Breakthrough in High-Purity Imidazole Acetate Ionic Liquid Manufacturing and Commercial Scalability
Introduction to Advanced Ionic Liquid Synthesis
The chemical industry is currently witnessing a paradigm shift towards functional materials that combine high performance with environmental sustainability, a trend vividly exemplified by the innovations detailed in patent CN110294712B. This pivotal intellectual property discloses a sophisticated preparation method for high-purity imidazole acetate ionic liquids, addressing long-standing challenges regarding impurity control and process scalability. Unlike conventional approaches that often struggle with residual halides and heavy metal contamination, this novel methodology employs a strategic two-step anion exchange mechanism. By first converting the starting imidazolium halide into a perchlorate intermediate, the process creates a unique opportunity for rigorous purification before the final acetate substitution occurs. This technical breakthrough is not merely an academic exercise but represents a viable pathway for the reliable ionic liquid supplier seeking to deliver materials with purity levels exceeding 99.0% and halide residues capped at 5ppm. For R&D directors and procurement specialists alike, understanding the nuances of this synthesis is critical for securing supply chains for next-generation green solvents and catalytic media.
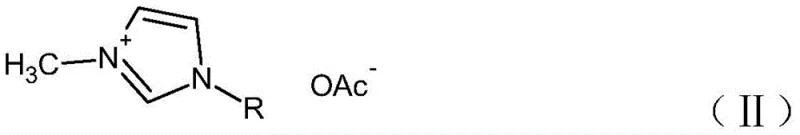
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of acetate-based ionic liquids has been plagued by significant technical and economic hurdles that hinder their widespread industrial adoption. Traditional methods often rely on expensive and light-sensitive reagents such as silver acetate, which not only drives up the cost of goods sold but also introduces stability issues during storage and handling. Alternative routes utilizing ion-exchange resins suffer from prolonged reaction times and the inherent instability of intermediate hydroxide species, making process control difficult and reproducibility poor. Furthermore, older metathesis techniques involving lead acetate introduce severe environmental liabilities due to toxic heavy metal residues that are notoriously difficult to remove completely from the final product matrix. Even direct metathesis using potassium acetate and imidazolium halides frequently results in products contaminated with thousands of parts per million of potassium and halide ions, rendering them unsuitable for sensitive applications in electronics or pharmaceuticals where trace impurities can catastrophically affect performance.
The Novel Approach
In stark contrast to these legacy methodologies, the approach outlined in CN110294712B introduces a robust, two-stage purification strategy that fundamentally alters the impurity profile of the final product. The core innovation lies in the isolation of a 1-alkyl-3-methylimidazolium perchlorate intermediate, which serves as a chemically distinct pivot point for purification. This intermediate allows for the effective removal of halide ions via aqueous washing of the organic phase, a step that is impossible when reacting halides directly with acetates. Subsequently, the reaction with potassium or ammonium acetate is conducted under conditions that favor the precipitation of inorganic by-products, which are then removed through freezing and filtration. This meticulous attention to phase behavior and solubility differences ensures that the final ionic liquid is virtually free from the metallic and halogenic contaminants that typically compromise quality. Consequently, this method offers a pathway for cost reduction in fine chemical intermediates manufacturing by utilizing cheap, stable raw materials while achieving superior purity specifications.
Mechanistic Insights into Two-Step Anion Exchange Metathesis
The chemical elegance of this synthesis lies in its exploitation of differential solubility and complexation thermodynamics to drive the reaction to completion while sequestering impurities. The first stage involves the reaction of the imidazolium halide with lithium perchlorate in a biphasic system of water and dichloromethane. In this environment, the lipophilic imidazolium cation preferentially partitions into the organic dichloromethane layer paired with the perchlorate anion, while the lithium halide by-product remains dissolved in the aqueous phase. This phase separation is the critical mechanistic step that allows for the physical removal of halide ions, reducing their concentration to negligible levels before the second reaction even begins. The resulting organic solution of the perchlorate intermediate is then washed repeatedly until silver nitrate testing confirms the absence of halide precipitation, ensuring a chemically clean substrate for the subsequent transformation.
