Advanced C-H Activation Strategy for Scalable Benzothiazinone Pharmaceutical Intermediates Manufacturing
Advanced C-H Activation Strategy for Scalable Benzothiazinone Pharmaceutical Intermediates Manufacturing
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN114181168A introduces a significant advancement in the preparation of N-(substituent)-2-substituted benzothiazine-4-ketones, a class of compounds renowned for their potential as COX-2 inhibitors and antihypertensive agents. This innovative methodology leverages a direct C-H activation strategy at the ortho-position of the sulfur atom, bypassing the need for pre-functionalized halides or harsh condensation conditions often seen in legacy processes. By utilizing 1-chloromethyl-4-fluoro-1,4-diazabicyclo[2.2.2]octane bis(tetrafluoroborate) salt, commonly known as Selectfluor, in conjunction with trifluoroacetic acid and acetic anhydride, the process achieves high efficiency and operational simplicity. For R&D directors and procurement specialists, this represents a pivotal shift towards greener, more cost-effective manufacturing of high-purity pharmaceutical intermediates that can be reliably sourced for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzothiazinone derivatives has relied heavily on the condensation of thiosalicylic acid with corresponding aldehydes and amines, typically requiring reflux conditions in toluene. This traditional approach suffers from significant drawbacks, including consistently low yields of the target product and the generation of pungent, noxious odors that pose safety and environmental hazards in a production facility. Furthermore, alternative literature methods involving N-substituted-2-mercaptobenzamide and alkynyl methyl propionate necessitate the use of potassium phosphate additives and prolonged heating at 110°C for up to 20 hours, which increases energy consumption and process time. The reliance on thiol-containing starting materials in these conventional routes not only complicates waste treatment due to sulfur emissions but also limits the substrate scope due to sensitivity issues. These inefficiencies create bottlenecks in the supply chain, leading to higher costs and longer lead times for acquiring high-purity intermediates needed for clinical trials and commercial API production.
The Novel Approach
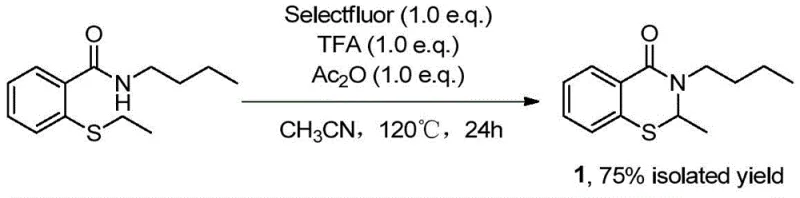
In stark contrast, the novel methodology disclosed in the patent utilizes a transition-metal-free C-H activation protocol that streamlines the construction of the benzothiazinone core. The reaction proceeds by sequentially adding N-(substituent)-2-sulfanylbenzamide, Selectfluor, acetic anhydride, and trifluoroacetic acid into acetonitrile, followed by vigorous stirring at 120°C for 24 hours. This single-pot transformation effectively activates the carbon-hydrogen bond ortho to the sulfur atom, facilitating simultaneous C-H cleavage and C-N bond construction without the need for external metal catalysts. The use of Selectfluor acts as a potent oxidant and fluorinating source that drives the cyclization forward with remarkable selectivity, resulting in isolated yields ranging from 60% to 75% across various substrates. This approach eliminates the malodorous byproducts associated with thiosalicylic acid routes and simplifies the purification process, making it an ideal candidate for reliable agrochemical intermediate supplier networks and pharmaceutical manufacturers seeking process intensification.
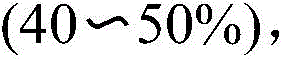
Mechanistic Insights into Selectfluor-Mediated C-H Activation and Cyclization
The core innovation of this synthesis lies in the unique mechanistic pathway enabled by the synergistic action of Selectfluor and trifluoroacetic acid (TFA). Mechanistically, the reaction initiates with the activation of the sulfanylbenzamide substrate, where the electrophilic nature of the Selectfluor reagent likely generates a reactive sulfur-centered radical or cationic intermediate. This activation renders the ortho-carbon position susceptible to nucleophilic attack or radical coupling, effectively breaking the inert C-H bond. The presence of acetic anhydride serves a dual purpose: it acts as a dehydrating agent to drive the equilibrium towards the cyclic ketone product and potentially acetylates intermediate species to stabilize the transition state. TFA enhances the acidity of the medium, protonating key intermediates and facilitating the final elimination steps to aromatize or stabilize the benzothiazine ring system. This intricate balance of reagents ensures that the cyclization occurs with high regioselectivity, minimizing the formation of regioisomers or over-oxidized byproducts that often plague C-H functionalization reactions.
