Advanced Manufacturing of 2-Hydroxybiphenyl: Technical Upgrades and Commercial Scalability
Introduction to Novel Synthetic Pathways
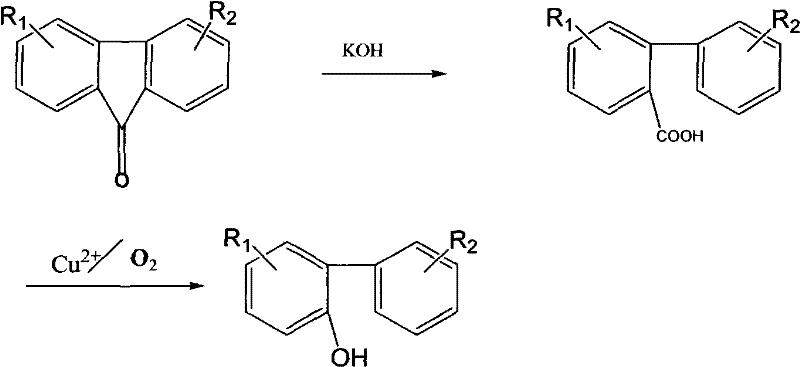
The chemical industry is constantly seeking more efficient routes for producing key intermediates like 2-hydroxybiphenyl, a vital component in fungicides, preservatives, and polymer modifiers. A significant breakthrough in this domain is detailed in patent CN101955415A, which outlines a robust method for preparing 2-hydroxybiphenyl compounds starting from readily available 9-fluorenone derivatives. This technology represents a paradigm shift away from traditional, costly, and impurity-prone methods, offering a streamlined two-step sequence that combines base-mediated ring opening with copper-catalyzed oxidative decarboxylation. For R&D directors and procurement specialists, understanding this pathway is crucial as it promises not only higher yields but also a dramatic simplification of the supply chain by utilizing commodity chemicals instead of specialized amines. The strategic value of this patent lies in its ability to deliver high-purity products suitable for sensitive applications in pharmaceuticals and agrochemicals while maintaining economic viability through the use of air as the terminal oxidant.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 2-hydroxybiphenyl has been plagued by significant technical and economic hurdles that limit scalability and product quality. One traditional method involves recovering the compound from the distillation residues of phenolic production via sulfonation; however, this approach suffers from inherent purity issues due to the unavoidable presence of the 4-hydroxybiphenyl isomer, which is difficult to separate and detrimental to downstream applications. Another existing route relies on the gas-phase dehydrogenation of substituted cyclohexenylcyclohexanone over metal-loaded alumina, a process that is energetically intensive and dependent on expensive, specialized raw materials that strain procurement budgets. Furthermore, the diazotization of 2-aminobiphenyl followed by hydrolysis, while chemically feasible, is characterized by low overall yields and the handling of hazardous diazonium salts, creating safety liabilities and waste disposal challenges that modern manufacturing facilities strive to avoid. These legacy methods collectively result in higher production costs, inconsistent quality, and supply chain vulnerabilities that hinder the reliable sourcing of high-purity intermediates.
The Novel Approach
In stark contrast to these legacy technologies, the novel approach described in the patent utilizes 9-fluorenone as a strategic starting material, leveraging its widespread availability and low cost to drive down manufacturing expenses. The process initiates with a highly efficient ring-opening reaction using potassium hydroxide to generate biphenyl-2-carboxylic acid intermediates with exceptional conversion rates, effectively bypassing the isomer contamination issues of sulfonation routes. The subsequent step employs a catalytic oxidative decarboxylation using inexpensive divalent copper salts and air, eliminating the need for stoichiometric oxidants or precious metal catalysts that typically inflate production costs. This methodology not only achieves superior yields, as evidenced by experimental data showing conversions exceeding 86% in the oxidation step, but also operates under conditions that are amenable to large-scale industrial implementation without the extreme energy penalties of gas-phase dehydrogenation. By integrating these steps, the new route offers a cleaner, safer, and more economically attractive alternative for the commercial scale-up of complex biaryl compounds.
Mechanistic Insights into Copper-Catalyzed Oxidative Decarboxylation
The core of this innovative synthesis lies in the mechanistic elegance of the copper-catalyzed transformation, which converts the carboxylic acid intermediate into the desired phenol functionality. The reaction proceeds through a mechanism where the divalent copper species, such as copper chloride or copper acetate, facilitates the activation of molecular oxygen from the air stream at elevated temperatures ranging from 220°C to 300°C. This thermal activation promotes the formation of reactive copper-oxygen species that interact with the carboxylate group, leading to the extrusion of carbon dioxide and the simultaneous installation of the hydroxyl group at the ortho-position of the biphenyl system. The use of air as the oxidant is particularly advantageous from a green chemistry perspective, as it generates water as the primary byproduct alongside CO2, minimizing the environmental footprint compared to methods requiring harsh chemical oxidants. Furthermore, the choice of copper salt loading between 1% and 5% by mass strikes an optimal balance between catalytic activity and cost efficiency, ensuring that the reaction proceeds to completion within a practical timeframe of 1 to 3 hours without requiring excessive catalyst quantities.
