Scalable Synthesis of Rosuvastatin Intermediates: A Safer, High-Yield Commercial Route
The pharmaceutical industry continuously seeks robust and safe methodologies for producing critical active pharmaceutical ingredient (API) intermediates, particularly for high-volume drugs like statins. Patent CN1301977C introduces a groundbreaking preparation method for aminopyrimidine compounds, specifically focusing on 2-(N-methyl-N-methylsulfonylamino)pyrimidine derivatives which serve as pivotal precursors for cholesterol-lowering agents such as Rosuvastatin. This technology represents a significant leap forward in process chemistry, addressing long-standing safety and efficiency concerns associated with traditional synthetic routes. By leveraging a novel sequence of condensation, oxidation, and substitution reactions, this patent provides a framework for generating high-purity intermediates without relying on extremely hazardous reagents. The structural versatility of the compounds described allows for broad application in medicinal chemistry, ensuring that manufacturers can meet stringent regulatory requirements for impurity profiles.
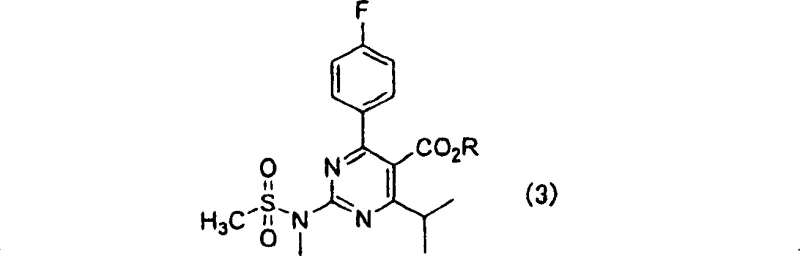
Furthermore, the economic implications of adopting this synthesis route are profound for supply chain stakeholders aiming to secure a reliable pharmaceutical intermediates supplier. The process utilizes readily available starting materials such as isobutyl acetate, 4-fluorobenzaldehyde, and urea, which stabilizes raw material costs and mitigates supply risks often associated with exotic reagents. The ability to conduct these reactions under relatively mild conditions using common metal salt catalysts like copper(I) chloride enhances the accessibility of the technology for contract development and manufacturing organizations (CDMOs). As the global demand for cardiovascular medications continues to rise, the capacity to produce key building blocks like the compound of formula (3) efficiently becomes a strategic asset. This patent not only secures intellectual property around a superior chemical route but also establishes a benchmark for sustainable and scalable manufacturing practices in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in WO01/04100, have historically relied on the use of cyanogen chloride to construct the pyrimidine ring system necessary for statin intermediates. This approach presents severe disadvantages for industrial production, primarily due to the extreme toxicity and handling hazards associated with cyanogen chloride gas. The requirement for such dangerous reagents necessitates specialized containment equipment and rigorous safety protocols, which drastically inflate capital expenditure and operational costs for manufacturing facilities. Moreover, the conventional routes often suffer from suboptimal yields, with documented overall yields based on starting esters hovering around 45.5%, leading to significant material loss and increased waste generation. These inefficiencies create bottlenecks in the supply chain, making it difficult to meet the high-volume demands of the generic pharmaceutical market while maintaining cost competitiveness. The environmental burden of disposing of cyanide-containing waste streams further complicates regulatory compliance, posing a long-term liability for producers.
