Advanced Manufacturing of Ethylene Glycol Sulfonamide Intermediates for Cardiovascular Therapeutics
The pharmaceutical industry continuously seeks robust synthetic routes for complex active pharmaceutical ingredients, particularly for cardiovascular therapeutics like endothelin receptor antagonists. Patent CN1178921C introduces a groundbreaking methodology for the preparation of 1,2-diheteroethylene sulfonamides, a critical structural motif found in drugs such as Bosentan. This innovation addresses long-standing challenges in process chemistry by utilizing monoprotected 1,2-diheteroethylene anions to react with pyrimidine monohalides, significantly streamlining the production workflow. The core of this technology lies in the strategic manipulation of protecting groups and solvent systems to enhance both safety and efficiency. By shifting away from traditional glycol-based solvents, the method offers a cleaner reaction profile that is highly attractive for industrial scale-up. 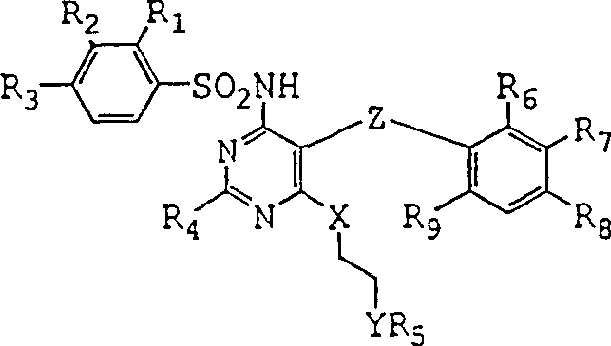 This structural versatility allows for the synthesis of various derivatives where R groups can be tailored for specific biological activities, ensuring that the process remains relevant across multiple drug development pipelines. For a reliable pharmaceutical intermediates supplier, mastering such patented methodologies is essential to meeting the rigorous demands of global regulatory bodies.
This structural versatility allows for the synthesis of various derivatives where R groups can be tailored for specific biological activities, ensuring that the process remains relevant across multiple drug development pipelines. For a reliable pharmaceutical intermediates supplier, mastering such patented methodologies is essential to meeting the rigorous demands of global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ethylene glycol sulfonamide derivatives has been plagued by significant chemical and operational inefficiencies that hinder large-scale manufacturing. Conventional protocols typically rely on reacting pyrimidine monohalides with monoanionic ethylene glycol, often using ethylene glycol itself as the reaction solvent. This approach suffers from a critical flaw: the formation of unwanted ethylene glycol bis-sulfonamides, where two molecules of the pyrimidine halide couple with a single glycol molecule. This side reaction creates difficult-to-remove impurities that necessitate costly and laborious isolation steps to achieve pharmaceutically acceptable purity levels. Furthermore, the use of ethylene glycol as a solvent presents severe downstream processing challenges due to its high boiling point and toxicity. Removing this solvent requires substantial energy input and time, making the process economically unviable for commercial production. Additionally, traditional routes often require the isolation of pyrimidine dihalide intermediates, which are identified as strong sensitizers, posing significant occupational health risks and requiring specialized handling equipment. These cumulative factors result in a process with low atom economy, high waste generation, and extended lead times, creating a bottleneck for the cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The methodology described in the patent represents a paradigm shift by introducing a monoprotected strategy that fundamentally alters the reaction landscape. Instead of using free ethylene glycol, the process employs monoprotected 1,2-diheteroethylene anions, such as those protected with tert-butyl groups. This simple yet profound modification sterically and chemically blocks the second hydroxyl group, effectively eliminating the possibility of bis-sulfonamide formation. Consequently, the reaction selectivity is drastically improved, leading to higher crude yields and simplified purification workflows. Moreover, the process transitions to non-polar aprotic solvents like toluene, tetrahydrofuran, or 2-methyltetrahydrofuran. These solvents are not only easier to recover and recycle via distillation but also facilitate better heat transfer and mixing in large reactors. A key feature of this novel approach is the telescoping of reaction steps, allowing the pyrimidine dihalide to be generated and consumed in situ without isolation. This avoids exposing personnel to hazardous sensitizers and reduces the overall number of unit operations. 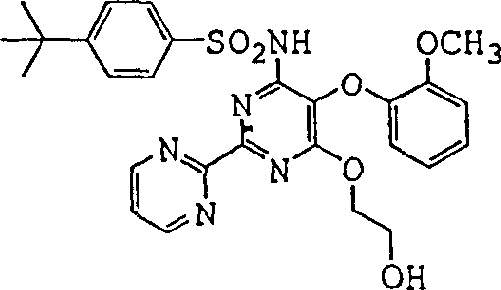 As illustrated by the structure of Bosentan, the final product requires precise substitution patterns that this method delivers with high fidelity. For procurement teams, this translates to a more stable supply chain with reduced dependency on complex purification infrastructure.