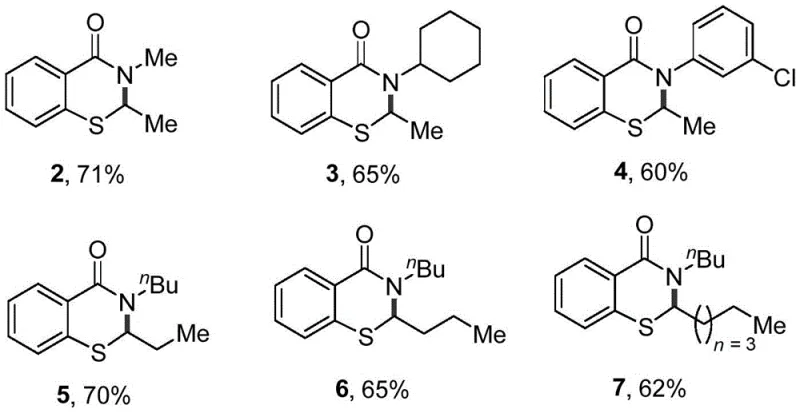
Furthermore, the substrate scope analysis reveals the robustness of this catalytic system against steric and electronic variations, which is crucial for impurity control in GMP manufacturing. As demonstrated in the experimental data, the reaction tolerates diverse N-substituents such as n-butyl, methyl, cyclohexyl, and even electron-deficient 3-chlorophenyl groups without significant loss in yield. Similarly, variations in the sulfur substituent, including ethyl, propyl, butyl, and n-hexyl chains, are well-accommodated, indicating that the steric bulk of the alkyl groups does not hinder the critical C-H activation step. This broad compatibility suggests that the impurity profile remains consistent and manageable across different analogues, allowing for standardized purification protocols like column chromatography to be effectively deployed. Such predictability in reaction outcome is highly valued by quality control teams aiming to maintain stringent purity specifications for complex polymer additives or fine chemical intermediates destined for sensitive biological applications.
How to Synthesize N-(substituent)-2-substituted benzothiazine-4-one Efficiently
To implement this high-efficiency synthetic route in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and thermal conditions to maximize yield and safety. The process begins with the careful charging of reagents into a sealed pressure vessel to contain the volatile acetonitrile solvent and acidic vapors generated during the exothermic activation phase. Maintaining the reaction temperature at exactly 120°C is critical, as deviations could lead to incomplete conversion or decomposition of the sensitive Selectfluor reagent. Following the 24-hour reaction period, the workup involves simple concentration followed by standard chromatographic separation, avoiding the need for complex aqueous extractions or heavy metal scavenging steps. Detailed standardized synthesis steps see the guide below for specific operational parameters and safety precautions regarding the handling of fluorinating agents.
- Charge a sealed tube with acetonitrile solvent, N-(substituent)-2-sulfanylbenzamide substrate, Selectfluor reagent, trifluoroacetic acid, and acetic anhydride in a 1: 1:1:1 molar ratio.
- Heat the reaction mixture to 120°C and stir vigorously for 24 hours to facilitate ortho-C-H bond activation and cyclization.
