Advanced Catalytic Synthesis of Imidazo[1,2-b]pyridazine Derivatives for Commercial Herbicide Production
Advanced Catalytic Synthesis of Imidazo[1,2-b]pyridazine Derivatives for Commercial Herbicide Production
The development of efficient synthetic routes for heterocyclic intermediates is a critical bottleneck in the agrochemical industry, particularly for high-activity sulfonylurea herbicides. Patent CN100341875C discloses a groundbreaking methodology for the preparation of imidazo[1,2-b]pyridazine derivatives substituted at the 6-position carbon atom. This technology addresses long-standing challenges in regioselectivity and operational safety, offering a robust pathway for generating key scaffolds used in modern crop protection agents. By leveraging transition metal catalysis, specifically utilizing nickel, palladium, or iron complexes, the process enables the direct coupling of organometallic reagents with halo-substituted pyridazine precursors. This innovation not only streamlines the synthesis of complex heterocycles but also aligns with modern green chemistry principles by avoiding hazardous solvents and extreme reaction conditions typically associated with traditional nucleophilic substitutions on electron-deficient rings.

For procurement managers and supply chain directors, the implications of this technology extend beyond mere chemical novelty. The ability to introduce diverse alkyl, alkenyl, or cycloalkyl groups at the 6-position with high fidelity translates directly into supply chain resilience. Traditional methods often suffer from low yields or require multi-step protection-deprotection sequences that inflate lead times and raw material costs. In contrast, this catalytic approach utilizes readily available organomagnesium or organozinc reagents, which are commercially accessible or easily generated in situ. The process described in the patent operates under mild thermal conditions, typically ranging from -20°C to 50°C, which significantly reduces energy consumption compared to high-temperature reflux protocols. Furthermore, the compatibility of this method with various functional groups ensures that downstream processing remains straightforward, minimizing the formation of difficult-to-remove impurities that often plague bulk chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of the imidazo[1,2-b]pyridazine ring at the 6-position has been fraught with difficulties due to the electronic nature of the heterocycle. Conventional synthetic strategies often relied on harsh nucleophilic substitution conditions that required polar aprotic solvents such as hexamethylphosphoryl triamide (HMPT) or dimethylacetamide (DMA). These solvents are not only suspected to be toxic and carcinogenic but also pose significant challenges in waste disposal and worker safety compliance. Moreover, traditional routes frequently lacked regioselectivity, leading to mixtures of isomers where substitution occurred at undesired positions or where the existing 2-position substituents were compromised. The necessity for high temperatures to drive these reactions further exacerbated safety risks and increased the formation of thermal degradation byproducts, complicating purification and reducing overall process efficiency. Such limitations made the commercial scale-up of these intermediates economically unviable for many manufacturers seeking cost-effective herbicide production.
The Novel Approach
The novel approach detailed in the patent revolutionizes this landscape by employing a transition metal-catalyzed cross-coupling mechanism that operates under remarkably mild conditions. Instead of relying on brute-force thermal energy, the process utilizes the catalytic power of nickel or palladium complexes to facilitate the bond formation between the heterocyclic core and the incoming organic fragment. This allows the reaction to proceed efficiently in safer, non-polar solvents like tetrahydrofuran (THF), diethyl ether, or toluene. The method demonstrates exceptional tolerance for various leaving groups at the 6-position, including halogens and sulfonates, providing flexibility in starting material selection. Crucially, the reaction preserves the integrity of substituents at the 2-position, ensuring high structural fidelity of the final product. This shift from stoichiometric harsh chemistry to catalytic precision represents a paradigm shift in how these valuable agrochemical intermediates are manufactured, offering a cleaner, safer, and more economically attractive alternative for industrial applications.
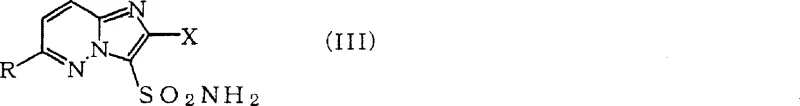
Mechanistic Insights into Nickel-Catalyzed Cross-Coupling
The core of this technological advancement lies in the mechanistic efficiency of the transition metal catalyst, particularly when nickel complexes such as dichloro[1,3-bis(diphenylphosphino)propane]nickel(II) are employed. The catalytic cycle likely initiates with the oxidative addition of the nickel(0) species into the carbon-halogen bond at the 6-position of the imidazo[1,2-b]pyridazine ring. This step is facilitated by the electron-deficient nature of the pyridazine nitrogen atoms, which activate the adjacent carbon centers towards metal insertion. Following oxidative addition, the organometallic reagent, typically a Grignard or organozinc compound, undergoes transmetallation with the nickel center, transferring the alkyl or alkenyl group to the metal complex. The final reductive elimination step releases the coupled product and regenerates the active nickel catalyst, allowing the cycle to continue with minimal catalyst loading. This mechanism explains the high turnover numbers observed in the experimental data, where catalyst loadings as low as 0.0001 equivalents can still drive the reaction to completion effectively.
From an impurity control perspective, the specificity of this catalytic cycle is paramount. The patent data highlights that when the 2-position substituent is a halogen, such as chlorine, it remains untouched during the coupling at the 6-position. This chemoselectivity is critical for R&D directors focused on purity profiles, as it eliminates the need for additional protection steps that would otherwise be required to mask the 2-position reactivity. The use of organozinc reagents, in particular, offers enhanced functional group tolerance compared to Grignard reagents, further reducing the risk of side reactions with sensitive moieties like esters or nitriles that might be present in more complex derivatives. The ability to conduct these reactions at temperatures as low as -20°C also suppresses thermal decomposition pathways, ensuring that the final crude product contains fewer degradation impurities. This results in a cleaner reaction profile that simplifies downstream purification, whether via crystallization or chromatography, ultimately yielding a high-purity intermediate suitable for stringent pharmaceutical or agrochemical standards.