As illustrated by the structure of Bosentan, the final product requires precise substitution patterns that this method delivers with high fidelity. For procurement teams, this translates to a more stable supply chain with reduced dependency on complex purification infrastructure.
Mechanistic Insights into Telescoped Nucleophilic Substitution
The chemical elegance of this process is rooted in a carefully orchestrated sequence of nucleophilic substitutions and deprotection events that maximize efficiency while minimizing hazard. The synthesis initiates with the conversion of a pyrimidine dione, such as the structure shown in 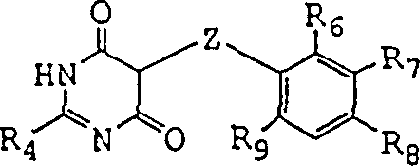 , into a reactive pyrimidine dihalide using a dehydrohalogenating reagent like phosphorus oxychloride. Crucially, this highly reactive and sensitizing dihalide is not isolated; instead, the reaction mixture is quenched and directly subjected to coupling with a sulfonamide in the presence of a base and a phase transfer catalyst. This in-situ generation ensures that the concentration of the hazardous dihalide remains low, mitigating safety risks. The subsequent step involves the nucleophilic attack of the monoprotected glycol anion on the resulting pyrimidine monohalide. The use of a strong base like sodium hydroxide generates the alkoxide anion efficiently in the non-polar medium. The protecting group, typically a tert-butyl ether, remains stable under these basic conditions but is designed to be labile under acidic conditions later in the sequence. This orthogonality is vital for maintaining the integrity of the intermediate during the coupling phase. The final deprotection is achieved using mild organic acids like formic acid, which cleaves the ether to reveal the free hydroxyl group, often passing through a formyloxy intermediate that is easily hydrolyzed. This mechanistic pathway ensures that impurities are kept to a minimum, satisfying the stringent purity specifications required for high-purity pharmaceutical intermediates.
, into a reactive pyrimidine dihalide using a dehydrohalogenating reagent like phosphorus oxychloride. Crucially, this highly reactive and sensitizing dihalide is not isolated; instead, the reaction mixture is quenched and directly subjected to coupling with a sulfonamide in the presence of a base and a phase transfer catalyst. This in-situ generation ensures that the concentration of the hazardous dihalide remains low, mitigating safety risks. The subsequent step involves the nucleophilic attack of the monoprotected glycol anion on the resulting pyrimidine monohalide. The use of a strong base like sodium hydroxide generates the alkoxide anion efficiently in the non-polar medium. The protecting group, typically a tert-butyl ether, remains stable under these basic conditions but is designed to be labile under acidic conditions later in the sequence. This orthogonality is vital for maintaining the integrity of the intermediate during the coupling phase. The final deprotection is achieved using mild organic acids like formic acid, which cleaves the ether to reveal the free hydroxyl group, often passing through a formyloxy intermediate that is easily hydrolyzed. This mechanistic pathway ensures that impurities are kept to a minimum, satisfying the stringent purity specifications required for high-purity pharmaceutical intermediates.
How to Synthesize Ethylene Glycol Sulfonamide Efficiently
Implementing this synthesis requires precise control over reaction parameters to ensure the telescoped steps proceed without accumulation of byproducts. The process begins with the chlorination of the pyrimidine dione at elevated temperatures, followed by a controlled quench to destroy excess reagents. The subsequent coupling with the sulfonamide is facilitated by potassium carbonate and a phase transfer catalyst like tetrabutylammonium bromide in toluene, with water removal via azeotropic distillation driving the equilibrium forward. Once the monohalide is formed, the addition of the monoprotected glycol and sodium hydroxide completes the carbon-oxygen bond formation. The final deprotection and hydrolysis steps are conducted under mild acidic and basic conditions respectively, ensuring the final product is obtained with minimal degradation. This streamlined protocol exemplifies how modern process chemistry can simplify complex molecular architectures.
