Scalable Synthesis of 3-Bromo-5-Chlorophenol via Diazonium Hydrolysis for Commercial API Production
Introduction to Advanced Synthetic Methodology
The pharmaceutical industry continuously seeks robust and cost-effective pathways for critical organic intermediates, particularly those serving as backbones for antiviral and neurodegenerative therapeutics. Patent CN101735023A introduces a significant technological advancement in the preparation of 3-bromo-5-chlorophenol, a pivotal building block utilized in the synthesis of non-nucleoside reverse transcriptase inhibitors and treatments for Alzheimer's disease. This patent discloses a streamlined method that circumvents the historical limitations of high cost and complex operations associated with previous synthetic routes. By leveraging a diazonium salt hydrolysis strategy, the process achieves superior productivity and purity while utilizing easily accessible raw materials. The structural integrity of the target molecule, as depicted below, requires precise halogen positioning which this new methodology addresses with high regioselectivity.
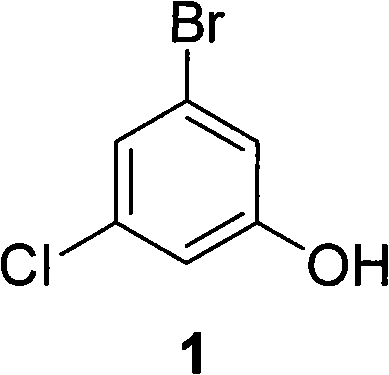
For global procurement teams and R&D directors, understanding the shift from legacy methods to this novel approach is essential for securing a reliable 3-bromo-5-chlorophenol supplier. The technology not only promises enhanced supply chain stability but also aligns with modern green chemistry principles by reducing energy consumption and waste generation. As market demand for HIV and cognitive dysfunction medications grows, the ability to manufacture this intermediate efficiently becomes a strategic asset. The following analysis dissects the technical merits and commercial implications of this patented process, providing a comprehensive view for stakeholders involved in fine chemical manufacturing and API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-bromo-5-chlorophenol has been plagued by inefficient and hazardous methodologies that hinder large-scale adoption. One prominent prior art route, documented in J.Am.Chem.Soc., relies on the ozonolysis of chloro-bromobenzene derivatives followed by esterification with expensive borane reagents. This approach is fundamentally flawed for industrial application due to the high energy requirements of ozonization and the necessity for specialized, novel apparatus that increases capital expenditure. Furthermore, the side reactions inherent in ozonolysis often lead to lower yields and complex impurity profiles that are difficult to purge. Another traditional pathway involves the Sandmeyer reaction starting from 3-amino-5-chlorophenol, a raw material that is notoriously difficult to source commercially and commands a premium price, thereby inflating the overall cost of goods.
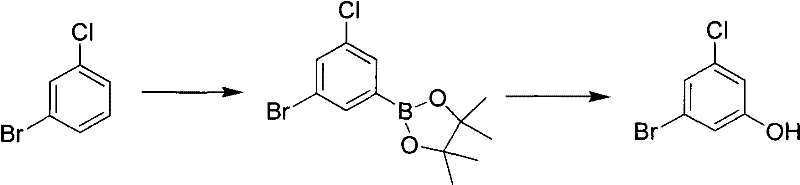
Additionally, alternative strategies involving nucleophilic aromatic substitution on fluorinated precursors, such as 3-chloro-5-bromofluorobenzene, present their own set of challenges. These methods typically suffer from low yields and require multi-step sequences involving methoxy group replacement followed by demethylation under harsh acidic conditions. The cumulative effect of these inefficiencies is a synthetic bottleneck that limits the availability of high-purity material for downstream drug development. For procurement managers, these legacy routes represent significant supply chain risks, including volatile pricing and potential discontinuation of key starting materials. The reliance on such cumbersome chemistries underscores the urgent need for a more direct and economically viable manufacturing process.
The Novel Approach
In stark contrast to the convoluted legacy methods, the novel approach detailed in CN101735023A offers a direct and highly efficient pathway via the hydrolysis of a diazonium intermediate. This method initiates with the reduction of readily available 3-bromo-5-chloronitrobenzene to the corresponding aniline, followed by diazotization and subsequent hydrolysis in an acidic medium. The elegance of this route lies in its simplicity and the use of commodity chemicals such as sulfuric acid and sodium nitrite, which drastically reduces raw material costs. The reaction conditions are mild yet effective, operating within a temperature range of 60°C to 150°C, which is easily manageable in standard stainless steel reactors without the need for cryogenic or high-pressure equipment. This accessibility translates directly into cost reduction in pharmaceutical intermediates manufacturing, making the process attractive for both laboratory scale-up and full commercial production.
