Advanced Synthetic Routes for Indazole Compounds: Enhancing Commercial Scalability and Purity
The pharmaceutical industry continuously seeks robust synthetic methodologies for complex heterocyclic scaffolds, particularly those serving as potent protein kinase inhibitors. Patent CN101052633A presents a significant advancement in the preparation of indazole compounds of Formula 1, which are critical modulators for targets such as VEGF-R, FGF-R, and CDK complexes. These molecules hold immense therapeutic potential for treating cancer and diseases associated with angiogenesis. The disclosed methods address the longstanding need for efficient, cost-effective, and scalable routes that overcome the limitations of earlier syntheses described in prior art like US Patents 6,534,524 and 6,531,491. By leveraging advanced transition metal catalysis and strategic protection group chemistry, this technology enables the production of high-purity intermediates suitable for commercial drug substance manufacturing.
Traditional methods for synthesizing substituted indazoles often suffered from poor regioselectivity, harsh reaction conditions, and difficulties in controlling stereochemistry at the C-3 position. Earlier routes frequently resulted in complex mixtures of isomers or required extensive purification steps that diminished overall yield and increased production costs. Furthermore, the removal of heavy metal catalysts, particularly palladium, to meet stringent regulatory limits was often inefficient, posing risks to supply chain continuity and product safety. The novel approach detailed in this patent introduces a streamlined sequence involving Pd-catalyzed cross-coupling and Heck reactions that significantly mitigate these issues. By utilizing specific ligand systems and optimized reaction parameters, the process achieves superior conversion rates and simplifies the isolation of the desired pharmacological active structures.
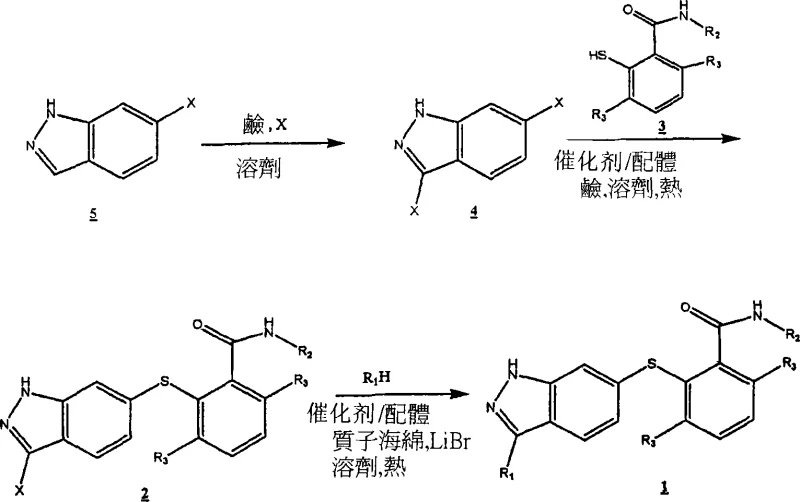
The core of this innovative methodology lies in the precise execution of palladium-catalyzed coupling reactions. As illustrated in the general scheme, the synthesis begins with the coupling of a halogenated indazole (Formula 4) with a thiol-containing benzamide (Formula 3). This step is critical for establishing the core sulfanyl linkage. The reaction is facilitated by catalysts such as Pd2(dba)3 complexed with bulky phosphine ligands like Xantphos, which enhance oxidative addition and reductive elimination rates. The use of strong bases like CsOH in polar aprotic solvents such as NMP or DMF at temperatures around 70°C to 80°C ensures complete consumption of starting materials. This mechanistic precision minimizes the formation of des-halo impurities and bis-coupled byproducts, thereby enhancing the purity profile of the intermediate Formula 2 before it proceeds to the final functionalization step.
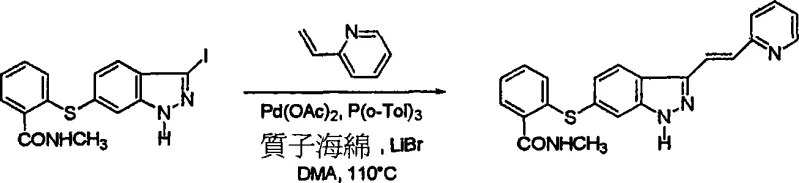
Following the initial coupling, the installation of the vinyl heteroaryl side chain is achieved through a Heck reaction, a pivotal transformation for generating the biologically active conformation. The patent specifies the use of Pd(OAc)2 with tri-o-tolylphosphine as the ligand system, combined with Proton Sponge as a non-nucleophilic base and LiBr as an additive. Conducting this reaction in DMA or NMP at elevated temperatures of 110°C drives the equilibrium towards the thermodynamically stable E-isomer. The mechanistic role of LiBr is particularly noteworthy, as it aids in the solubilization of the palladium species and accelerates the catalytic cycle. For scenarios requiring the Z-isomer, an alternative Sonogashira coupling followed by stereoselective reduction offers a viable pathway, demonstrating the versatility of this platform in accessing diverse chemical space for structure-activity relationship studies.
