Scalable Vilsmeier-Mediated Dehydration of Aldoximes for High-Purity Nitrile Production
The chemical industry is constantly seeking more efficient pathways to produce essential nitrile intermediates, which serve as critical building blocks for pharmaceuticals and agrochemicals. Patent CN100500650C introduces a robust and scalable chemical synthesis method that addresses many of the limitations found in traditional dehydration protocols. This innovative approach utilizes bis(trichloromethyl) carbonate (BTC) reacting with N,N-disubstituted amides to generate a Vilsmeier reagent in situ, which subsequently dehydrates aldoximes into the corresponding nitrile substances. The process operates under remarkably mild conditions, typically ranging from -5°C to 5°C for reagent formation and 0°C to 120°C for the conversion step, ensuring high operational safety. By leveraging this specific catalytic system, manufacturers can achieve reaction yields generally greater than 85%, representing a significant improvement in process efficiency. The versatility of this method allows for the processing of diverse substrates, including substituted phenyl groups and various heterocycles, making it a valuable asset for complex molecule synthesis.
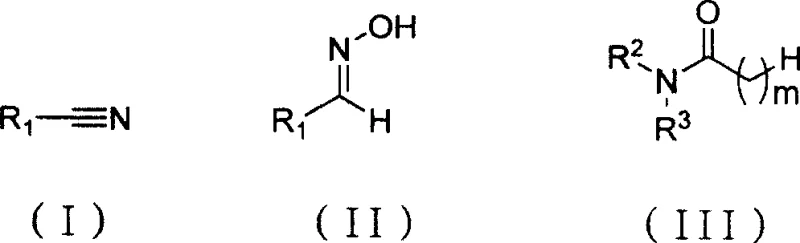
Traditional methods for preparing nitrile substances have long relied on the dehydration of primary amides or the reaction of carboxylic acid derivatives with ammonia, often requiring harsh thermal conditions and generating substantial waste. Another common historical approach involves the dehydration of aldoximes using strong mineral acids, which poses severe challenges regarding equipment corrosion and environmental compliance due to the generation of large volumes of acidic wastewater. These conventional routes frequently suffer from moderate yields and difficult purification steps, as the aggressive reaction environments can lead to the degradation of sensitive functional groups on the substrate. Furthermore, the handling of gaseous reagents or highly corrosive liquids in legacy processes increases the operational risk profile for manufacturing facilities. In contrast, the novel approach detailed in the patent utilizes a solid source of phosgene equivalent, BTC, which is safer and easier to handle than gaseous alternatives. This shift to a solid-state reagent combined with mild organic solvents drastically simplifies the engineering requirements for the reactor setup.
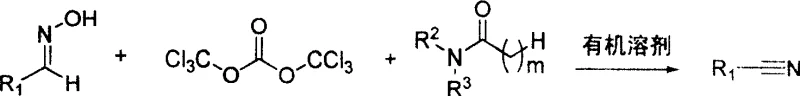
The mechanistic pathway of this synthesis relies on the in situ generation of a highly reactive Vilsmeier-Haack type reagent, which acts as a potent dehydrating agent for the aldoxime functionality. Initially, the bis(trichloromethyl) carbonate reacts with the N,N-disubstituted amide, such as N,N-dimethylformamide (DMF), to form an iminium salt intermediate. This electrophilic species then activates the hydroxyl group of the aldoxime, facilitating the elimination of water and the formation of the carbon-nitrogen triple bond characteristic of nitriles. The reaction kinetics are favorable, with the conversion typically completing within a short window of 2 to 60 minutes, depending on the specific substrate and temperature employed. Crucially, the mild temperature range of 10°C to 40°C during the main reaction phase prevents the thermal decomposition of the product or the formation of polymeric byproducts that often plague high-temperature dehydrations. This controlled environment ensures that the impurity profile remains manageable, reducing the burden on downstream purification units like crystallization or distillation columns.
Impurity control is a paramount concern for R&D directors overseeing the production of high-value intermediates, and this process offers distinct advantages in selectivity. The use of specific molar ratios, such as an aldoxime to BTC to amide ratio of 3:1:3, optimizes the consumption of reagents while minimizing the presence of unreacted starting materials or chlorinated side products. The patent data highlights that even with electron-rich or electron-deficient aromatic rings, the reaction maintains high specificity for the nitrile formation without affecting other sensitive moieties like esters or halogens. For instance, substrates containing chloro, nitro, or methoxy groups on the phenyl ring are converted efficiently with purities often exceeding 99% after simple recrystallization. This high level of chemoselectivity is attributed to the specific reactivity of the Vilsmeier complex, which targets the oxime hydroxyl group preferentially over other nucleophilic sites. Consequently, the final product requires less extensive chromatographic purification, aligning well with the cost and time constraints of industrial manufacturing.
How to Synthesize Nitrile Substances Efficiently
To implement this synthesis route effectively, operators must adhere to precise stoichiometric controls and temperature management protocols as outlined in the patent specifications. The process begins with the dissolution of bis(trichloromethyl) carbonate in a suitable organic solvent, followed by the controlled addition of the amide component at low temperatures to ensure stable reagent formation. Once the Vilsmeier reagent is generated, the aldoxime is introduced, and the reaction is allowed to proceed under mild heating or at ambient temperature until completion is confirmed. Detailed standardized synthetic steps, including specific workup procedures involving neutralization and filtration, are provided in the technical guide below to ensure reproducibility and safety.
