Advanced Synthesis of NF-kappaB Inhibitors DHM2EQ and DHM3EQ for Commercial Scale-up
Advanced Synthesis of NF-kappaB Inhibitors DHM2EQ and DHM3EQ for Commercial Scale-up
The pharmaceutical landscape is constantly evolving towards targeted therapies that minimize systemic side effects, and patent CN1185212C represents a significant breakthrough in this domain by disclosing novel salicylamide derivatives known as DHM2EQ and DHM3EQ. These compounds exhibit potent inhibitory activity against NF-kappaB activation, a central transcription factor involved in immune responses and inflammation, positioning them as superior alternatives to traditional steroidal agents. The technical depth of this patent provides a robust framework for the industrial production of these high-value pharmaceutical intermediates, detailing a convergent synthetic strategy that balances chemical complexity with operational feasibility. For R&D directors and procurement specialists, understanding the nuances of this synthesis is critical for securing a reliable supply chain for next-generation immunosuppressive drugs. The disclosed methodology not only ensures high structural fidelity but also offers multiple pathways to optimize yield and purity, making it an attractive candidate for commercial scale-up in the competitive fine chemical sector.
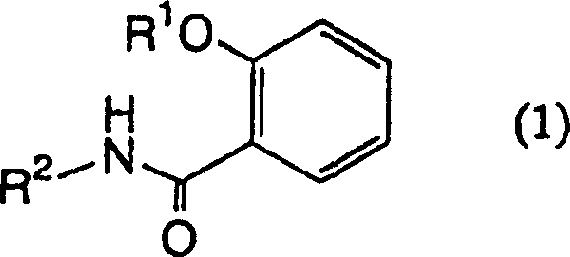
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of anti-inflammatory and immunosuppressive agents has been dominated by steroidal compounds and calcineurin inhibitors like cyclosporine and FK506, which, while effective, are plagued by severe long-term side effects and narrow therapeutic windows. Conventional synthetic routes for complex heterocyclic systems often rely on harsh conditions, heavy metal catalysts, or non-selective reactions that generate difficult-to-remove impurities, complicating the purification process and increasing manufacturing costs. Furthermore, many existing scaffolds lack the specific structural motifs required to selectively inhibit NF-kappaB without affecting other essential cellular pathways, leading to off-target toxicity. The reliance on multi-step sequences with low overall yields in traditional methods further exacerbates supply chain vulnerabilities, making it challenging to secure consistent quality for clinical and commercial demands. These limitations necessitate a paradigm shift towards more precise, atom-economical, and scalable synthetic methodologies that can deliver novel chemical entities with improved safety profiles.
The Novel Approach
The innovative strategy outlined in the patent leverages a sophisticated sequence involving hypervalent iodine chemistry and selective reductions to construct the unique epoxy-cyclohexenone core found in DHM2EQ and DHM3EQ. Unlike traditional methods that might struggle with regioselectivity, this approach utilizes diacetoxyiodobenzene to effect a controlled oxidative transformation of the aromatic ring, creating a reactive dienone intermediate that serves as a versatile pivot point for further functionalization. This method allows for the precise installation of the epoxide moiety and the hydroxyl groups essential for biological activity, ensuring that the final product meets stringent purity specifications required for pharmaceutical applications. By avoiding transition metal catalysts in key bond-forming steps, the process simplifies downstream processing and reduces the risk of heavy metal contamination, a critical consideration for regulatory compliance. The flexibility of this route, which branches from a common intermediate to produce two distinct active compounds, maximizes resource efficiency and provides manufacturers with strategic options to meet diverse market needs.
Mechanistic Insights into Hypervalent Iodine Oxidation and Epoxidation
The core of this synthetic innovation lies in the mechanistic elegance of the oxidative spirocyclization and subsequent epoxidation steps, which transform a simple aniline derivative into a highly functionalized bioactive scaffold. The reaction begins with the formation of an amide bond between 2,5-dimethoxyaniline and an O-alkanoylsalicyloyl halide, establishing the fundamental connectivity of the molecule. Subsequent treatment with diacetoxyiodobenzene in a protic solvent like methanol triggers a single-electron transfer or ligand coupling mechanism that oxidizes the electron-rich aromatic ring to a cyclohexadienone system. This step is critical as it introduces the oxygen functionality at the 4-position while maintaining the integrity of the amide linkage, setting the stage for the introduction of chirality and three-dimensional complexity. The careful control of temperature and stoichiometry during this oxidation is paramount to prevent over-oxidation or decomposition, highlighting the need for precise process engineering during scale-up.
