks of hydrolysis and impurity formation that have plagued previous attempts. By re-engineering the reaction sequence and optimizing solvent systems, this innovation provides a reliable foundation for the mass production of high-performance electronic chemicals.
For R&D directors and process engineers, the significance of this patent lies in its ability to deliver a product with an HPLC purity exceeding 99 percent, a specification that is non-negotiable for advanced node manufacturing. The method employs a strategic three-step sequence that begins with the formation of a stable hydroxyimide intermediate, followed by a controlled etherification, and concludes with a precise triflation. This approach not only enhances the chemical integrity of the final molecule but also simplifies the purification workflow, thereby reducing the overall environmental footprint of the manufacturing process. As we delve deeper into the mechanistic advantages and commercial implications, it becomes clear that this synthesis route represents a substantial leap forward in the field of specialty electronic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex naphthalimide sulfonates has been hindered by the inherent instability of the anhydride precursor under basic conditions. Traditional routes often attempt to introduce the ether functionality early in the synthesis using strong bases and elevated temperatures. However, as illustrated in the failure analysis of prior art, these conditions are detrimental to the acid anhydride structure, leading to rapid and irreversible hydrolysis. 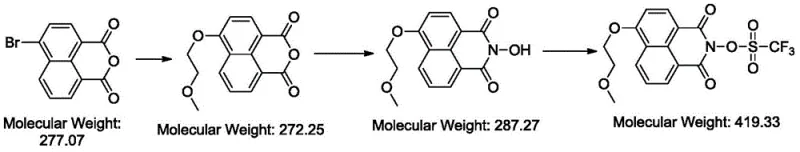 This hydrolytic degradation results in the formation of dicarboxylic acid byproducts rather than the desired imide, effectively shutting down the reaction pathway before the target intermediate can be generated. Furthermore, even if the intermediate is formed, subsequent steps involving strong alkali can cause the hydrolysis of the triflate group, rendering the final product useless for lithographic applications.
This hydrolytic degradation results in the formation of dicarboxylic acid byproducts rather than the desired imide, effectively shutting down the reaction pathway before the target intermediate can be generated. Furthermore, even if the intermediate is formed, subsequent steps involving strong alkali can cause the hydrolysis of the triflate group, rendering the final product useless for lithographic applications.
The consequences of these conventional failures extend beyond mere yield loss; they introduce significant complexity into the purification process. The presence of hydrolyzed byproducts and partially reacted species creates a difficult-to-separate impurity profile that can compromise the performance of the final photoresist. For procurement managers, this translates to higher costs associated with raw material waste and extensive downstream processing. The inability to consistently achieve high purity using these older methods poses a supply chain risk, as batch-to-batch variability can disrupt the delicate balance required in semiconductor fabrication lines. Therefore, a fundamental rethinking of the synthetic strategy was necessary to overcome these thermodynamic and kinetic barriers.
The Novel Approach
The innovative strategy outlined in patent CN109384720B circumvents these pitfalls by reversing the order of operations and utilizing milder reaction conditions that preserve the integrity of the sensitive functional groups. Instead of exposing the anhydride to harsh bases immediately, the new method first converts the anhydride into a more stable hydroxyimide structure using hydroxylamine hydrochloride. This initial transformation protects the carbonyl functionality while setting the stage for subsequent modifications. The subsequent etherification step is carefully controlled, using ethylene glycol monomethyl ether as both solvent and reagent, which allows for the introduction of the methoxyethoxy group without triggering the catastrophic hydrolysis seen in prior art. This strategic sequencing ensures that the core naphthalimide scaffold remains intact throughout the synthesis.
Furthermore, the final triflation step is executed under strictly controlled low-temperature conditions, utilizing pyridine as a base to scavenge the generated acid without promoting decomposition. This meticulous attention to reaction parameters allows for the isolation of the target trifluoromethanesulfonic acid derivative with exceptional fidelity. The result is a process that not only avoids the formation of hydrolytic impurities but also facilitates easier purification, as the impurity profile is significantly cleaner compared to conventional methods. For supply chain leaders, this reliability means a more predictable production schedule and a consistent supply of high-quality material, which is essential for maintaining the uptime of high-volume semiconductor manufacturing facilities.