The Novel Approach
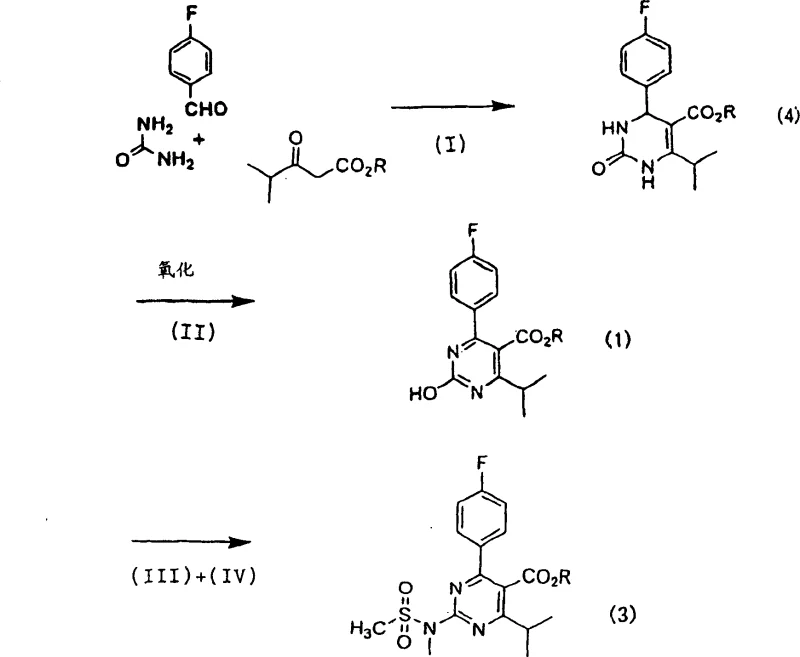
In stark contrast, the methodology outlined in CN1301977C circumvents these critical pitfalls by employing a Biginelli-type multicomponent condensation followed by oxidative aromatization. This novel approach initiates with the reaction of isobutyl acetate, 4-fluorobenzaldehyde, and urea in the presence of a protic compound and a metal salt, effectively building the dihydropyrimidinone core without toxic cyanating agents. The subsequent oxidation step utilizes nitric acid, a common and manageable oxidant, to convert the dihydro-intermediate into the desired hydroxypyrimidine with high efficiency. This shift in synthetic strategy not only enhances operator safety by eliminating carcinogenic gases but also improves the overall atom economy of the process. The final functionalization steps involve activation with organic sulfonyl halides and subsequent amination, which proceed under controllable conditions to deliver the target 2-(N-methyl-N-methylsulfonylamino)pyrimidine in high purity. This comprehensive redesign of the synthetic pathway offers a viable solution for cost reduction in pharmaceutical intermediates manufacturing by streamlining operations and reducing hazard mitigation costs.
Mechanistic Insights into Metal-Catalyzed Condensation and Oxidation
The core of this innovative synthesis lies in the initial cyclization step, which resembles a modified Biginelli reaction mechanism facilitated by Lewis acid metal salts. In this stage, isobutyl acetate reacts with 4-fluorobenzaldehyde and urea, where the metal salt, preferably copper(I) chloride or iron(III) chloride, acts as a catalyst to promote the formation of the carbon-nitrogen bonds required for the pyrimidine ring. The presence of a protic compound, such as sulfuric acid or p-toluenesulfonic acid, is crucial for activating the carbonyl groups and facilitating the dehydration steps that drive the equilibrium towards the dihydropyrimidinone product. Experimental data suggests that optimizing the molar ratio of the metal salt to the ester substrate, typically between 0.01 to 0.1 mol per mole of ester, is key to maximizing conversion rates. This catalytic system allows the reaction to proceed at moderate temperatures ranging from 30 to 100°C, preventing thermal degradation of sensitive functional groups and ensuring a clean reaction profile that simplifies downstream purification.
Following the cyclization, the oxidative aromatization step is critical for establishing the fully conjugated pyrimidine system required for biological activity. This transformation is achieved using nitric acid, which serves as a potent dehydrogenating agent to remove hydrogen atoms from the dihydropyrimidinone ring. The mechanism likely involves the formation of nitrosonium ions or similar reactive nitrogen species that abstract hydrides, leading to the formation of the aromatic heterocycle and water as a byproduct. The patent highlights that controlling the concentration of nitric acid, preferably between 50% to 70%, and maintaining the reaction temperature between 0 to 50°C is essential to prevent over-oxidation or nitration of the aromatic ring. This precise control ensures that the hydroxypyrimidine intermediate is formed with minimal side products, preserving the integrity of the fluorophenyl and ester substituents. The high yields observed in this step, often exceeding 90%, underscore the robustness of this oxidative protocol for large-scale applications.
How to Synthesize 2-(N-methyl-N-methylsulfonylamino)pyrimidine Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to ensure reproducibility and high quality, particularly when transitioning from laboratory benchtop to pilot plant scales. The process begins with the condensation of the beta-keto ester equivalent, aldehyde, and urea, followed by isolation of the solid dihydropyrimidinone precipitate which can be purified by simple washing. The subsequent oxidation is exothermic and requires efficient cooling systems to maintain the specified low-temperature range, ensuring safety and selectivity. Finally, the activation of the hydroxyl group and the nucleophilic displacement by the sulfonamide moiety must be conducted in anhydrous conditions with appropriate bases like potassium carbonate or sodium tert-amylate to drive the reaction to completion. Detailed standard operating procedures regarding reagent addition rates, stirring speeds, and workup protocols are essential for maintaining the stringent purity specifications required for API intermediates. For a complete breakdown of the specific operational steps and conditions validated in the patent examples, please refer to the technical guide below.