- Convert pyrimidine dione to dihalide using phosphorus oxychloride without isolation.
- React dihalide with sulfonamide in toluene with phase transfer catalyst to form monohalide.
- Couple monohalide with monoprotected ethylene glycol anion followed by deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented process offers substantial strategic advantages for organizations focused on cost efficiency and supply reliability. The elimination of the bis-sulfonamide impurity is perhaps the most significant value driver, as it removes the need for expensive chromatographic separations or multiple recrystallizations that typically erode profit margins. By preventing this side reaction at the source, the overall yield of the desired intermediate is significantly increased, directly lowering the cost of goods sold. Furthermore, the switch from high-boiling ethylene glycol to volatile non-polar solvents like toluene dramatically reduces energy consumption during solvent recovery. Distilling toluene requires far less thermal energy and time compared to stripping glycol, leading to faster batch cycle times and increased reactor throughput. This efficiency gain allows manufacturers to respond more agilely to market demand fluctuations. In terms of supply chain resilience, the avoidance of isolating strong sensitizers like pyrimidine dihalides simplifies logistics and storage requirements. Facilities do not need specialized containment for hazardous intermediates, reducing compliance costs and insurance premiums. The robustness of the chemistry also implies a lower risk of batch failures, ensuring a consistent flow of materials to downstream API manufacturers. For partners seeking cost reduction in pharmaceutical intermediates manufacturing, this technology provides a clear pathway to optimized operational expenditures without compromising on quality.
- Cost Reduction in Manufacturing: The process inherently lowers production costs by eliminating the formation of difficult-to-separate bis-sulfonamide impurities, thereby reducing the need for extensive purification steps and increasing overall material throughput. The use of readily available, low-cost solvents like toluene further decreases raw material expenses compared to specialized glycol solvents. Additionally, the telescoped nature of the synthesis reduces the number of isolation steps, saving on filtration media, drying time, and labor costs associated with handling multiple intermediates.
- Enhanced Supply Chain Reliability: By avoiding the isolation of hazardous sensitizing intermediates, the process minimizes safety-related shutdowns and regulatory hurdles that can disrupt supply continuity. The use of common industrial solvents ensures that raw material availability is not a bottleneck, as these chemicals are widely sourced globally. The simplified workflow also reduces the complexity of the manufacturing schedule, allowing for more predictable lead times and better inventory management for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The transition to non-polar solvents facilitates easier solvent recovery and recycling, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing process. The lower energy requirements for solvent removal make the process highly scalable from pilot plant to commercial tonnage without significant engineering redesigns. Furthermore, the reduction in waste generation from fewer purification steps simplifies effluent treatment, ensuring compliance with increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this route for their specific projects. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation. These insights help clarify the operational benefits and chemical rationale behind the process improvements.
Q: How does this process prevent bis-sulfonamide impurity formation?
A: By using a monoprotected ethylene glycol derivative, only one hydroxyl group is available for nucleophilic attack, chemically preventing the formation of the unwanted bis-sulfonamide byproduct.
Q: Why is avoiding the isolation of pyrimidine dihalides beneficial?
A: Pyrimidine dihalides are known strong sensitizers. Telescoping the reaction avoids worker exposure and eliminates the costly purification steps associated with handling hazardous intermediates.
Q: What solvents are preferred for this synthesis?
A: The process utilizes non-polar aprotic solvents like toluene, which are easier to remove via distillation compared to high-boiling ethylene glycol used in conventional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethylene Glycol Sulfonamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe manufacturing processes for complex cardiovascular intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patented routes like CN1178921C are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch meets the highest international standards. Our capability to handle telescoped reactions and hazardous intermediates safely positions us as a preferred partner for global pharmaceutical companies. We understand that consistency and quality are paramount in the supply of active pharmaceutical ingredients and their precursors.
We invite you to engage with our technical procurement team to discuss how we can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this optimized synthesis route. We encourage potential partners to contact us for specific COA data and route feasibility assessments to ensure seamless integration into your supply chain. Let us collaborate to bring life-saving medications to market faster and more efficiently.
