Advanced Manufacturing of 2-Ethylaminopyridine Derivatives for High-Performance Agrochemicals
Advanced Manufacturing of 2-Ethylaminopyridine Derivatives for High-Performance Agrochemicals
The global demand for high-efficiency agrochemicals continues to drive innovation in intermediate synthesis, particularly for heterocyclic compounds that serve as the backbone of modern fungicides and insecticides. Patent CN101080390A introduces a transformative methodology for the preparation of 2-ethylaminopyridine derivatives, addressing critical bottlenecks in yield and process complexity that have historically plagued this sector. This technical disclosure outlines a robust, scalable route that converts readily available 2-halopyridine derivatives into valuable ethylamine intermediates through a streamlined sequence of alkylation, catalytic hydrogenation, and hydrolysis. For R&D directors and procurement specialists, understanding the nuances of this patented pathway is essential for securing a competitive edge in the reliable agrochemical intermediate supplier market. The process not only enhances purity profiles but also drastically simplifies the operational workflow, eliminating the need for isolation of unstable intermediates that often degrade during traditional multi-step syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, specifically referenced in patent application WO 2004/016088, relied on a disjointed synthetic strategy that suffered from severe efficiency losses at the reduction stage. In these conventional protocols, the conversion of 2-methylcyanopyridine derivatives to the corresponding 2-ethylaminopyridine derivatives was achieved via reduction in a protic solvent with a metal catalyst, yet the reported yields were unacceptably low for commercial viability. Historical data indicates that such methods often resulted in overall yields as low as 19%, rendering the process economically prohibitive for large-scale cost reduction in pesticide manufacturing. Furthermore, the traditional approach necessitated two distinct, separate steps to first generate the 2-methylcyanopyridine precursor from the halopyridine starting material, compounding the time, energy, and solvent consumption required. These inefficiencies created significant supply chain vulnerabilities, as low throughput and high waste generation made it difficult to maintain consistent inventory levels for downstream drug substance production.
The Novel Approach
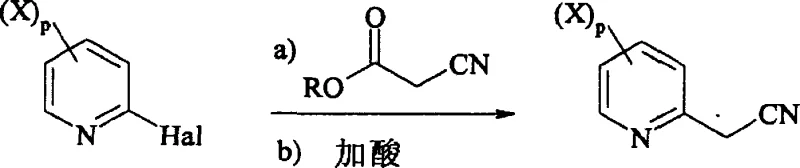
The methodology presented in CN101080390A overcomes these historical deficiencies by integrating the alkylation and functionalization steps into a cohesive, high-yielding sequence. By reacting 2-halopyridine derivatives directly with alkyl cyanoacetate in a polar solvent under basic conditions, followed by immediate acidification, the process achieves the formation of the nitrile intermediate with exceptional efficiency. Crucially, the subsequent reduction step is performed in the presence of an acylating agent, which protects the newly formed amine in situ, preventing side reactions and facilitating easier purification. This strategic modification boosts the yield of the reduction step to a range of 65% to 95%, a dramatic improvement that validates the process for commercial scale-up of complex agrochemical intermediates. The ability to operate this synthesis on an industrial scale without compromising on purity or throughput represents a paradigm shift for manufacturers seeking to optimize their production lines for next-generation crop protection agents.
Mechanistic Insights into Catalytic Hydrogenation and Acylation
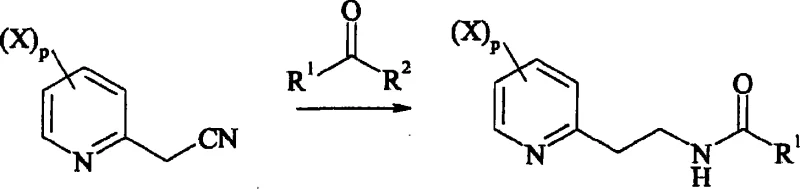
The core of this innovative synthesis lies in the sophisticated management of the reduction-acylation cascade, which transforms the nitrile functionality into a protected ethylamine moiety. In the second step of the reaction scheme, the 2-methylcyanopyridine derivative undergoes catalytic hydrogenation under a controlled hydrogen pressure of 4 to 40 bar. Unlike standard reductions that might stop at the primary amine or lead to over-reduction, the presence of an acylating agent, such as acetic anhydride, ensures that the intermediate imine or amine is immediately trapped as a stable amide. This mechanism utilizes metal catalysts like palladium on carbon (Pd/C) to facilitate the addition of hydrogen across the triple bond of the nitrile group while simultaneously promoting nucleophilic attack by the acylating agent. The result is a highly selective transformation that minimizes the formation of secondary amines or polymeric byproducts, which are common impurities in reductive amination processes. This level of control is vital for maintaining the high-purity agrochemical intermediate standards required by regulatory bodies for final pesticide registration.
Following the formation of the N-[2-(2-pyridyl)ethyl]carboxamide derivative, the final step involves a straightforward hydrolysis to reveal the free amine. This hydrolysis is conducted by adding a mineral acid, such as hydrochloric acid or sulfuric acid, in molar equivalents ranging from 1 to 20 relative to the substrate. The reaction mixture is heated to temperatures between 20°C and reflux, effectively cleaving the amide bond to release the target 2-ethylaminopyridine derivative. The mechanistic elegance of this step ensures that any remaining protecting groups or side products are converted into water-soluble salts, which can be easily separated from the organic phase during workup. This robustness in the final deprotection stage contributes significantly to the overall mass balance of the process, ensuring that the theoretical yield improvements seen in the earlier steps are realized in the final isolated product. Such mechanistic reliability is a key factor for supply chain heads evaluating the risk profile of adopting new synthetic routes for critical raw materials.