- Condense isobutyl acetate with 4-fluorobenzaldehyde and urea using a metal salt catalyst like copper(I) chloride and a protic acid.
- Oxidize the resulting dihydropyrimidinone intermediate using concentrated nitric acid to form the hydroxypyrimidine derivative.
- Activate the hydroxyl group with a sulfonyl halide and react with N-methyl-N-methanesulfonamide in the presence of a base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers substantial strategic benefits that extend beyond mere chemical efficiency. By eliminating the need for cyanogen chloride, facilities can significantly reduce the costs associated with specialized safety infrastructure, hazardous waste disposal, and regulatory compliance monitoring. This simplification of the safety profile translates directly into lower overheads and reduced insurance premiums, contributing to a more favorable cost structure for the final intermediate. Furthermore, the use of commodity chemicals like nitric acid and common metal salts ensures a stable supply chain that is less susceptible to geopolitical disruptions or vendor monopolies that often plague specialty reagent markets. The high yields reported in the patent examples indicate a drastic reduction in raw material consumption per kilogram of product, which inherently lowers the variable cost of goods sold and improves margin potential for manufacturers.
- Cost Reduction in Manufacturing: The elimination of toxic cyanogen chloride removes the necessity for expensive scrubbing systems and specialized containment vessels, leading to significant capital expenditure savings. Additionally, the high yield of the condensation step, reported at approximately 85%, minimizes the loss of valuable starting materials like 4-fluorobenzaldehyde, thereby optimizing material utilization rates. The ability to perform the final substitution steps in common solvents like acetonitrile or butyl acetate further reduces solvent recovery costs compared to more exotic solvent systems. These cumulative efficiencies result in a leaner manufacturing process that is highly competitive in price-sensitive generic drug markets.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved as the key reagents, including urea, aldehydes, and nitric acid, are globally available bulk chemicals with multiple qualified suppliers. This diversification of the supply base mitigates the risk of production stoppages due to single-source failures or logistics bottlenecks. The robustness of the reaction conditions, which tolerate a range of temperatures and pressures, ensures consistent output even when minor variations in utility supplies occur. Consequently, manufacturers can offer more reliable lead times and maintain higher inventory levels of finished intermediates, providing greater security to downstream API producers who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the feasibility of scaling this process to 200L reactors, proving its readiness for commercial production without extensive re-engineering. The waste streams generated are primarily aqueous acids and organic solvents which are easier to treat and recycle compared to heavy metal or cyanide wastes. This alignment with green chemistry principles facilitates smoother environmental permitting and reduces the long-term ecological footprint of the manufacturing site. Companies adopting this technology can leverage these sustainability credentials to meet corporate social responsibility goals while ensuring uninterrupted production capacity.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into existing production lines. The following questions address common inquiries regarding safety, scalability, and quality control based on the data provided in the patent documentation. These insights are derived directly from the experimental examples and comparative analysis presented in the intellectual property filing, offering a transparent view of the technology's capabilities. By clarifying these points, we aim to facilitate informed decision-making for technical teams assessing the feasibility of this route for their specific portfolio needs.
Q: Why is this synthesis method safer than previous art like WO01/04100?
A: This method eliminates the use of highly toxic cyanogen chloride, which was required in prior art processes, significantly improving operational safety and reducing hazardous waste handling requirements.
Q: What yields can be expected from this patented process?
A: Experimental data indicates high efficiency, with the initial condensation step achieving yields around 85%, oxidation steps reaching over 90%, and the final amination step yielding approximately 86%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability with examples conducted in 200L glass-lined reactors, proving the feasibility of commercial scale-up for pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(N-methyl-N-methylsulfonylamino)pyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a supply of high-quality intermediates for the production of life-saving cardiovascular medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical partners. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of 2-(N-methyl-N-methylsulfonylamino)pyrimidine meets the highest industry standards. Our facility is equipped to handle the specific safety requirements of oxidative and substitution chemistries, guaranteeing a safe and compliant manufacturing environment that protects both our workforce and the environment.
We invite you to collaborate with us to optimize your supply chain for Rosuvastatin intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. Please contact us to request specific COA data and route feasibility assessments that demonstrate how our implementation of patent CN1301977C can enhance your production efficiency. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable partner dedicated to driving innovation and reliability in the pharmaceutical supply chain.