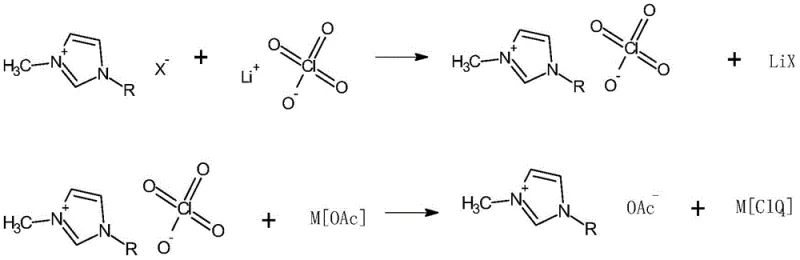
Following the isolation of the perchlorate intermediate, the second mechanistic phase involves a metathesis reaction with solid potassium acetate or ammonium acetate in an ethanol solvent. As the acetate anion exchanges with the perchlorate, the resulting potassium perchlorate or ammonium perchlorate exhibits low solubility in the reaction medium, driving the equilibrium forward according to Le Chatelier's principle. However, trace amounts of alkali metal ions often remain entrained in the viscous ionic liquid. To address this, the process employs 18-crown-6 ether in a dichloromethane wash during the final purification stage. The crown ether acts as a highly selective host molecule, forming stable complexes with residual potassium or ammonium ions. These complexes are either insoluble or easily separated during the final solvent evaporation, effectively scavenging the last traces of metal contamination. This dual-mechanism of precipitation followed by molecular recognition ensures the production of high-purity imidazole acetate with metal ion content below 200ppm, a specification that is exceptionally difficult to achieve with standard recrystallization techniques.
How to Synthesize 1-Alkyl-3-methylimidazolium Acetate Efficiently
The operational protocol for synthesizing these high-value ionic liquids requires precise control over stoichiometry, temperature, and phase separation to maximize yield and purity. The process begins with the dissolution of the imidazolium halide precursor and lithium perchlorate in water, followed by extraction into dichloromethane to isolate the perchlorate intermediate. Once the intermediate is secured and verified to be halide-free, it is redissolved in ethanol for the anion exchange with acetate salts. The reaction mixture is subjected to vigorous stirring and controlled cooling to induce the crystallization of inorganic salts, which are filtered off. The final polishing step involves treatment with a crown ether solution to chelate residual metals, followed by solvent removal under vacuum. For a comprehensive understanding of the specific molar ratios, temperature profiles, and safety precautions required for this synthesis, the detailed standardized synthesis steps are provided in the guide below.
- React 1-alkyl-3-methylimidazole halide with lithium perchlorate in a biphasic water/dichloromethane system to form the perchlorate intermediate.
- Perform metathesis with potassium acetate in ethanol, followed by freezing to precipitate impurities.
- Purify the crude product using a dichloromethane and 18-crown-6 mixture to scavenge residual alkali metal ions.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers profound advantages that extend beyond mere technical superiority, directly impacting the bottom line and supply chain resilience. By eliminating the need for precious metal catalysts like silver or toxic reagents like lead acetate, the process drastically simplifies the raw material procurement landscape. The reliance on commodity chemicals such as lithium perchlorate, potassium acetate, and common organic solvents ensures that the supply chain is robust and less susceptible to the volatility associated with specialty reagents. Furthermore, the absence of heavy metals removes the necessity for expensive and time-consuming heavy metal clearance steps, which are often regulatory bottlenecks in pharmaceutical and electronic grade manufacturing. This streamlining of the production workflow translates into substantial cost savings and a more predictable manufacturing timeline, enabling suppliers to offer competitive pricing without compromising on the stringent quality standards required by global clients.