- Dissolve bis(trichloromethyl) carbonate in an organic solvent such as methylene dichloride and cool the mixture to between -5°C and 5°C.
- Slowly add N,N-disubstituted amide (e.g., DMF) dropwise while stirring to generate the Vilsmeier reagent in situ, maintaining the low temperature for approximately 1 hour.
- Add the aldoxime substrate to the reaction mixture, allow the temperature to rise to 10-40°C, and stir for 5 to 30 minutes before neutralizing and isolating the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this BTC-mediated synthesis route offers compelling economic and logistical benefits over traditional acid-catalyzed dehydration methods. The primary advantage lies in the substantial reduction of waste treatment costs, as the process avoids the generation of large volumes of corrosive acidic effluent that requires expensive neutralization and disposal. Additionally, the use of solid BTC instead of gaseous reagents simplifies logistics and storage, reducing the need for specialized high-pressure containment systems and enhancing overall site safety. The high reaction yields, consistently reported above 85% and often nearing 96% in optimized examples, directly translate to better raw material utilization and lower cost of goods sold per kilogram of finished product. Furthermore, the short reaction times of merely minutes allow for higher throughput in existing reactor vessels, effectively increasing plant capacity without the need for capital-intensive expansion projects.
- Cost Reduction in Manufacturing: The elimination of harsh mineral acids and the associated corrosion-resistant equipment requirements leads to significant capital expenditure savings and lower maintenance costs over the lifecycle of the production facility. By utilizing widely available and relatively inexpensive reagents like BTC and DMF, the direct material costs are kept competitive while achieving superior yields that minimize waste. The simplified workup procedure, which often involves basic neutralization and filtration rather than complex extraction sequences, further reduces labor and utility consumption during the isolation phase. This holistic reduction in operational complexity ensures that the manufacturing process remains economically viable even at varying scales of production.
- Enhanced Supply Chain Reliability: The reliance on stable, solid reagents like bis(trichloromethyl) carbonate mitigates the supply risks associated with transporting and storing hazardous gases or unstable liquids. Since the reaction conditions are mild and the process is robust across a wide range of substrates, batch-to-batch variability is minimized, ensuring consistent delivery schedules to downstream customers. The ability to source common organic solvents such as methylene dichloride or acetonitrile further strengthens the supply chain resilience, as these materials are readily available in the global chemical market. This stability allows supply chain heads to forecast production timelines with greater accuracy and reduce the safety stock levels required for critical intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exothermic nature being easily managed through controlled dosing and standard cooling systems, making it suitable for transfer from pilot plants to multi-ton commercial reactors. From an environmental perspective, the reduction in acidic waste and the potential for solvent recovery align with increasingly stringent global regulations on industrial emissions and effluent discharge. The high atom economy of the dehydration reaction ensures that the majority of the input mass is converted into the desired product, supporting sustainability goals and reducing the carbon footprint of the manufacturing operation. These factors collectively make the technology an attractive option for companies aiming to modernize their production portfolios with greener chemistry solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitrile synthesis technology, derived directly from the patent's experimental data and claims. Understanding these aspects is crucial for evaluating the feasibility of integrating this method into existing production lines or new product development pipelines. The answers provided reflect the specific advantages and operational parameters documented in the intellectual property, offering a clear view of the technology's capabilities.
Q: What are the safety advantages of using BTC over traditional dehydrating agents?
A: Unlike traditional methods that often require corrosive strong acids or high-temperature dehydration which pose significant safety risks and equipment corrosion issues, this process utilizes bis(trichloromethyl) carbonate (BTC) under mild conditions (-5°C to 40°C). This significantly reduces the risk of thermal runaway and minimizes the generation of hazardous acidic waste streams, making it safer for large-scale industrial operations.
Q: What is the typical purity profile of nitriles produced via this method?
A: The patent data indicates that nitriles synthesized through this Vilsmeier-mediated pathway consistently achieve high purity levels, often exceeding 98% and reaching up to 99.6% after simple recrystallization. The mild reaction conditions help suppress side reactions such as polymerization or over-chlorination, resulting in a cleaner crude product that requires less intensive purification downstream.
Q: Can this synthesis method be applied to heterocyclic substrates?
A: Yes, the method demonstrates excellent versatility across a broad range of substrates. The patent explicitly confirms successful conversion of various heterocyclic aldoximes, including furyl, thienyl, pyrryl, and pyridyl derivatives, with yields generally remaining above 85%. This makes the process highly suitable for producing complex intermediates required in modern pharmaceutical and agrochemical development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrile Intermediate Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the Vilsmeier-mediated dehydration are executed with precision and reliability. Our technical team is adept at optimizing reaction parameters to meet stringent purity specifications, leveraging our rigorous QC labs to guarantee that every batch of nitrile intermediate meets the highest international standards. We understand the critical nature of supply continuity for our partners and have established robust procurement channels for key reagents like BTC and specialized amides to prevent any disruption in your manufacturing schedule. Our commitment to quality and safety makes us an ideal partner for bringing advanced chemical technologies from the laboratory to the global marketplace.
We invite you to contact our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this more efficient production route for your target molecules. We are ready to provide specific COA data from our pilot runs and comprehensive route feasibility assessments to support your decision-making process. Let us collaborate to enhance your supply chain efficiency and drive down costs while maintaining the highest standards of product quality.
