Advanced Synthesis of High-Selectivity N2-Alkyl Substituted Triazole Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing nitrogen-rich heterocycles, particularly the 1,2,3-triazole scaffold, which serves as a critical pharmacophore in numerous bioactive molecules. Addressing the longstanding challenge of regioselective alkylation, Chinese Patent CN116178286A discloses a groundbreaking synthetic strategy for producing high-selectivity N2-alkyl substituted triazole derivatives. This innovation leverages a DBU-mediated alkylation process that operates under remarkably mild conditions, overcoming the thermodynamic preferences that typically favor N1-substitution. The significance of this technology lies in its ability to deliver complex intermediates with superior purity and yield, directly impacting the efficiency of downstream drug discovery and development pipelines. By utilizing N-sulfonyl-1,2,3-triazoles as key starting materials, the method bypasses the need for bulky protecting groups that require subsequent removal steps, thereby streamlining the overall synthetic sequence.
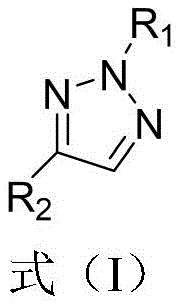
The structural versatility of these derivatives, as illustrated in the general formula, allows for extensive modification at both the N2 and C4 positions, enabling medicinal chemists to fine-tune physicochemical properties for optimal biological activity. This patent represents a significant leap forward for reliable pharmaceutical intermediate suppliers aiming to diversify their catalog with high-value heterocyclic building blocks. The ability to access these specific isomers efficiently opens new avenues for the synthesis of anticancer agents and other therapeutic classes where the triazole ring plays a pivotal role in receptor binding and metabolic stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the regioselective synthesis of N2-substituted 1,2,3-triazoles has been plagued by significant technical hurdles that hinder efficient manufacturing. Traditional approaches often rely on the inherent electronic density differences between the nitrogen atoms, where the terminal N1 and N3 positions are naturally more nucleophilic than the internal N2 position. To overcome this, prior art methods frequently necessitate the use of harsh reaction conditions, including elevated temperatures exceeding 65°C and prolonged reaction times lasting more than two hours. Furthermore, many existing strategies depend on the introduction of sterically bulky protecting groups to force substitution at the desired position, which subsequently requires additional synthetic steps for deprotection. These multi-step sequences not only increase the overall cost of goods but also accumulate impurities, complicating the purification process and reducing the final yield. The reliance on such inefficient protocols creates bottlenecks in the supply chain for high-purity OLED material and pharmaceutical precursors, limiting the speed at which new candidates can be advanced.
The Novel Approach
In stark contrast, the methodology described in the patent introduces a streamlined, one-pot transformation that achieves high regioselectivity without the need for extreme thermal energy or complex protecting group manipulations. By employing 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) as a specific additive in conjunction with an organic solvent like acetonitrile, the reaction proceeds rapidly at room temperature, typically completing within 10 to 30 minutes. This dramatic reduction in reaction time and energy consumption translates directly into substantial cost savings in electronic chemical manufacturing and related sectors. The process utilizes readily available N-sulfonyl-1,2,3-triazoles and simple alkyl halides, reacting them in a molar ratio of approximately 1:1.2 to 1:1.5 to ensure complete conversion. The result is a highly efficient pathway that delivers the desired N2-alkyl products with yields often exceeding 80 percent and selectivity ratios greater than 6:1. This novel approach effectively resolves the competitive pathway issues that have long frustrated synthetic chemists, providing a scalable solution for the commercial scale-up of complex polymer additives and drug intermediates.