- Upon completion, concentrate the reaction liquid and purify the crude product via column chromatography to isolate the target benzothiazinone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this Selectfluor-mediated protocol offers substantial strategic advantages for procurement managers and supply chain heads tasked with optimizing raw material costs and ensuring continuity. The elimination of transition metal catalysts removes the necessity for expensive and time-consuming metal scavenging processes, which significantly reduces the overall cost of goods sold (COGS) and shortens the production cycle time. Additionally, the avoidance of malodorous thiosalicylic acid derivatives improves the working environment and reduces the regulatory burden associated with volatile organic compound (VOC) emissions and sulfur waste disposal. The use of commodity chemicals like acetic anhydride and acetonitrile ensures that the supply chain remains resilient against fluctuations in the availability of exotic reagents, thereby enhancing supply chain reliability for long-term contracts. This operational simplicity translates directly into cost reduction in pharmaceutical intermediates manufacturing, allowing companies to allocate resources towards further process optimization or scale-up activities.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by utilizing a one-pot reaction design that minimizes unit operations and solvent usage compared to multi-step traditional syntheses. By removing the requirement for precious metal catalysts, the method eliminates the capital expenditure associated with catalyst recovery systems and the recurring cost of catalyst replenishment. The high isolated yields reported, such as 75% for the n-butyl derivative, indicate excellent atom economy, meaning less raw material is wasted as byproduct, further driving down the effective price per kilogram of the active intermediate. These factors collectively contribute to a leaner manufacturing model that is highly attractive for cost-sensitive generic drug production or large-volume specialty chemical orders.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents like Selectfluor and acetic anhydride mitigates the risk of supply disruptions often caused by the degradation of sensitive organometallic reagents. Since the reaction does not require inert atmosphere techniques beyond standard sealing, it can be performed in standard glass-lined reactors available in most contract manufacturing organizations (CMOs), widening the pool of potential suppliers. This flexibility ensures reducing lead time for high-purity pharmaceutical intermediates, as production slots can be secured more easily without needing specialized equipment setups. Consequently, procurement teams can negotiate better terms and secure inventory buffers more effectively, safeguarding against market volatility.
- Scalability and Environmental Compliance: The protocol's inherent safety profile, characterized by the absence of pyrophoric reagents and toxic heavy metals, facilitates seamless commercial scale-up of complex pharmaceutical intermediates from gram to ton scale. The waste stream is primarily composed of organic salts and solvents that are easier to treat than heavy metal-contaminated effluents, aligning with increasingly strict global environmental regulations. This compliance advantage reduces the liability and permitting hurdles for manufacturing sites, ensuring uninterrupted production schedules. Moreover, the robust nature of the reaction conditions suggests that the process can withstand minor variations in scale-up parameters, providing a stable platform for sustained commercial supply.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzothiazinone synthesis technology, derived directly from the patent specifications and comparative data. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the specific advantages in yield, safety, and substrate tolerance that distinguish this method from prior art, providing clarity for decision-makers evaluating technology transfer opportunities.
Q: What are the advantages of this Selectfluor-mediated method over traditional thiosalicylic acid routes?
A: Traditional methods often involve refluxing thiosalicylic acid with aldehydes and amines, which can result in low yields and the generation of pungent, harmful odors. The novel Selectfluor-mediated C-H activation pathway described in CN114181168A operates under cleaner conditions with higher efficiency and avoids the handling of malodorous thiol-based starting materials typically associated with older synthetic protocols.
Q: What is the substrate scope for the N-substituent in this benzothiazinone synthesis?
A: The process demonstrates strong substrate adaptability, successfully accommodating diverse N-substituents including n-butyl, methyl, cyclohexyl, and 3-chlorophenyl groups. This versatility allows for the rapid generation of a library of benzothiazinone derivatives, which is critical for structure-activity relationship (SAR) studies in drug discovery programs targeting COX-2 inhibition or hypertension.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method utilizes readily available reagents like acetic anhydride and trifluoroacetic acid in acetonitrile, avoiding expensive transition metal catalysts that require complex removal steps. The operational simplicity and the use of standard heating conditions (120°C) suggest that the process can be scaled up effectively for industrial manufacturing of fine chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(substituent)-2-substituted benzothiazine-4-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner who can translate innovative patent technologies into commercial reality with speed and precision. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability regardless of volume. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify identity and assay levels against the highest industry standards. By leveraging our expertise in C-H activation chemistry and heterocyclic synthesis, we can offer you a secure source of high-quality benzothiazinone intermediates that accelerate your drug development timelines.
We invite you to collaborate with us to explore how this advanced synthetic route can optimize your specific project requirements and reduce your overall manufacturing footprint. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs, along with specific COA data and route feasibility assessments. Our experts are ready to discuss how we can support your supply chain goals with flexible manufacturing solutions and competitive pricing structures designed for long-term partnership success.