Controlling impurities in this process is achieved through the specificity of the initial ring-opening step and the robustness of the subsequent oxidation. The base-mediated cleavage of the 9-fluorenone ketone bridge is highly regioselective, ensuring that the resulting carboxylic acid possesses the correct substitution pattern required for the final product, thereby preventing the formation of meta- or para-substituted byproducts that plague sulfonation methods. During the workup phase, the protocol specifies the removal of inorganic copper salts via filtration and the use of activated carbon for decolorization, which effectively strips away trace organic impurities and residual catalyst species. This purification strategy, combined with recrystallization from ethanol, allows manufacturers to achieve purity levels exceeding 99%, meeting the stringent specifications required for pharmaceutical intermediates and high-performance agrochemical active ingredients. The mechanistic clarity of this pathway provides R&D teams with a predictable and controllable process window for optimization.
How to Synthesize 2-Hydroxybiphenyl Efficiently
The synthesis of 2-hydroxybiphenyl via this patented route is designed for operational simplicity and high throughput, making it an ideal candidate for technology transfer and process validation. The procedure begins with the preparation of the biphenyl-2-carboxylic acid intermediate by reacting 9-fluorenone with solid potassium hydroxide in toluene at reflux temperatures, followed by acidic workup to isolate the acid in near-quantitative yields. The subsequent oxidative decarboxylation is performed in a melt or high-boiling solvent system where the acid is mixed with a catalytic amount of copper salt and heated under a steady stream of air. Detailed standardized operating procedures regarding specific stirring rates, gas flow dynamics, and crystallization parameters are critical for maximizing yield and ensuring batch-to-batch consistency. For a comprehensive guide on the exact molar ratios, temperature ramping profiles, and isolation techniques validated in the patent examples, please refer to the structured synthesis guide below.
- Perform base-mediated ring opening of 9-fluorenone using KOH in toluene at 112°C to generate biphenyl-2-carboxylic acid.
- Add divalent copper salt (1-5% mass ratio) to the carboxylic acid intermediate and heat to 220-300°C.
- Pass air through the reaction mixture for 1-3 hours to effect oxidative decarboxylation, followed by ethanol recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this fluorenone-based synthesis route offers transformative benefits that directly impact the bottom line and operational resilience. By shifting away from reliance on 2-aminobiphenyl or specialized cyclohexenyl precursors, companies can leverage the abundant global supply of 9-fluorenone, a commodity chemical produced in vast quantities for other industries, thereby securing a stable and cost-effective raw material base. The elimination of hazardous diazotization steps and the use of air instead of stoichiometric oxidants significantly reduce the costs associated with waste treatment, safety compliance, and raw material consumption, leading to substantial overall cost savings in fine chemical intermediates manufacturing. Moreover, the high selectivity of the reaction minimizes the formation of difficult-to-remove isomers, reducing the burden on purification units and increasing the effective capacity of existing production assets without the need for major capital expenditure on new separation equipment.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by the replacement of expensive amine starting materials with low-cost fluorenone derivatives and the utilization of air as a free oxidant. By avoiding the use of precious metal catalysts or complex reagents, the variable cost per kilogram of the final product is drastically reduced, allowing for more competitive pricing in the global market. Additionally, the high yield of the initial ring-opening step ensures that raw material utilization is maximized, further enhancing the cost efficiency of the overall process and providing a buffer against fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: Sourcing 9-fluorenone is significantly less risky than procuring specialized amines or residues from sulfonation processes, as it is a standard industrial chemical with multiple suppliers worldwide. This diversification of the supply base mitigates the risk of shortages and price spikes, ensuring continuous production schedules and reliable delivery to customers. The robustness of the synthesis also means that production can be scaled up rapidly to meet surges in demand without the long lead times typically associated with setting up complex multi-step syntheses involving hazardous intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard reactor configurations capable of handling high-temperature melt reactions or high-boiling solvents, which facilitates the commercial scale-up of complex biaryl compounds. From an environmental standpoint, the generation of benign byproducts like CO2 and water, coupled with the absence of heavy metal waste streams typical of other catalytic systems, simplifies regulatory compliance and reduces the environmental liability of the manufacturing site. This alignment with green chemistry principles not only lowers waste disposal costs but also enhances the corporate sustainability profile of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and industry best practices. Understanding these details is essential for evaluating the feasibility of adopting this route for your specific production needs. The answers provided reflect the technical realities of the process as described in the intellectual property documentation.
Q: What are the primary advantages of using 9-fluorenone over 2-aminobiphenyl for this synthesis?
A: The use of 9-fluorenone eliminates the need for expensive 2-aminobiphenyl starting materials and avoids the hazardous diazotization process, resulting in significantly lower raw material costs and improved operational safety.
Q: How does this method address the purity issues associated with sulfonation residues?
A: Unlike the recovery from sulfonation distillation residues which inevitably contains 4-hydroxybiphenyl impurities, this synthetic route builds the molecule from a clean fluorenone precursor, ensuring high regioselectivity and purity levels exceeding 99%.
Q: Is the copper catalyst recoverable in this oxidative decarboxylation process?
A: The process involves filtering off inorganic matter after the reaction. While the patent specifies removing inorganic salts, standard industrial practices for heterogeneous or semi-heterogeneous copper catalysis often allow for catalyst recovery or recycling strategies to further minimize waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxybiphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global fine chemicals market. Our team of expert chemists has extensively evaluated the fluorenone oxidation pathway and possesses the technical capability to implement this route with precision and efficiency. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require pilot quantities for clinical trials or metric tons for commercial manufacturing, our facilities are equipped to deliver. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this cost-effective and high-yielding technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can drive value and reliability for your organization.
