Advanced Synthesis of Indole-3-formaldehyde Phenylenediamine Bis-Schiff Bases for Commercial Scale-up
Advanced Synthesis of Indole-3-formaldehyde Phenylenediamine Bis-Schiff Bases for Commercial Scale-up
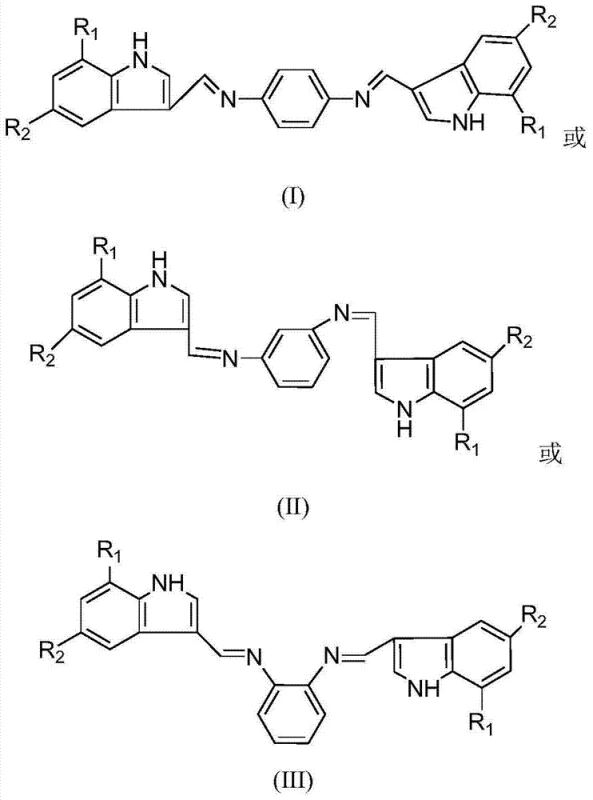
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for heterocyclic intermediates that balance high purity with operational simplicity. Patent CN102584670B introduces a significant advancement in this domain by detailing the preparation of indole-3-formaldehyde phenylenediamine bis-Schiff bases. This technology leverages the condensation reaction between various indole-3-carbaldehyde derivatives and inexpensive diamine precursors to generate a series of conjugated structures. The innovation lies not just in the novel chemical entities produced, which possess dual functionality as antibacterial agents and fluorescent materials, but in the remarkably mild and scalable reaction conditions employed. By utilizing a straightforward thermal process ranging from 25°C to 100°C, the method eliminates the need for complex catalytic systems or extreme pressures, thereby presenting a highly attractive pathway for reliable pharmaceutical intermediate supplier operations aiming for cost-effective manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of Schiff bases, particularly those involving sensitive heterocyclic aldehydes like indole-3-carbaldehyde, often suffers from significant process inefficiencies that hinder commercial viability. Conventional protocols frequently require stringent anhydrous conditions, expensive Lewis acid catalysts, or prolonged reaction times that lead to the degradation of the indole ring system. Furthermore, the purification of mono-Schiff bases versus bis-Schiff bases can be notoriously difficult, often requiring column chromatography which is impractical for multi-kilogram production. The presence of unreacted amine or aldehyde impurities can severely impact the biological efficacy of the final API intermediate, necessitating multiple recrystallization steps that drive down overall yield. Additionally, many older methods rely on volatile organic solvents that pose safety hazards and environmental compliance issues, increasing the operational expenditure for waste treatment and solvent recovery in large-scale facilities.
The Novel Approach
The methodology described in CN102584670B overcomes these historical bottlenecks through a streamlined condensation strategy that prioritizes atom economy and operational safety. By reacting indole-3-formaldehyde analogues directly with phenylenediamines in common solvents like methanol or dioxane, the process achieves high conversion rates without the need for exotic reagents. The reaction tolerates a wide temperature window of 25-100°C, allowing process engineers to optimize kinetics based on available equipment rather than rigid chemical constraints. Crucially, the formation of the bis-Schiff base is favored by controlling the molar ratio of aldehyde to diamine between 2:1 and 5:1, effectively minimizing the formation of mono-substituted byproducts. The resulting yellow powder solids are easily isolated via simple vacuum distillation and purified through standard recrystallization techniques, ensuring a high-purity product suitable for downstream pharmaceutical applications without the need for complex chromatographic separation.
Mechanistic Insights into Condensation Reaction Dynamics
The core chemical transformation driving this synthesis is the nucleophilic addition-elimination reaction between the primary amine groups of the phenylenediamine and the carbonyl carbon of the indole-3-carbaldehyde. Under the specified thermal conditions, the lone pair electrons on the nitrogen atom attack the electrophilic carbonyl carbon, forming a unstable carbinolamine intermediate. Subsequent dehydration leads to the formation of the characteristic imine (C=N) double bond, creating the conjugated Schiff base linkage. The presence of electron-withdrawing or electron-donating substituents on the indole ring (such as nitro, methoxy, chloro, or fluoro groups) modulates the electrophilicity of the aldehyde, which is why the patent specifies a flexible reaction time of 2 to 12 hours to accommodate different derivative reactivities. This mechanistic flexibility ensures that even less reactive substrates can be fully converted given sufficient time, preventing the accumulation of starting materials that could complicate purification.