Following the formation of the dienone intermediate, the pathway diverges into an epoxidation sequence using alkaline hydrogen peroxide, which selectively targets the electron-deficient double bond of the cyclohexenone ring. This nucleophilic epoxidation proceeds with high stereoselectivity, generating the 5,6-epoxy motif that is crucial for the compound's interaction with the NF-kappaB pathway. The mechanism likely involves the formation of a hydroperoxide anion that attacks the beta-carbon of the enone system, followed by ring closure to form the epoxide. Subsequent steps involve either acidic deprotection to reveal the diketone system or selective reduction with sodium borohydride to install the allylic alcohol functionality. Understanding these mechanistic details allows chemists to troubleshoot potential impurities, such as ring-opened byproducts or over-reduced species, ensuring that the final API intermediate maintains the structural integrity required for potent biological activity.
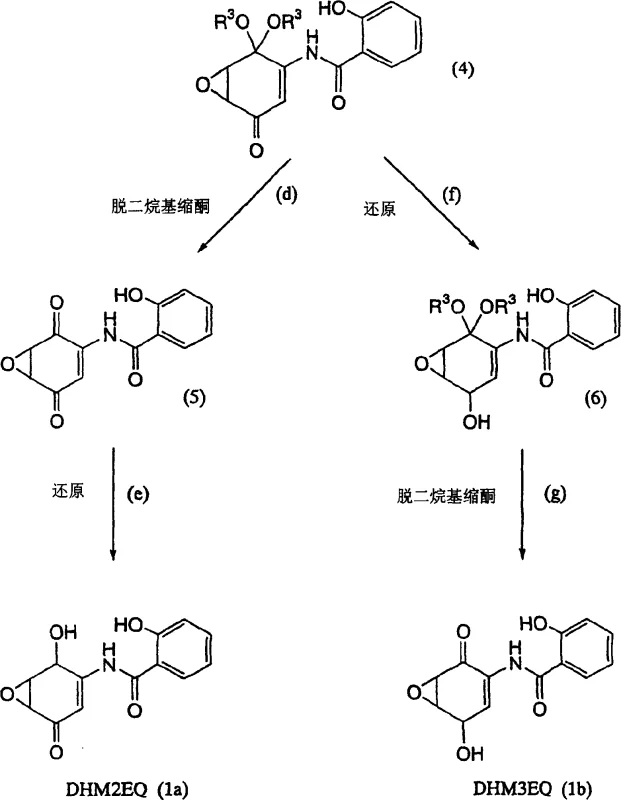
How to Synthesize DHM2EQ Efficiently
The synthesis of DHM2EQ and its analog DHM3EQ follows a logical progression from readily available starting materials, utilizing standard unit operations that are well-suited for kilogram-scale production. The process initiates with the acylation of 2,5-dimethoxyaniline, followed by the pivotal oxidative rearrangement to form the spiro-dienone core, which acts as the central hub for the entire synthetic tree. From this common intermediate, the pathway splits: one branch leads to DHM2EQ via epoxidation, deprotection, and selective ketone reduction, while the other leads to DHM3EQ through a slightly different reduction and deprotection sequence. Each step has been optimized in the patent examples to provide isolable solids with defined melting points and spectral characteristics, facilitating quality control at every stage. The detailed experimental procedures provided in the patent serve as a blueprint for process chemists to adapt these conditions to larger reactors, ensuring that heat transfer and mixing efficiencies are maintained to preserve reaction selectivity.
- Prepare N-(2-alkanoyloxybenzoyl)-2,5-dimethoxyaniline by reacting 2,5-dimethoxyaniline with O-alkanoylsalicyloyl chloride in pyridine at low temperatures.
- Perform oxidative transformation using diacetoxyiodobenzene in methanol to generate the 4,4-dialkoxy-2,5-cyclohexadienone intermediate.
