Advanced Manufacturing of 4-Amino-3-Quinolinecarbonitriles for Kinase Inhibitor Development
Introduction to Next-Generation Kinase Inhibitor Intermediates
The pharmaceutical industry's relentless pursuit of potent protein kinase (PK) inhibitors has placed significant demand on the supply chain for specialized heterocyclic intermediates. Patent CN1835923A introduces a transformative methodology for the preparation of 4-amino-3-quinolinecarbonitriles and 7-amino-thieno[3,2-b]pyridine-6-carbonitriles, compounds that serve as critical scaffolds in the development of therapeutics for polycystic kidney disease, colon polyps, cancer, and stroke. As a reliable pharmaceutical intermediate supplier, understanding the nuances of this patented process is essential for R&D directors seeking to optimize their synthetic routes. The disclosed technology moves away from historically problematic high-temperature cyclizations, offering a robust pathway that ensures high purity and operational simplicity. By leveraging specific condensation and cyclodehydration techniques, this process addresses the long-standing stability issues associated with amino-quinoline species, providing a commercially viable solution for the mass production of complex API precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-amino-substituted quinolines relied heavily on intramolecular Friedel-Crafts reactions of N-(2-carboxyvinyl)-aniline derivatives or electrocyclization ring closures. These conventional pathways are fraught with significant technical challenges that hinder efficient commercial scale-up. Primarily, these reactions necessitate extremely high temperatures, which often lead to the decomposition of sensitive functional groups and the formation of undesirable side products. Literature indicates that under such harsh thermal conditions, the desired amino-quinoline species are frequently unstable, leading to a preference for chloro-quinoline formation instead. Furthermore, attempts to utilize Vilsmeier-type intermediates for cyclodehydration have been documented as lacking robustness, often failing to achieve complete conversion or requiring complex purification steps to isolate the target molecule from a mixture of regioisomers and degradation products. For procurement managers, these inefficiencies translate into higher costs of goods sold (COGS) and unpredictable lead times due to low yields and batch-to-batch variability.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a温和 (mild) yet highly effective sequence involving cyanoacetamide condensation followed by phosphorus oxychloride-mediated cyclization. This method circumvents the thermal instability issues of previous generations by operating at moderate temperatures ranging from 80°C to 110°C. The process begins with the facile preparation of a cyanoacetamide moiety from cyanoacetic acid and the desired aniline, utilizing either acid catalysis or peptide coupling reagents. This intermediate is then condensed with a substituted aniline in the presence of a trialkyl orthoformate to generate a 3-amino-2-cyanoacrylamide. The final ring closure is achieved through dehydration with phosphorus oxychloride in solvents such as acetonitrile, butyronitrile, toluene, or xylene. This strategic shift allows for the isolation of product salts directly from the reaction mixture, which can be neutralized to yield the final high-purity quinoline nitrile. 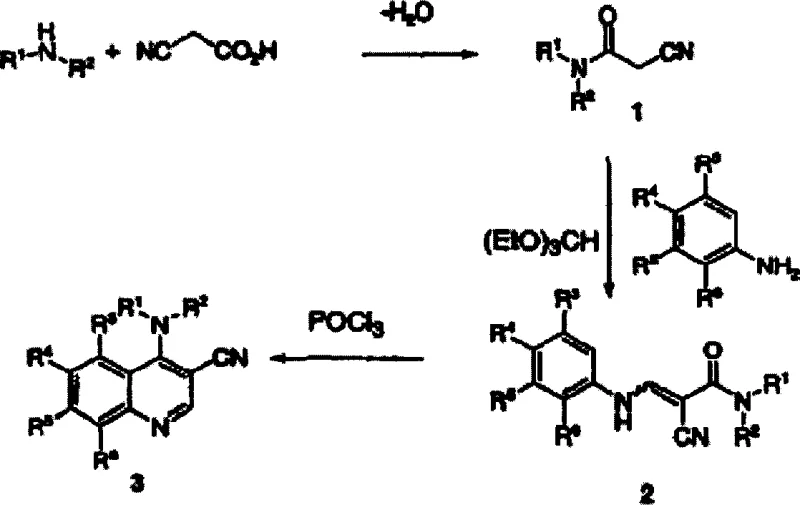
Mechanistic Insights into POCl3-Mediated Cyclodehydration
The core of this technological advancement lies in the precise control of the cyclodehydration mechanism. The reaction proceeds through the activation of the amide carbonyl by phosphorus oxychloride, facilitating an intramolecular nucleophilic attack by the aromatic ring. Unlike traditional methods that might rely on brute force thermal energy, this chemical pathway leverages the electrophilicity of the phosphorylated intermediate to drive ring closure under kinetic control. The use of solvents like acetonitrile or toluene is critical, as they provide the necessary polarity to solubilize the reactants while allowing the product salt to precipitate upon completion. This precipitation phenomenon is a key mechanistic feature that drives the equilibrium forward and simplifies downstream processing. For R&D teams, understanding this mechanism is vital for troubleshooting; for instance, the addition of tertiary amine bases such as pyridine or triethylamine can provide significant rate acceleration by scavenging the HCl byproduct, thereby preventing acid-catalyzed degradation of the sensitive amino group.
Impurity control is inherently built into the reaction design through the physical properties of the intermediates. The initial formation of the cyanoacetamide allows for a filtration step where urea byproducts (from coupling reagents like DCC or DIC) are physically removed before the condensation step. This prevents the carryover of nitrogenous impurities into the final cyclization vessel, which could otherwise complicate purification. Furthermore, the condensation with trialkyl orthoformate generates water as a byproduct, which is managed by the choice of solvent and reaction temperature to prevent hydrolysis of the nitrile group. The result is a process that consistently delivers intermediates with purity specifications exceeding 96% to 99% as measured by HPLC, minimizing the burden on analytical QC labs and ensuring that the material is suitable for subsequent medicinal chemistry transformations without further chromatographic purification.