Furthermore, the novel approach demonstrates exceptional versatility regarding the acidic medium used for hydrolysis, accommodating various ratios of aqueous sulfuric acid mixed with organic inert solvents like toluene or chloroform. This flexibility allows process chemists to optimize solubility and reaction kinetics to maximize yield, which has been reported to reach impressive levels in pilot studies. The workup procedure is equally straightforward, requiring only standard extraction, concentration, and filtration steps to isolate the product with high purity. By eliminating the need for exotic reagents and complex purification protocols, this methodology establishes a new benchmark for the commercial scale-up of complex pharmaceutical intermediates. It effectively resolves the long-standing issues of yield and operability that have constrained the supply of this critical molecule.
Mechanistic Insights into Diazonium Salt Hydrolysis
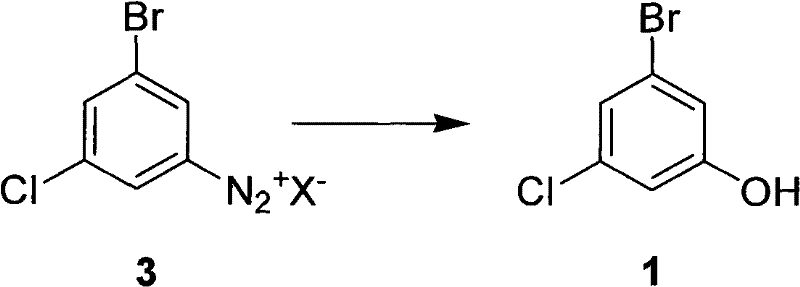
The core of this innovative synthesis lies in the precise control of the diazotization and hydrolysis mechanisms, which dictate the final quality of the 3-bromo-5-chlorophenol. The process begins with the formation of the diazonium salt (Compound 3) from 3-bromo-5-chloroaniline (Compound 2) under strongly acidic conditions. The stability of this diazonium intermediate is paramount; the patent specifies maintaining the reaction temperature between -5°C and 10°C during the addition of sodium nitrite to prevent premature decomposition. The choice of acid anion (X-) plays a crucial role, with chloride, bromide, bisulfate, or nitrate ions all being viable, though sulfuric acid is preferred for its dual role as both solvent and catalyst. This careful control ensures that the diazonium species remains intact until it is introduced into the hydrolysis medium, minimizing the formation of tar-like byproducts that often plague azo-coupling side reactions.
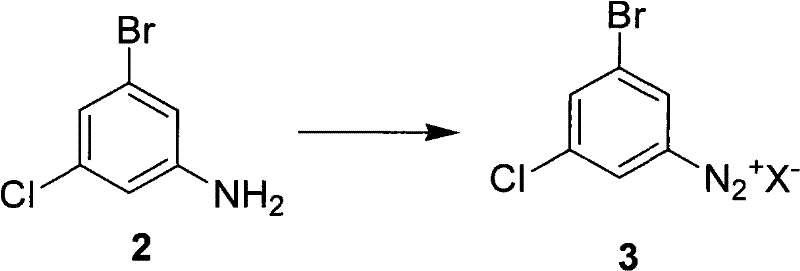
Upon heating the diazonium solution in the acidic hydrolysis medium, the nitrogen molecule is expelled, generating a highly reactive aryl cation that is immediately trapped by water to form the phenolic hydroxyl group. The patent highlights that the concentration of the acid and the presence of organic co-solvents significantly influence the rate of this substitution. By optimizing the molar ratio of acid to substrate (preferably 8 to 12 times), the reaction environment promotes the desired hydrolysis over competing pathways such as reduction or coupling. Impurity control is rigorously managed through HPLC monitoring, ensuring that the reaction is driven to completion before workup. This mechanistic understanding allows for the consistent production of material with purity exceeding 98%, meeting the stringent specifications required for GMP-grade API synthesis. The robustness of this mechanism against variations in raw material quality further enhances its suitability for industrial application.
How to Synthesize 3-Bromo-5-Chlorophenol Efficiently
The synthesis of 3-bromo-5-chlorophenol via this patented route is designed for operational simplicity, making it an ideal candidate for technology transfer from R&D to manufacturing. The process generally follows a logical sequence: first, the reduction of the nitro precursor to the aniline using iron powder or catalytic hydrogenation; second, the conversion of the aniline to the diazonium salt using sodium nitrite in acid; and finally, the thermal hydrolysis of the salt to yield the phenol. Each step has been optimized to minimize handling time and maximize throughput. For detailed technical execution, operators should refer to the specific molar ratios and temperature profiles outlined in the patent examples, which demonstrate reproducibility across different scales. The standardized nature of these steps ensures that the detailed standardized synthesis steps see the guide below can be implemented with minimal training.