Mechanistic Insights into Sequential Functionalization and Triflation
The success of this synthesis hinges on a deep understanding of nucleophilic substitution mechanisms and the stabilization of transition states. In the first step, the reaction between 4-bromo-1,8-naphthalic anhydride and hydroxylamine hydrochloride proceeds via a nucleophilic attack on one of the carbonyl carbons of the anhydride ring. 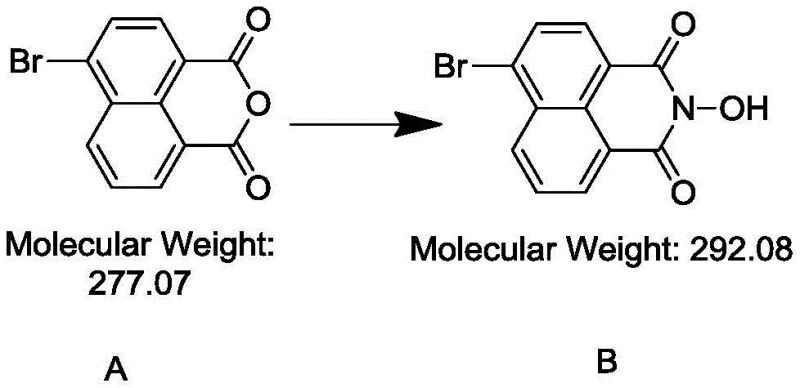 The presence of sodium bicarbonate serves to neutralize the hydrochloric acid generated during the reaction, driving the equilibrium towards the formation of the N-hydroxyimide intermediate (Compound B). This step is crucial because it converts the highly reactive anhydride into a more robust imide structure that can withstand the basic conditions required for the subsequent etherification. The use of ethanol as a solvent in this step provides an optimal medium for the dissolution of reactants while allowing for easy removal of the solvent post-reaction, facilitating the isolation of the intermediate in high purity.
The presence of sodium bicarbonate serves to neutralize the hydrochloric acid generated during the reaction, driving the equilibrium towards the formation of the N-hydroxyimide intermediate (Compound B). This step is crucial because it converts the highly reactive anhydride into a more robust imide structure that can withstand the basic conditions required for the subsequent etherification. The use of ethanol as a solvent in this step provides an optimal medium for the dissolution of reactants while allowing for easy removal of the solvent post-reaction, facilitating the isolation of the intermediate in high purity.
The final triflation step represents the culmination of the synthetic effort, where the hydroxyl group on the nitrogen atom is converted into a triflate leaving group. 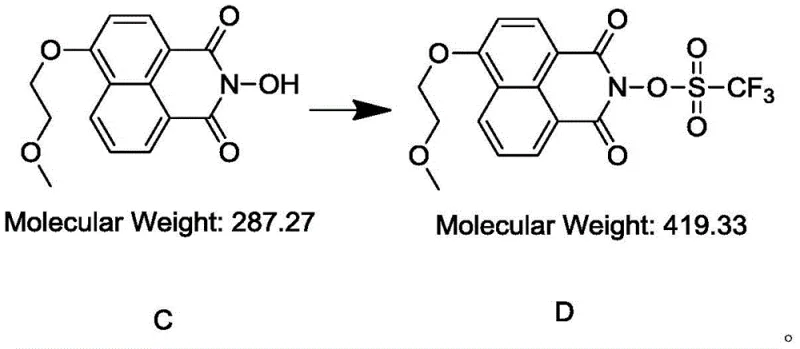 This transformation is achieved by reacting the intermediate (Compound C) with trifluoromethanesulfonic anhydride in the presence of pyridine. The mechanism involves the nucleophilic attack of the nitrogen-bound oxygen on the sulfur atom of the anhydride, facilitated by the electron-withdrawing nature of the trifluoromethyl group. Pyridine acts as a proton scavenger, neutralizing the triflic acid byproduct and preventing acid-catalyzed degradation of the product. The strict temperature control, starting at -5 to 0°C and gradually warming to 10-20°C, is critical to managing the exothermicity of the reaction and preventing the formation of sulfonate esters on other potential nucleophilic sites. This precise control ensures that the triflate group is installed exclusively at the desired position, resulting in a product with the structural fidelity required for high-performance photoacid generation.
This transformation is achieved by reacting the intermediate (Compound C) with trifluoromethanesulfonic anhydride in the presence of pyridine. The mechanism involves the nucleophilic attack of the nitrogen-bound oxygen on the sulfur atom of the anhydride, facilitated by the electron-withdrawing nature of the trifluoromethyl group. Pyridine acts as a proton scavenger, neutralizing the triflic acid byproduct and preventing acid-catalyzed degradation of the product. The strict temperature control, starting at -5 to 0°C and gradually warming to 10-20°C, is critical to managing the exothermicity of the reaction and preventing the formation of sulfonate esters on other potential nucleophilic sites. This precise control ensures that the triflate group is installed exclusively at the desired position, resulting in a product with the structural fidelity required for high-performance photoacid generation.
How to Synthesize 6-(2-Methoxyethoxy)-N-Hydroxynaphthalimide Trifluoromethanesulfonic Acid Efficiently
Implementing this synthesis in a commercial setting requires adherence to strict operational protocols to ensure safety and reproducibility. The process is divided into three distinct stages, each with specific temperature and stoichiometric requirements that must be monitored closely. The initial condensation sets the foundation for the entire sequence, while the etherification and triflation steps demand precise control over reaction kinetics to minimize byproduct formation. Operators must be trained to recognize the visual cues of reaction progress, such as color changes and precipitation, which serve as indicators of reaction completion alongside analytical data. The following guide outlines the standardized approach derived from the patent examples, providing a roadmap for scaling this chemistry from the laboratory to pilot and commercial production scales.