- Cost Reduction in Manufacturing: The economic viability of this process is anchored in the substitution of high-cost reagents with inexpensive, widely available alternatives. Traditional methods utilizing silver acetate incur significant material costs due to the price of silver and the losses associated with its recovery or disposal. In contrast, the lithium perchlorate and potassium acetate route utilizes bulk chemicals that are stable and easy to handle, reducing both direct material costs and waste management expenses. Additionally, the high reaction yield exceeding 90% minimizes raw material waste, further enhancing the overall process efficiency. The elimination of complex resin regeneration or specialized equipment for handling light-sensitive materials also reduces capital expenditure and operational overhead, making the commercial scale-up of complex ionic liquids far more economically attractive.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on niche reagents that have limited global production capacity. By shifting to a synthesis route based on fundamental inorganic salts and common solvents, manufacturers can mitigate the risk of supply disruptions. Lithium perchlorate and acetates are produced at scale for various industries, ensuring a steady flow of inputs. Moreover, the stability of these raw materials means they can be stored for extended periods without degradation, allowing for strategic stockpiling against market fluctuations. This reliability is crucial for maintaining consistent production schedules and meeting the just-in-time delivery expectations of downstream customers in the pharmaceutical and agrochemical sectors who depend on uninterrupted access to high-quality intermediates.
- Scalability and Environmental Compliance: The environmental footprint of chemical manufacturing is increasingly becoming a decisive factor in vendor selection. This synthesis route is inherently greener, avoiding the generation of hazardous heavy metal waste streams that require costly treatment and disposal. The by-products, primarily lithium halides and potassium perchlorate, are manageable and do not pose the same ecological risks as lead or silver waste. The process is designed for scalability, utilizing standard unit operations such as liquid-liquid extraction, filtration, and distillation that are easily transferred from pilot plants to multi-ton production facilities. This ease of scale-up ensures that the technology can meet growing market demand for ionic liquids in applications ranging from biomass processing to electrochemistry, all while adhering to strict environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their operations, we have compiled answers to common inquiries regarding the synthesis and application of these ionic liquids. The following responses are derived directly from the technical data and experimental results presented in the patent documentation, providing a factual basis for assessing the method's capabilities. These insights address critical concerns regarding impurity profiles, reaction mechanisms, and the practical implications of the purification steps involved. Understanding these details is essential for R&D teams planning to utilize these materials in sensitive applications where trace contaminants could compromise product performance or regulatory compliance.
Q: How does this method reduce halide residues compared to traditional metathesis?
A: By converting the initial halide salt into a perchlorate intermediate first, the process utilizes dichloromethane extraction to effectively separate and wash away lithium halides before the final acetate exchange, reducing halide residues to ≤5ppm.
Q: Why is 18-crown-6 used in the final purification step?
A: 18-crown-6 acts as a phase-transfer catalyst and complexing agent that selectively binds residual potassium or ammonium ions, allowing them to be separated from the ionic liquid product during the final solvent extraction and evaporation stages.
Q: What are the purity specifications achievable with this synthesis route?
A: The patented process consistently achieves product purity higher than 99.0%, with specific control over critical impurities such as halogen residues (≤5ppm) and metal ions like potassium or ammonium (≤200ppm).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazole Acetate Ionic Liquid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN110294712B can be reliably replicated at the volumes your business demands. We understand that high-purity imidazole acetate ionic liquids are not just commodities but critical enablers for advanced applications, which is why our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications including halide and metal ion limits. Our commitment to quality assurance means that every batch is scrutinized to ensure it meets the exacting standards required for use in pharmaceutical intermediates, electronic chemicals, and other high-value sectors.
We invite you to collaborate with us to explore how this advanced synthesis technology can optimize your supply chain and enhance your product formulations. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to reach out to request specific COA data from our recent pilot runs and to discuss route feasibility assessments for your unique projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable ionic liquid supplier dedicated to delivering excellence in both product quality and service reliability.
