Advanced C-H Acetoxylation Strategy for High-Purity Ortho-Substituted Aryl Pyrimidines
Advanced C-H Acetoxylation Strategy for High-Purity Ortho-Substituted Aryl Pyrimidines
The landscape of fine chemical synthesis is constantly evolving, driven by the demand for more efficient and selective pathways to complex heterocyclic scaffolds. Patent CN101812022A introduces a groundbreaking methodology for the preparation of ortho-monovinylphenyl substituted compounds of aryl pyrimidine, a class of molecules that serves as a critical backbone in both pharmaceutical chemistry and advanced electronic materials. This innovation addresses long-standing challenges in regioselective C-H functionalization, offering a robust route to access valuable intermediates that were previously difficult to synthesize with high purity. By leveraging a dual catalytic system involving palladium and copper, coupled with a hypervalent iodine oxidant, this technology enables the direct installation of acetoxy groups under remarkably mild conditions. For industry leaders seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this synthetic breakthrough is essential for securing supply chains for next-generation APIs and OLED materials.
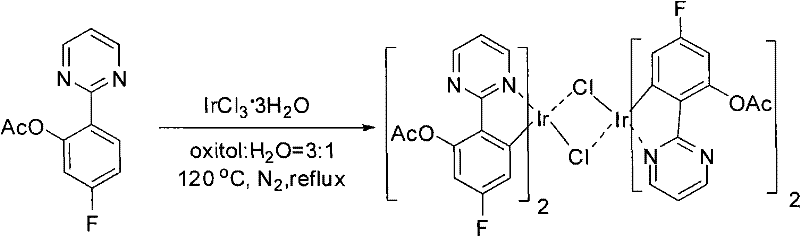
The significance of this technology extends far beyond simple molecule construction; it unlocks access to high-value functional materials. As illustrated in the downstream applications, the resulting ortho-acetoxy aryl pyrimidines can be readily transformed into phenolic compounds or utilized directly in the coordination chemistry of iridium complexes. These iridium complexes are pivotal in the fabrication of organic light-emitting diodes (OLEDs), where they function as highly efficient phosphorescent dopants. The ability to synthesize the precursor ligands with high regiocontrol and yield directly impacts the performance and color purity of the final display devices. Consequently, this patent represents not just a chemical curiosity, but a strategic asset for manufacturers aiming to reduce lead time for high-purity electronic chemical precursors and pharmaceutical building blocks alike.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ortho-acetoxylated aryl compounds has been plagued by significant operational and chemical hurdles that hindered large-scale adoption. Traditional methods often relied on the use of acetate solvents under the catalysis of palladium in the presence of high-pressure oxygen gas. This approach, while conceptually straightforward, introduced severe safety risks associated with handling pressurized oxygen in the presence of organic solvents and catalysts. Furthermore, these reactions frequently suffered from poor selectivity, leading to the generation of complex by-product mixtures that required tedious and costly purification steps. The harsh reaction conditions also limited the scope of compatible substrates, particularly those containing sensitive functional groups that could degrade under oxidative stress. For a procurement manager, these factors translate into unpredictable batch quality, increased waste disposal costs, and potential supply chain disruptions due to safety incidents or low throughput.
The Novel Approach
In stark contrast, the methodology disclosed in CN101812022A utilizes a sophisticated yet operationally simple system employing iodobenzene diacetate (PhI(OAc)2) as the terminal oxidant. This shift from gaseous oxygen to a solid, bench-stable oxidant fundamentally changes the risk profile of the manufacturing process, allowing reactions to proceed safely at atmospheric pressure. The reaction is conducted in a mixture of acetic anhydride and acetic acid at a moderate temperature of 75°C, which is significantly milder than many traditional oxidative protocols. This novel approach ensures high regioselectivity for the ortho-position, driven by the coordinating ability of the pyrimidine nitrogen atoms, thereby minimizing the formation of unwanted isomers. The result is a streamlined process that delivers yields as high as 88%, providing a compelling case for cost reduction in pharmaceutical intermediate manufacturing through improved material efficiency and simplified downstream processing.
Mechanistic Insights into Pd/Cu Co-Catalyzed Oxidative Acetoxylation
To fully appreciate the robustness of this synthetic route, one must delve into the mechanistic interplay between the palladium catalyst, the copper co-catalyst, and the hypervalent iodine oxidant. The reaction initiates with the coordination of the palladium species to the nitrogen atom of the pyrimidine ring, which directs the metal center to the adjacent ortho-C-H bond. Through a concerted metalation-deprotonation (CMD) pathway or a similar electrophilic palladation mechanism, a stable five-membered palladacycle intermediate is formed. This step is crucial as it dictates the regioselectivity of the entire transformation, ensuring that the acetoxy group is installed exclusively at the desired position. The presence of copper trifluoroacetate acts as a crucial promoter, likely facilitating the re-oxidation of the reduced palladium species back to its active Pd(II) state, thus closing the catalytic cycle efficiently. This synergistic effect allows the reaction to proceed with lower catalyst loading and higher turnover numbers compared to systems relying solely on palladium.