Implementing this synthesis protocol requires careful attention to reaction parameters to ensure reproducibility and safety on a large scale. The process generally involves three main stages: first, the construction of the indazole-thioether core via copper or palladium catalysis; second, the optional protection of the indazole nitrogen to prevent side reactions; and third, the final carbon-carbon bond formation via Heck or Sonogashira coupling. Each step is designed to be telescoped where possible, reducing solvent usage and processing time. Critical control points include maintaining strict anhydrous conditions during the coupling phases and monitoring reaction progress via HPLC to prevent over-reaction. The final deprotection and crystallization steps are equally vital, as they determine the physical form and purity of the isolated material, directly impacting its suitability for downstream formulation.
From a procurement and supply chain perspective, the adoption of this synthetic route offers substantial strategic advantages. The reliance on commercially available starting materials, such as substituted indazoles and vinyl pyridines, reduces the risk of raw material shortages and price volatility. Furthermore, the robustness of the catalytic systems allows for the use of standard reactor equipment found in most multipurpose pharmaceutical plants, facilitating rapid technology transfer and commercial scale-up of complex pharmaceutical intermediates. The ability to consistently produce material with greater than 95% purity minimizes the need for repetitive recrystallizations, thereby reducing solvent consumption and waste generation. This efficiency translates directly into a more reliable supply chain for high-purity pharmaceutical intermediates, ensuring that clinical and commercial timelines are met without compromise.
Cost reduction in API manufacturing is significantly driven by the efficient removal of residual catalysts, a common bottleneck in metal-catalyzed processes. The patent details a highly effective purification strategy using cysteine-silica or chelating agents like 1,2-diaminopropane to scavenge palladium. This capability allows manufacturers to reduce residual metal content to levels below 10 ppm, eliminating the need for expensive chromatographic purification steps that often limit batch size. Additionally, the control over polymorphic forms, specifically the generation of Form IV crystals through specific solvent systems involving THF, DMF, and MeOH, ensures consistent physical properties. This level of control over solid-state characteristics is essential for regulatory filings and guarantees batch-to-batch consistency, ultimately lowering the cost of goods sold through improved yield and reduced rejection rates.

Scalability and environmental compliance are further enhanced by the optimization of solvent systems and reaction conditions. The use of solvents like NMP and DMA, while requiring careful handling, allows for high concentration reactions that maximize reactor throughput. The described workup procedures, which often involve aqueous washes and filtration rather than complex extractions, simplify the isolation of the product and reduce the volume of organic waste generated. Moreover, the ability to recycle mother liquors or recover solvents contributes to a greener manufacturing footprint. By integrating these efficient unit operations, manufacturers can achieve substantial cost savings and adhere to increasingly stringent environmental regulations, making this process not only chemically elegant but also economically and ecologically sustainable for long-term production.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and purification of these indazole derivatives. Understanding these aspects is crucial for R&D teams evaluating the feasibility of this route for their specific pipeline candidates. The answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for process development scientists.
Q: How is palladium content controlled in the final indazole API intermediate?
A: The patent describes a rigorous purification process using 10% cysteine-silica scavengers or chelating agents like 1,2-diaminopropane and DIPHOS. This reduces residual palladium from levels around 1000 ppm down to less than 10 ppm, ensuring compliance with strict pharmaceutical safety standards.
Q: Can the stereochemistry of the double bond in the side chain be controlled?
A: Yes, the process allows for the preparation of both E and Z isomers. The E-isomer is typically formed directly via the Heck reaction. The Z-isomer can be accessed via a Sonogashira coupling to form an alkyne intermediate (Formula 10), followed by stereoselective reduction using Lindlar's catalyst or hydrazine.
Q: What are the advantages of using N-1 protection groups like THP or Boc?
A: Protecting the N-1 position of the indazole ring prevents unwanted alkylation or side reactions during the harsh conditions of the Heck coupling. It ensures higher regioselectivity and purity of the final product, simplifying downstream purification and improving overall yield.
- Couple a halogenated indazole (Formula 4) with a thiol-benzamide derivative (Formula 3) using a Pd or Cu catalyst (e.g., Pd2(dba)3/Xantphos) and a base like CsOH in DMF or NMP at 70-80°C to form intermediate Formula 2.
- Optionally protect the indazole N-1 position with a group like THP or Boc to prevent side reactions during subsequent coupling steps, forming Formula 7.
- Perform a Heck reaction between the iodinated intermediate (Formula 2 or 7) and a vinyl heteroaryl compound (e.g., 2-vinylpyridine) using Pd(OAc)2, P(o-Tol)3, Proton Sponge, and LiBr in DMA at 110°C to install the side chain.
- Remove any protecting groups using acidic conditions (e.g., TsOH/MeOH or TFA) and purify the final product via crystallization, employing Pd scavengers like cysteine-silica to ensure metal content below 10 ppm.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indazole Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the production of complex kinase inhibitor intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of indazole compounds meets the highest international standards for pharmaceutical applications. Our commitment to quality extends to our ability to manage complex chemistries, including sensitive palladium-catalyzed reactions and precise polymorph control.
We invite you to collaborate with us to optimize your supply chain for these vital therapeutic intermediates. By leveraging our technical expertise, you can achieve a Customized Cost-Saving Analysis tailored to your specific project requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your drug development timeline with our proven manufacturing capabilities and dedication to excellence in fine chemical synthesis.