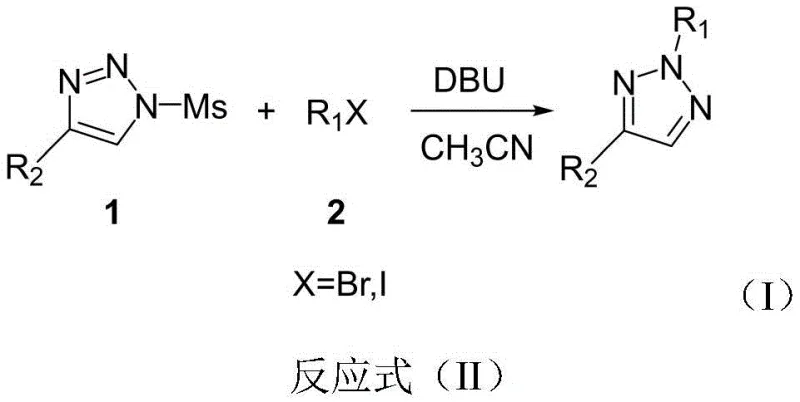
Mechanistic Insights into DBU-Mediated Regioselective Alkylation
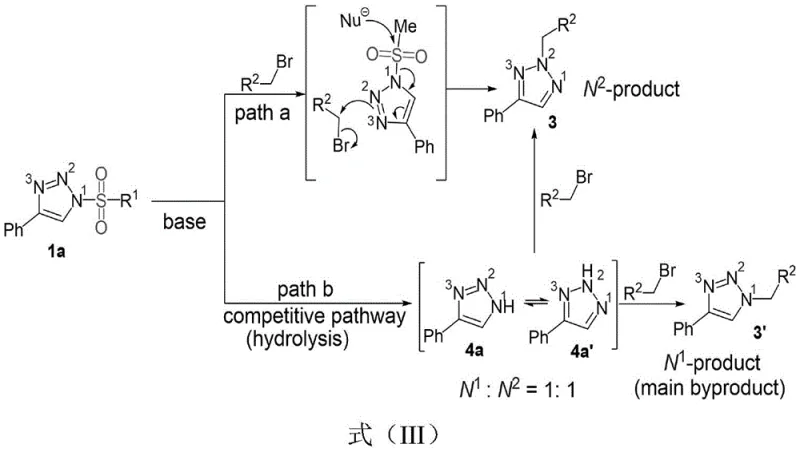
The success of this synthetic route hinges on a sophisticated understanding of the reaction mechanism, specifically how the DBU additive influences the nucleophilic attack on the triazole ring. The proposed mechanism suggests that the base facilitates the generation of a reactive intermediate from the N-sulfonyl triazole, effectively modulating the electron density distribution across the nitrogen atoms. Unlike traditional base-mediated alkylations where the most accessible nitrogen reacts, the presence of the sulfonyl group combined with DBU creates a unique electronic environment that favors attack at the N2 position. This is critical because the N2-isomer is often the thermodynamically less stable but biologically more relevant configuration for certain drug targets. The reaction avoids the formation of N1-substituted byproducts, which are typically the major impurities in non-selective processes. By suppressing these competitive pathways, the method ensures a cleaner crude reaction mixture, significantly reducing the burden on downstream purification units.
Furthermore, the mechanistic pathway elucidates why hydrolysis and other side reactions are minimized under these specific conditions. The rapid kinetics of the alkylation step outcompete potential degradation pathways, preserving the integrity of sensitive functional groups on the R1 and R2 substituents. This tolerance for diverse functional groups, including halogens, esters, and heterocycles, underscores the robustness of the chemistry. For R&D directors focused on impurity control, this mechanism offers a predictable profile where the primary concern is simply unreacted starting material rather than a complex array of regioisomers. The ability to consistently produce the N2-isomer with high fidelity allows for more accurate structure-activity relationship studies, accelerating the optimization of lead compounds. Understanding this catalytic cycle is essential for scaling the process, as it confirms that strict control of stoichiometry and base concentration is sufficient to maintain selectivity without needing exotic catalysts.
How to Synthesize N2-Alkyl Substituted Triazole Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and selectivity. The process begins by dissolving the N-sulfonyl triazole substrate in a suitable organic solvent, with acetonitrile being the preferred choice due to its polarity and ability to solubilize both the ionic intermediates and organic reactants. The concentration of the triazole compound is typically maintained between 0.05M and 0.15M to ensure optimal reaction kinetics without causing precipitation or viscosity issues. Once the substrate is in solution, the alkyl halide electrophile is introduced, followed by the addition of DBU. It is crucial to add the reagents in the correct order and at room temperature to prevent exothermic spikes that could degrade selectivity. The detailed standardized synthesis steps see the guide below.
- Dissolve the N-sulfonyl-1,2,3-triazole substrate in an organic solvent such as acetonitrile to achieve a concentration of approximately 0.10M.