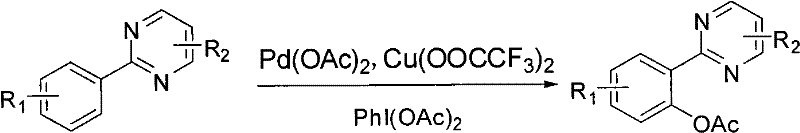
Furthermore, the choice of PhI(OAc)2 as the oxidant plays a dual role in both driving the thermodynamics of the reaction and serving as the source of the acetoxy group. Upon oxidation of the organopalladium intermediate, a high-valent Pd(IV) species is generated, which subsequently undergoes reductive elimination to forge the C-O bond and release the desired product. This mechanism effectively suppresses the formation of homocoupling by-products that are common in oxidative coupling reactions. From an impurity control perspective, the mildness of the oxidant prevents the over-oxidation of the electron-rich aromatic rings, preserving sensitive substituents such as methoxy or fluoro groups. For R&D directors focused on purity profiles, this mechanistic elegance ensures a cleaner crude reaction mixture, reducing the burden on purification teams and ensuring that the final API intermediate meets stringent regulatory specifications without extensive recrystallization or chromatographic intervention.
How to Synthesize 2-(2-acetoxyl group phenyl) pyrimidine Efficiently
The practical implementation of this chemistry is designed for scalability and ease of execution in a standard multipurpose reactor. The process begins by charging the aryl pyrimidine substrate along with the copper and palladium catalysts into a reaction vessel containing the acetic anhydride/acetic acid solvent system. The addition of the solid oxidant is controlled to manage any exotherm, although the reaction is generally well-behaved at the prescribed 75°C setpoint. Monitoring the reaction progress via thin-layer chromatography (TLC) allows operators to determine the precise endpoint, ensuring maximum conversion while preventing potential degradation from prolonged heating. Once the starting material is consumed, the workup involves a straightforward aqueous quench and extraction, followed by standard purification techniques. The detailed standardized synthesis steps for this specific transformation are outlined in the guide below, providing a clear roadmap for technology transfer teams.
- Dissolve aryl pyrimidine substrate, copper trifluoroacetate, and iodobenzene diacetate in a mixed solvent of acetic anhydride and acetic acid (15: 1 ratio).
- Add palladium catalyst and heat the reaction mixture to 75°C, stirring until the starting material is completely consumed as monitored by TLC.
- Remove solvents, neutralize with saturated sodium bicarbonate, extract with ethyl acetate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders managing the bottom line and logistical continuity, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere chemical yield. The elimination of high-pressure oxygen infrastructure removes a significant capital expenditure barrier and reduces the ongoing maintenance and safety compliance costs associated with hazardous gas handling. Moreover, the use of commercially available and stable reagents like PhI(OAc)2 simplifies inventory management and reduces the risk of supply interruptions caused by the specialized logistics required for compressed gases. The mild reaction conditions also imply lower energy consumption for heating and cooling cycles, contributing to a smaller carbon footprint and aligning with modern sustainability goals. These factors collectively contribute to a more resilient and cost-effective supply chain for critical heterocyclic intermediates.
- Cost Reduction in Manufacturing: The transition to a liquid-phase oxidative system using solid oxidants drastically simplifies the reactor engineering requirements, allowing for the use of standard glass-lined or stainless steel vessels without the need for expensive pressure-rated autoclaves. This reduction in equipment complexity directly lowers the depreciation costs allocated to each batch. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to by-products, thereby improving the overall mass balance and reducing the cost of goods sold (COGS). The ability to recycle solvent streams further enhances the economic viability of the process, making it highly competitive for large-scale production runs.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals such as acetic acid, acetic anhydride, and iodobenzene diacetate, the manufacturing process is decoupled from the volatility of specialized reagent markets. These raw materials are produced globally in massive quantities, ensuring a stable supply even during periods of regional disruption. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in temperature or mixing efficiency, leading to consistent batch-to-batch quality. This reliability is paramount for pharmaceutical customers who require guaranteed delivery schedules to maintain their own production timelines for finished dosage forms.
- Scalability and Environmental Compliance: The absence of high-pressure gases and the use of relatively benign solvents facilitate a smoother scale-up from kilogram to tonne production scales. Safety hazards are inherently minimized, reducing the regulatory burden and insurance premiums associated with chemical manufacturing. Furthermore, the waste stream generated is primarily composed of iodobenzene and acetate salts, which are easier to treat and dispose of compared to the complex heavy metal waste often associated with stoichiometric oxidants like chromates or permanganates. This environmental compatibility ensures long-term operational continuity in increasingly regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the scope, safety, and applicability of this ortho-acetoxylation technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, aimed at clarifying the operational parameters for potential licensees or contract manufacturing partners. Understanding these details is crucial for assessing the fit of this technology within existing production portfolios and for anticipating any specific handling requirements during tech transfer.
Q: What is the primary advantage of using PhI(OAc)2 over oxygen gas in this acetoxylation?
A: Using iodobenzene diacetate (PhI(OAc)2) eliminates the need for high-pressure oxygen equipment, significantly enhancing operational safety and simplifying the reaction setup while maintaining high regioselectivity.
Q: Can this method tolerate electron-withdrawing groups on the phenyl ring?
A: Yes, the patented method demonstrates excellent compatibility with various substituents including p-F, p-COOEt, and p-OTs, overcoming limitations of previous methods which struggled with electron-deficient substrates.
Q: What are the downstream applications of the resulting ortho-acetoxy compounds?
A: These compounds serve as versatile intermediates that can be hydrolyzed to phenols for drug synthesis or converted into tosylates for cross-coupling reactions, and are also precursors for iridium-based OLED phosphorescent materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ortho-Acetoxy Phenylpyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development and material science programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of complex aryl pyrimidine derivatives or large-scale supply of established intermediates, our infrastructure is designed to support your growth and innovation.
We invite you to engage with our technical procurement team to discuss how this advanced acetoxylation technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring that you have the most reliable partner for your fine chemical requirements.