![Specific example of Ni-catalyzed coupling reaction producing 2-chloro-6-n-propylimidazo[1,2-b]pyridazine](/insights/img/imidazo-pyridazine-derivative-synthesis-agrochemical-supplier-20260307013303-016.webp)
How to Synthesize 6-Substituted Imidazo[1,2-b]pyridazine Efficiently
The practical implementation of this synthesis route involves a straightforward protocol that balances reactivity with safety. The process begins with the suspension of the halogenated imidazo[1,2-b]pyridazine substrate and the chosen transition metal catalyst in an anhydrous solvent system, typically under an inert nitrogen atmosphere to prevent catalyst deactivation by oxygen or moisture. The organometallic coupling partner is then introduced slowly to manage the exotherm, maintaining strict temperature control to optimize selectivity. Following the reaction period, standard aqueous workup procedures are sufficient to quench excess reagents and separate the organic product. The detailed standardized synthesis steps below outline the precise conditions required to replicate the high yields reported in the patent examples, ensuring reproducibility for process development teams.
- Prepare the reaction mixture by suspending the 6-halo-imidazo[1,2-b]pyridazine substrate and a nickel or palladium catalyst in an anhydrous ether or hydrocarbon solvent under inert atmosphere.
- Add the organometallic reagent, such as an alkylmagnesium halide or alkylzinc halide, dropwise while maintaining the internal temperature between -20°C and 50°C to ensure selectivity.
- Quench the reaction with water or acid, separate the organic layer, and purify the resulting 6-substituted derivative via silica gel column chromatography or crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for sourcing and logistics, the adoption of this catalytic technology offers substantial strategic benefits that go beyond simple yield improvements. The elimination of toxic solvents like HMPT and DMA removes a significant regulatory burden, simplifying environmental compliance and reducing the costs associated with hazardous waste disposal. This shift towards greener solvents such as THF or toluene also enhances the safety profile of the manufacturing facility, potentially lowering insurance premiums and improving operational continuity. Furthermore, the use of earth-abundant metals like nickel, as opposed to more expensive palladium in some embodiments, provides a direct avenue for raw material cost optimization without sacrificing reaction efficiency. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or reagent quality, leading to more consistent batch-to-batch performance and reliable supply schedules for downstream customers.
- Cost Reduction in Manufacturing: The transition to a catalytic process significantly lowers the consumption of expensive reagents and solvents. By avoiding the need for stoichiometric amounts of harsh activating agents and eliminating toxic solvent disposal costs, the overall cost of goods sold is drastically reduced. The ability to use lower catalyst loadings while maintaining high conversion rates further contributes to economic efficiency, making the production of these complex intermediates financially viable even at large scales. Additionally, the simplified purification requirements reduce the consumption of silica gel and elution solvents, adding another layer of cost savings to the manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on commercially available organometallic reagents and common transition metal salts ensures a stable supply of raw materials. Unlike specialized reagents that may have long lead times or single-source dependencies, the inputs for this process are widely produced by multiple chemical suppliers globally. This diversification of the supply base mitigates the risk of shortages and price volatility, ensuring that production schedules can be maintained consistently. The mild reaction conditions also reduce the strain on manufacturing equipment, extending asset life and minimizing unplanned downtime due to maintenance or corrosion issues associated with harsher chemical environments.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with exotherms that are manageable through standard cooling systems rather than requiring specialized cryogenic equipment. This makes the technology transfer from laboratory to pilot plant and finally to commercial production much smoother and less capital intensive. From an environmental standpoint, the avoidance of persistent organic pollutants and toxic solvents aligns with increasingly stringent global environmental regulations. This proactive compliance strategy future-proofs the manufacturing operation against potential regulatory changes, ensuring long-term operational viability and enhancing the corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity for engineering and procurement teams evaluating this technology for adoption. Understanding these nuances is essential for accurate process design and risk assessment.
Q: What catalysts are most effective for this cross-coupling reaction?
A: The patent data indicates that nickel complexes, particularly dichloro[1,3-bis(diphenylphosphino)propane]nickel(II), provide superior yields compared to palladium or iron catalysts for this specific heterocyclic system.
Q: Can this process avoid toxic polar solvents like HMPT?
A: Yes, the novel process operates efficiently in ether or hydrocarbon solvents such as THF, diethyl ether, or toluene, eliminating the need for suspected toxic amide solvents like HMPT or DMA used in conventional methods.
Q: Is the 2-position substituent stable during the 6-position coupling?
A: The method demonstrates high chemoselectivity, allowing the 2-position halogen or alkyl group to remain intact while the leaving group at the 6-position is selectively substituted by the organometallic reagent.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazo[1,2-b]pyridazine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of imidazo[1,2-b]pyridazine derivatives meets the exacting standards required for herbicide synthesis. Our commitment to quality and safety mirrors the innovations described in patent CN100341875C, allowing us to deliver high-performance intermediates that empower your R&D and production goals.
We invite you to collaborate with us to leverage this advanced catalytic technology for your specific supply chain needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient process can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to secure a reliable, cost-effective, and sustainable supply of these vital chemical building blocks for the global agrochemical market.