- Add the alkyl halide (bromide or iodide) electrophile to the reaction mixture at room temperature, maintaining a molar ratio of roughly 1: 1.2 relative to the triazole.
- Introduce DBU base as an additive and stir the reaction at 25°C for 10 to 30 minutes, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DBU-mediated synthesis offers tangible benefits that extend beyond mere chemical elegance. The shift from high-temperature, long-duration reactions to a room temperature process completed in minutes drastically reduces energy consumption and reactor occupancy time. This efficiency gain allows manufacturing facilities to increase throughput without expanding physical infrastructure, effectively lowering the unit cost of production. Moreover, the high selectivity of the reaction minimizes the generation of difficult-to-separate isomers, which simplifies the purification workflow. In traditional processes, separating N1 and N2 isomers often requires expensive preparative HPLC or multiple recrystallizations, both of which drive up costs and extend lead times. By delivering a cleaner product directly from the reactor, this method reduces the demand for chromatographic media and solvents, contributing to a more sustainable and cost-effective operation.
- Cost Reduction in Manufacturing: The elimination of harsh reaction conditions and the reduction in reaction time from hours to minutes significantly lowers utility costs associated with heating and stirring. Additionally, the high yield and selectivity reduce the amount of raw material wasted on byproducts, optimizing the atom economy of the process. Since the method avoids the use of expensive transition metal catalysts or specialized ligands, the raw material costs are kept low, relying instead on commodity chemicals like DBU and acetonitrile. This economic efficiency makes the production of high-purity pharmaceutical intermediates more viable, allowing for competitive pricing in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on widely available and stable reagents ensures that the supply chain is resilient to disruptions. Unlike processes dependent on scarce metals or custom-synthesized catalysts, the inputs for this reaction are commodity chemicals with robust global supply networks. The simplicity of the workup procedure, which typically involves standard column chromatography or crystallization, means that the process can be easily transferred between different manufacturing sites or contract organizations. This flexibility enhances supply continuity, ensuring that critical intermediates for drug production are available when needed. Reducing lead time for high-purity intermediates becomes achievable as the bottleneck of lengthy reaction and purification cycles is removed.
- Scalability and Environmental Compliance: The mild nature of the reaction conditions facilitates safe scale-up from gram to kilogram and ton scales. Operating at ambient temperature reduces the risk of thermal runaway incidents, enhancing workplace safety and lowering insurance premiums. Furthermore, the reduced solvent usage and shorter reaction times contribute to a smaller environmental footprint, aligning with increasingly stringent regulatory requirements for green chemistry. The ability to recycle solvents like acetonitrile further improves the sustainability profile of the process. This compliance with environmental standards mitigates regulatory risks and supports the long-term viability of the manufacturing operation, making it an attractive option for companies committed to corporate social responsibility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These answers are derived directly from the experimental data and mechanistic insights provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production workflows.
Q: What is the primary advantage of the DBU-mediated method for N2-alkyl triazoles?
A: The primary advantage is the exceptional regioselectivity for the N2-position over the N1/N3 positions, achieved under mild room temperature conditions with short reaction times, avoiding the harsh temperatures required by conventional methods.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the method utilizes common solvents like acetonitrile and operates at ambient temperature without requiring specialized high-pressure or cryogenic equipment, making it highly suitable for commercial scale-up.
Q: What types of substrates are compatible with this alkylation protocol?
A: The protocol demonstrates broad substrate scope, accommodating various alkyl halides including benzyl, alkyl, and functionalized chains, as well as diverse triazole cores with electron-withdrawing or donating groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N2-Alkyl Substituted Triazole Derivatives Supplier
As the demand for specialized heterocyclic intermediates continues to grow, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM provides a strategic advantage. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from clinical trials to full-scale manufacturing. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of N2-alkyl triazole derivatives meets the highest industry standards. We understand the critical nature of supply chain consistency and are committed to delivering high-quality intermediates that support your drug development timelines.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how this innovative synthesis route can benefit your portfolio. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this DBU-mediated method. We are ready to provide specific COA data and route feasibility assessments to help you make informed decisions. Let us collaborate to accelerate your research and bring life-saving therapies to market faster and more efficiently.