- Condense 4-bromo-1,8-naphthalic anhydride with hydroxylamine hydrochloride and sodium bicarbonate in ethanol under reflux to form the hydroxyimide intermediate.
- Perform nucleophilic substitution using ethylene glycol monomethyl ether and sodium hydroxide at 78-80°C to introduce the methoxyethoxy group.
- React the intermediate with trifluoromethanesulfonic anhydride and pyridine in dichloromethane at low temperature (-5 to 0°C) followed by warming to complete the triflation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this novel synthetic route offers tangible benefits that extend far beyond the laboratory bench. The primary advantage lies in the significant reduction of raw material waste associated with the avoidance of hydrolytic side reactions. By eliminating the conditions that lead to the degradation of the anhydride precursor, the process maximizes the atom economy of the synthesis, ensuring that a higher proportion of expensive starting materials are converted into valuable product. This efficiency directly translates to cost reduction in electronic chemical manufacturing, as the need for excessive overcharging of reagents to compensate for poor yields is removed. Furthermore, the simplified impurity profile reduces the burden on purification resources, lowering the consumption of solvents and chromatography media.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis eliminates the need for complex protection and deprotection strategies that are often required in alternative routes. By utilizing a direct etherification strategy that tolerates the imide functionality, the number of unit operations is minimized, leading to reduced labor and energy costs. The high purity of the crude product emerging from the reactor means that less intensive purification is required to meet final specifications, further driving down the cost of goods sold. Additionally, the use of common industrial solvents like ethanol and dichloromethane ensures that material costs remain stable and predictable, shielding the supply chain from volatility associated with exotic or specialized reagents.
- Enhanced Supply Chain Reliability: The robustness of this chemical process ensures a consistent and reliable supply of critical photoacid generators. Unlike methods that are sensitive to minor fluctuations in temperature or moisture, this route demonstrates a wide operating window that accommodates the realities of large-scale manufacturing. This resilience reduces the risk of batch failures, which can otherwise lead to costly delays in the delivery of materials to semiconductor fabs. By securing a manufacturing process that is less prone to deviation, companies can offer their customers greater certainty regarding lead times and inventory availability, strengthening long-term partnerships in the highly competitive electronics sector.
- Scalability and Environmental Compliance: The synthetic pathway is designed with scalability in mind, utilizing reaction conditions that are easily transferable from glassware to stainless steel reactors. The absence of extremely hazardous reagents or extreme pressure conditions simplifies the engineering controls required for safe operation. Moreover, the improved selectivity of the reaction results in a cleaner waste stream, facilitating easier treatment and disposal of effluents in compliance with stringent environmental regulations. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturer, a factor of increasing importance to global technology clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized photoacid generator. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity on the capabilities and limitations of the described method. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this material into their existing supply chains or product formulations.
Q: Why do conventional synthesis routes for this photoacid generator often fail?
A: Conventional methods often utilize strong bases at high temperatures during the initial etherification step. Under these harsh alkaline conditions, the acid anhydride structure of the starting material is prone to rapid hydrolysis, preventing the formation of the target intermediate and leading to significant yield loss.
Q: What purity levels can be achieved with this novel synthetic method?
A: The patented process achieves exceptional purity standards suitable for semiconductor applications. Through optimized reaction conditions and purification steps including column chromatography and recrystallization, the final product consistently reaches an HPLC purity of greater than or equal to 99 percent, with严格控制 acid value and water content.
Q: What are the critical control points for ensuring high yield in the final step?
A: The final triflation step requires strict temperature control, initially maintaining -5 to 0°C during the addition of trifluoromethanesulfonic anhydride to prevent side reactions. Subsequent warming to 10-20°C ensures complete conversion, while monitoring via TLC is essential to confirm the disappearance of raw materials before workup.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-(2-Methoxyethoxy)-N-Hydroxynaphthalimide Trifluoromethanesulfonic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent concept to commercial reality requires a partner with deep technical expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results observed in the laboratory are faithfully reproduced at an industrial scale. We understand that for applications in semiconductor lithography, stringency is paramount; therefore, our manufacturing facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including HPLC purity greater than 99 percent and controlled acid values. Our dedication to process optimization allows us to deliver materials that meet the exacting standards of the global electronics industry.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific application needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to validate the feasibility of this route for your specific formulation, our technical procurement team is ready to assist. We encourage you to reach out to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven performance. Together, we can drive innovation in electronic materials while ensuring a secure and efficient supply of critical components for the future of technology.