How to Synthesize 2-Ethylaminopyridine Derivatives Efficiently
Implementing this patented synthesis requires precise control over reaction parameters, particularly regarding stoichiometry and pH management during the initial alkylation phase. The process begins with the reaction of the halopyridine substrate with an alkyl cyanoacetate, where the molar ratio of base to substrate must be carefully maintained between 1 and 4 to ensure complete conversion without excessive degradation. Operators must monitor the pH closely during the acidification step, aiming for a range of 1 to 5 to precipitate or stabilize the nitrile intermediate before proceeding to hydrogenation. The detailed standardized synthesis steps, including specific solvent choices like NMP or DMSO and temperature profiles for each stage, are critical for replicating the high yields reported in the patent documentation. For technical teams looking to validate this route in their pilot plants, adhering to these specific operational windows is the key to unlocking the full economic potential of this technology.
- React 2-halopyridine derivatives with alkyl cyanoacetate in a polar solvent with a base, followed by acidification to pH 1-5 to obtain 2-methylcyanopyridine derivatives.
- Perform catalytic reduction of the nitrile group under 4-40 bar hydrogen pressure in the presence of an acylating agent and metal catalyst to form the protected amine.
- Hydrolyze the resulting amide intermediate using mineral acid at reflux temperatures to yield the final 2-ethylaminopyridine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound benefits that extend beyond simple yield improvements, impacting the total cost of ownership for agrochemical manufacturers. By consolidating what were previously inefficient, low-yielding steps into a streamlined process, companies can achieve substantial cost savings through reduced raw material consumption and lower waste disposal fees. The elimination of transition metal catalysts in certain variations, or the efficient recovery of palladium catalysts in others, further reduces the burden on downstream purification units, thereby shortening the overall production cycle time. For procurement managers, this translates into a more predictable pricing structure for key intermediates, shielding the business from volatility associated with inefficient legacy manufacturing methods. The ability to source these materials from a reliable agrochemical intermediate supplier who utilizes this optimized technology ensures a steady flow of materials essential for meeting seasonal agricultural demands.
- Cost Reduction in Manufacturing: The significant increase in yield from roughly 19% to over 65% fundamentally alters the cost basis of the final product, allowing for much more competitive pricing in the global market. By reducing the number of discrete unit operations and minimizing the loss of valuable pyridine scaffolds during synthesis, the process inherently lowers the variable cost per kilogram of output. Additionally, the use of common, commodity chemicals such as acetic anhydride and standard mineral acids avoids the need for exotic or prohibitively expensive reagents that often inflate the BOM (Bill of Materials) costs in fine chemical synthesis. This economic efficiency enables manufacturers to reinvest savings into quality control or capacity expansion, strengthening their market position against competitors relying on outdated technologies.
- Enhanced Supply Chain Reliability: The robustness of this chemical pathway means that production schedules are less susceptible to disruptions caused by failed batches or off-spec material. Since the reaction conditions are well-defined and tolerant of various substituents on the pyridine ring, suppliers can maintain consistent output even when switching between different grades of the intermediate to meet specific customer formulations. This flexibility is crucial for reducing lead time for high-purity agrochemical intermediates, as it allows for rapid response to urgent orders without the need for extensive process re-validation. Furthermore, the reliance on stable, shelf-stable starting materials ensures that raw material inventory can be managed more effectively, reducing the risk of supply shortages due to precursor degradation.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial scale operation, with parameters such as hydrogen pressure and temperature set within ranges that are safe and manageable in standard stainless steel reactors. The improved atom economy resulting from higher yields means less chemical waste is generated per unit of product, aligning with increasingly stringent environmental regulations and sustainability goals. Waste streams are simpler to treat, primarily consisting of aqueous salt solutions and recoverable organic solvents, which simplifies the permitting process for manufacturing facilities. This environmental compatibility not only reduces compliance costs but also enhances the brand reputation of the end-product as being manufactured through greener, more responsible chemical processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the specifications and examples provided in the patent literature. Understanding these details helps stakeholders assess the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the specific conditions and outcomes observed during the development of the patent, providing a realistic baseline for expected performance.
Q: What is the primary advantage of this synthesis method over prior art?
A: The method disclosed in CN101080390A significantly improves the overall yield from approximately 19% in previous methods to between 65% and 95%, making it viable for industrial scale-up.
Q: Can this process be adapted for different pyridine substitutions?
A: Yes, the process is highly versatile and accommodates various substituents (X) at multiple positions on the pyridine ring, including halogens, alkyl groups, and trifluoromethyl groups.
Q: What catalysts are recommended for the hydrogenation step?
A: The patent specifies the use of palladium on carbon (Pd/C) as the preferred catalyst, though Raney nickel and platinum-based catalysts are also suitable alternatives depending on substrate sensitivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Ethylaminopyridine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of effective crop protection solutions. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of 2-ethylaminopyridine derivatives meets the exacting standards required for agrochemical applications. Our commitment to process excellence means we can leverage the advancements described in CN101080390A to deliver superior products that empower your R&D efforts.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthesis routes can reduce your overall production costs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your unique chemical needs, ensuring a partnership built on transparency, quality, and mutual growth.