- Execute epoxidation with hydrogen peroxide followed by selective reduction or deprotection steps to yield the final DHM2EQ or DHM3EQ compounds.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits in terms of cost stability and supply continuity, primarily driven by the use of commodity chemicals and the avoidance of exotic reagents. The starting materials, such as 2,5-dimethoxyaniline and salicylic acid derivatives, are widely available from global chemical suppliers, reducing the risk of raw material shortages that often plague specialized pharmaceutical syntheses. Furthermore, the elimination of expensive transition metal catalysts in favor of hypervalent iodine reagents, which can often be recycled or managed effectively, significantly lowers the cost of goods sold and simplifies waste disposal protocols. The ability to isolate stable intermediates allows for a modular manufacturing approach, where stockpiles of key precursors can be maintained to buffer against demand fluctuations, thereby enhancing the overall resilience of the supply chain. This strategic flexibility ensures that downstream drug manufacturers can rely on a consistent flow of high-quality intermediates without facing the volatility associated with more fragile synthetic routes.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by utilizing inexpensive oxidants and avoiding the need for complex chiral resolution steps, as the stereochemistry is controlled through substrate-directed reactions. By streamlining the purification workflow to rely on crystallization and standard chromatography rather than preparative HPLC, the operational expenditure is drastically reduced, making the final intermediates more price-competitive in the global market. Additionally, the high yields reported in the patent examples for key transformation steps indicate a material-efficient process that minimizes waste generation and maximizes the throughput of the production facility. These factors collectively contribute to a lower total cost of ownership for partners sourcing these critical building blocks for anti-inflammatory drug development.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route, characterized by mild reaction conditions and stable intermediates, translates directly into improved delivery performance and reduced lead times for customers. Since the chemistry does not depend on sensitive organometallic reagents that require strict inert atmospheres or cryogenic temperatures, the manufacturing process is less susceptible to equipment failures or utility interruptions. This operational stability allows the supplier to maintain higher inventory levels of finished goods and respond rapidly to urgent procurement requests, ensuring that clinical trials and commercial production schedules are never compromised by material delays. The scalability of the process from gram to multi-kilogram quantities further guarantees that supply can expand seamlessly alongside the growing demand for NF-kappaB inhibitors.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, the synthesis aligns well with green chemistry principles by minimizing the use of hazardous solvents and generating manageable aqueous waste streams. The workup procedures described, which involve standard extractions and washes with bicarbonate and brine, are easily adaptable to industrial separation equipment, ensuring compliance with increasingly strict environmental discharge regulations. The absence of heavy metal residues in the final product simplifies the regulatory filing process for drug developers, as extensive testing for metal impurities is not required, accelerating the time to market for new therapeutic candidates. This commitment to sustainable manufacturing practices not only mitigates environmental risk but also enhances the corporate social responsibility profile of the entire value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these salicylamide derivatives, providing clarity for stakeholders evaluating this technology. The answers are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these aspects is essential for integrating these intermediates into broader drug discovery pipelines and manufacturing strategies.
Q: What is the primary biological mechanism of DHM2EQ and DHM3EQ?
A: These salicylamide derivatives function as potent inhibitors of NF-kappaB activation, offering a novel mechanism for anti-inflammatory and immunosuppressive therapy distinct from traditional steroids.
Q: Are the synthetic intermediates stable enough for isolation?
A: Yes, key intermediates such as the 5,6-epoxy-4,4-dialkoxy-3-salicyloylamino-2-cyclohexenone are isolable solids that can be purified via column chromatography or crystallization, ensuring process control.
Q: What purification methods are recommended for the final API intermediates?
A: The patent describes purification through silica gel column chromatography and subsequent washing with solvents like methanol or ethyl acetate to achieve high-purity white solid products suitable for pharmaceutical use.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DHM2EQ Supplier
As a leader in the fine chemical industry, NINGBO INNO PHARMCHEM is uniquely positioned to support the global demand for advanced pharmaceutical intermediates like DHM2EQ and DHM3EQ through our state-of-the-art manufacturing capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet the highest standards of quality and consistency. Our rigorous QC labs employ advanced analytical techniques to verify stringent purity specifications, guaranteeing that every batch of salicylamide derivative performs reliably in downstream biological assays and formulation processes. By partnering with us, pharmaceutical companies can leverage our technical expertise to navigate the complexities of process optimization and regulatory compliance with confidence.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and project timelines. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing solutions can accelerate your drug development programs. Whether you are in the early stages of preclinical research or preparing for commercial launch, NINGBO INNO PHARMCHEM is committed to being your strategic partner in delivering high-performance chemical solutions that drive innovation in healthcare.