From an impurity control perspective, the rigid planar structure of the resulting bis-Schiff base facilitates effective crystallization, which acts as a powerful purification mechanism in itself. The conjugation across the indole-imine-phenyl-imine-indole backbone creates a stable electronic system that resists hydrolysis under neutral workup conditions, unlike simpler aliphatic Schiff bases. The patent data indicates that the proton NMR signals for the imine protons appear distinctly downfield (around 8.00-8.80 ppm), providing a clear analytical marker for quality control teams to verify the completeness of the reaction. By maintaining the solvent-to-reactant ratio between 1:10 and 1:100, the system ensures adequate solubility to prevent premature precipitation of oligomers while keeping the concentration high enough to drive the equilibrium toward the desired bis-condensed product, thus maximizing yield and minimizing waste.
How to Synthesize Indole-3-formaldehyde Phenylenediamine Bis-Schiff Base Efficiently
To implement this synthesis effectively in a pilot or production plant, operators must adhere to the specific stoichiometric and thermal parameters outlined in the patent examples. The process begins with the dissolution of the indole-3-formaldehyde derivative in a selected solvent, followed by the controlled addition of the diamine component. Maintaining the correct molar excess of the aldehyde is critical to driving the reaction to the bis-substituted state. The reaction mixture is then heated isothermally, with temperature selection depending on the boiling point of the solvent and the reactivity of the specific indole derivative used.
- Dissolve indole-3-formaldehyde derivatives in a suitable solvent such as methanol or dioxane at a molar ratio of 1: 10 to 1:100.
- Add diamine compounds (o-, m-, or p-phenylenediamine) to the solution maintaining a molar ratio of diamine to aldehyde between 1: 2 and 1:5.
- Stir the mixture isothermally at 25-100°C for 2-12 hours, remove solvent under reduced pressure, and purify the resulting yellow solid via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits regarding cost stability and sourcing reliability. The reliance on commodity chemicals such as phenylenediamine and substituted indole aldehydes means that raw material costs are insulated from the volatility often associated with specialized catalysts or rare earth metals. The simplicity of the workup procedure, which avoids chromatography and utilizes standard distillation and recrystallization, significantly reduces the consumption of silica gel and elution solvents, directly lowering the variable cost per kilogram of the active intermediate. Furthermore, the broad solvent compatibility allows manufacturers to switch between methanol, acetone, or dioxane based on real-time market pricing and availability, providing a hedge against supply chain disruptions for specific reagents.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of ambient to moderate pressure conditions drastically reduce capital expenditure requirements for reactor infrastructure. Since the reaction proceeds efficiently in standard glass-lined or stainless steel vessels without the need for high-pressure autoclaves, the barrier to entry for manufacturing is lowered. The high yield and ease of purification mean that fewer batches are required to meet production targets, effectively spreading fixed overhead costs over a larger volume of saleable product. Additionally, the ability to recover and reuse solvents like methanol and ethyl acetate further enhances the economic profile of the process, contributing to substantial cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is paramount for maintaining long-term contracts with downstream API manufacturers. Because the process tolerates a wide range of temperatures and reaction times, minor fluctuations in utility supply (such as steam pressure variations) are less likely to result in batch failures or off-spec material. The use of widely available starting materials reduces the risk of single-source supplier dependency, allowing procurement teams to diversify their vendor base for key inputs. This resilience translates to shorter lead times for high-purity intermediates, as production scheduling becomes more predictable and less prone to delays caused by reagent shortages or complex logistical hurdles.
- Scalability and Environmental Compliance: Scaling this process from laboratory grams to commercial tons is straightforward due to the absence of exothermic runaway risks typically associated with aggressive reagents. The mild thermal profile allows for efficient heat transfer management in large reactors, ensuring safe scale-up without the need for specialized cooling systems. From an environmental standpoint, the process generates minimal hazardous waste, as the primary byproduct is water formed during condensation. The solvents used are generally classified as Class 2 or 3 residual solvents, which are easier to manage and dispose of in compliance with ICH Q3C guidelines. This alignment with green chemistry principles simplifies regulatory filings and reduces the environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and application of this technology, derived directly from the experimental data and claims within the patent documentation. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this route into their existing portfolio.
Q: What are the primary applications of these indole-based bis-Schiff bases?
A: According to patent CN102584670B, these compounds exhibit significant antibacterial and bacteriostatic activities, making them valuable as pharmaceutical intermediates. Additionally, their conjugated rigid planar structure provides strong fluorescence properties suitable for material science applications.
Q: What solvents are compatible with this synthesis route?
A: The process demonstrates flexibility in solvent choice, successfully utilizing methanol, acetone, 1,4-dioxane, and tetrahydrofuran (THF). This versatility allows manufacturers to optimize for cost and recovery efficiency without compromising yield.
Q: How is the purity of the final product ensured?
A: The protocol specifies a straightforward purification strategy involving vacuum distillation to remove solvents followed by recrystallization using ethanol, ethyl acetate, or acetone. This ensures the removal of unreacted starting materials and oligomeric impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole-3-formaldehyde Phenylenediamine Bis-Schiff Base Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation therapeutics and functional materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the structural and spectral standards defined in patents like CN102584670B. Our facility is equipped to handle the specific solvent systems and thermal requirements of this condensation chemistry, guaranteeing a consistent supply of material for your clinical or commercial needs.
We invite potential partners to engage with our technical procurement team to discuss how this specific synthetic route can be optimized for your unique project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies can translate into better margins for your final product. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about securing a stable supply of these valuable fluorescent and antibacterial intermediates.