How to Synthesize 4-Amino-3-Quinolinecarbonitrile Efficiently
The synthesis protocol outlined in the patent offers a streamlined workflow that balances reaction efficiency with safety and scalability. The process is divided into three distinct operational phases: amide formation, enaminonitrile generation, and final cyclization. Each phase is optimized to maximize yield while minimizing the use of hazardous reagents and excessive solvent volumes. The following guide summarizes the critical operational parameters derived from the patent examples, serving as a foundational reference for process chemists aiming to replicate or adapt this route for specific analogues. Detailed standardized synthesis steps are provided in the section below to ensure reproducibility and compliance with GMP standards.
- Prepare cyanoacetamide by combining an amine compound with cyanoacetic acid and a peptide coupling reagent or acid catalyst, followed by filtration to remove byproducts.
- Condense the cyanoacetamide with a substituted aniline and trialkyl orthoformate in an alcoholic solvent to yield a 3-amino-2-cyanoacrylamide intermediate.
- Perform cyclodehydration by reacting the acrylamide intermediate with phosphorus oxychloride in a solvent like acetonitrile or toluene at 80-110°C to form the final quinoline nitrile.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic advantages beyond mere chemical elegance. The primary benefit lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures. By eliminating the need for high-temperature reactors capable of withstanding the extreme conditions of Friedel-Crafts reactions, facilities can utilize standard glass-lined or stainless steel equipment, thereby lowering capital expenditure requirements. Additionally, the ability to isolate intermediates via simple filtration rather than energy-intensive distillation or complex chromatography significantly reduces utility costs and processing time. This efficiency translates into a more resilient supply chain, as the reliance on specialized, hard-to-source catalysts is minimized in favor of commodity chemicals like cyanoacetic acid and phosphorus oxychloride.
- Cost Reduction in Manufacturing: The process achieves cost optimization through the elimination of expensive transition metal catalysts often required in cross-coupling alternatives and the reduction of solvent consumption due to high-concentration reaction conditions. The direct precipitation of products allows for minimal workup procedures, reducing the labor hours and solvent waste associated with multiple extraction and washing steps. Furthermore, the high yields reported in the patent examples, often exceeding 80%, mean that less raw material is required to produce the same amount of API intermediate, directly lowering the variable cost per kilogram. This economic efficiency makes the process highly attractive for cost reduction in pharmaceutical intermediate manufacturing, especially when scaling from pilot plant to commercial tonnage.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of widely available starting materials. Cyanoacetic acid, various substituted anilines, and trialkyl orthoformates are commodity chemicals with stable global supply chains, reducing the risk of bottlenecks associated with exotic reagents. The robustness of the reaction conditions, which tolerate a range of substituents on the aniline ring, allows for flexibility in sourcing; if one supplier of a specific substituted aniline faces disruption, the process can often accommodate alternate sources without extensive re-validation. This flexibility ensures reducing lead time for high-purity pharmaceutical intermediates, allowing drug developers to maintain their clinical trial timelines without interruption due to material shortages.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process is designed for safe scale-up. The exothermic nature of the initial coupling reaction is managed through controlled addition and cooling, preventing thermal runaways that could jeopardize plant safety. The use of solvents like isopropanol and acetonitrile, while requiring proper handling, allows for efficient recovery and recycling systems, aligning with modern green chemistry principles. The reduction in heavy metal waste and the minimization of aqueous waste streams through direct filtration contribute to lower environmental compliance costs. This makes the commercial scale-up of complex pharmaceutical intermediates not only technically feasible but also environmentally sustainable, meeting the rigorous ESG criteria demanded by modern multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity for technical teams evaluating the feasibility of this route for their specific projects. Understanding these details is crucial for making informed decisions about process adoption and vendor selection.
Q: What are the advantages of this cyclodehydration method over traditional Friedel-Crafts reactions?
A: Unlike traditional intramolecular Friedel-Crafts reactions which require high temperatures and often favor chloro-quinoline formation, this novel process utilizes mild cyclodehydration conditions (80-110°C) with phosphorus oxychloride. This results in higher stability of the amino-quinoline species and significantly improved yields without the need for harsh thermal conditions.
Q: How is purity controlled during the cyanoacetamide formation step?
A: The process employs water-soluble peptide coupling reagents or specific acid catalysts that allow the cyanoacetamide product to precipitate directly upon water treatment. The solid byproducts, such as urea derivatives, are removed via filtration before the subsequent condensation step, ensuring the intermediate enters the cyclization phase with high purity (>97%).
Q: Can this process be scaled for commercial production of kinase inhibitor intermediates?
A: Yes, the patent details scalable conditions using common solvents like isopropanol, acetonitrile, and toluene. The exothermic nature of the coupling reaction is managed by controlled addition and cooling (maintaining temperatures below 15°C initially), making it suitable for large-scale manufacturing with standard reactor equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Amino-3-Quinolinecarbonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful development of kinase inhibitors depends on the consistent quality and availability of key building blocks. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market supply is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4-amino-3-quinolinecarbonitrile meets the exacting standards required for GMP API synthesis. Our commitment to technical excellence means we can navigate the complexities of this cyclodehydration chemistry to deliver material that accelerates your drug development timeline.
We invite you to engage with our technical procurement team to discuss your specific requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how optimizing this synthetic route can impact your overall project budget. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecule. Let us collaborate to secure your supply chain and drive your innovative therapies forward with confidence.