- Reduction of 3-bromo-5-chloronitrobenzene to 3-bromo-5-chloroaniline using iron powder or catalytic hydrogenation.
- Diazotization of the resulting aniline derivative with sodium nitrite under strong acidic conditions at low temperature.
- Hydrolysis of the diazonium salt in an acidic medium at elevated temperatures (60-150°C) to yield the final phenol product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers profound advantages for procurement and supply chain management, primarily driven by the simplification of the value chain. The elimination of expensive and hazardous reagents like borane complexes and ozone generators removes significant cost drivers from the manufacturing equation. Instead, the process relies on bulk commodities such as nitrobenzenes, mineral acids, and sodium nitrite, which are widely available from multiple global suppliers. This shift in raw material strategy mitigates the risk of supply disruptions and price volatility, ensuring a more stable cost structure for long-term contracts. For procurement managers, this means the ability to negotiate better terms and secure a reliable 3-bromo-5-chlorophenol supplier who can guarantee continuity of supply even during market fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this process are substantial, stemming from the drastic simplification of the reaction sequence and the use of low-cost inputs. By avoiding the need for specialized equipment required for ozonolysis or high-pressure hydrogenation with noble metals, capital investment is significantly lowered. The operational expenses are further reduced due to the shorter reaction times and the ability to recycle solvents like toluene or chloroform. Additionally, the high yield and purity achieved reduce the burden on downstream purification units, lowering utility consumption and waste disposal costs. These factors combine to create a highly competitive cost profile that allows for significant margin improvement in the final API production.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials fundamentally strengthens the supply chain resilience. Unlike routes dependent on custom-synthesized fluorinated intermediates or scarce amino-phenols, the nitro-precursor used here is a standard chemical with a robust global supply network. This accessibility reduces lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond quickly to surges in demand. Furthermore, the scalability of the process means that production volumes can be ramped up rapidly without the need for extensive process re-engineering. For supply chain heads, this translates to reduced inventory holding costs and a lower risk of stockouts, ensuring that downstream drug manufacturing schedules are met without interruption.
- Scalability and Environmental Compliance: The environmental footprint of this method is markedly lower than conventional alternatives, aligning with increasingly strict global regulatory standards. The absence of heavy metal catalysts and the minimization of hazardous waste streams simplify the effluent treatment process, reducing the cost and complexity of environmental compliance. The process is inherently scalable, having been demonstrated to work efficiently from gram to multi-kilogram scales in the patent examples. This scalability ensures that the transition from pilot plant to commercial production is smooth and predictable. For organizations focused on sustainability, adopting this greener synthetic route enhances corporate social responsibility profiles while simultaneously driving operational efficiency and reducing the total cost of ownership.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 3-bromo-5-chlorophenol using the methodology described in CN101735023A. These answers are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of this route for potential partners. Understanding these details is crucial for making informed decisions about sourcing and process development. The responses cover aspects ranging from raw material selection to purity assurance, providing a comprehensive overview of the technology's capabilities.
Q: What are the primary advantages of the diazonium hydrolysis route over ozonolysis methods?
A: The diazonium hydrolysis route described in CN101735023A eliminates the need for expensive borane reagents and energy-intensive ozonolysis equipment, significantly reducing operational costs and safety risks associated with high-energy intermediates.
Q: How is product purity controlled in this synthesis method?
A: The process utilizes HPLC monitoring to ensure complete consumption of reactants during both the reduction and hydrolysis steps, consistently achieving purity levels exceeding 98% after simple extraction and crystallization.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method uses readily available raw materials like 3-bromo-5-chloronitrobenzene and common acids, avoiding complex purification steps, which makes it highly adaptable for commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Bromo-5-Chlorophenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our team of expert chemists has extensively evaluated the synthetic route disclosed in CN101735023A and confirmed its viability for large-scale manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of material. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to verify that every batch meets stringent purity specifications, typically exceeding 98% by HPLC. We are committed to delivering excellence in every aspect of our service, from initial process validation to final delivery.
We invite pharmaceutical companies and contract research organizations to collaborate with us to leverage this cost-effective technology for their pipeline projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your drug development timelines with our premium grade 3-bromo-5-chlorophenol.
